1,2-Dioxetanedione

| |
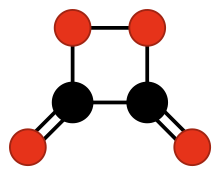
| |
| Names | |
|---|---|
| Preferred IUPAC name
1,2-Dioxetanedione | |
| Other names
Peroxyacid ester
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2O4 | |
| Molar mass | 88.018 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The
dimer of carbon dioxide.[1]
In ordinary conditions, it quickly decomposes to carbon dioxide (CO2) even at 180 K (−93.1 °C), but can be detected by mass spectrometry and other techniques.[2][3]
1,2-Dioxetanedione is an intermediate in the
biradical intermediate.[6]
Recently it has been found that a high-energy intermediate in one of these reactions (between
reducing agents are removed from the system, and the reactions are carried out in an inert atmosphere.[7]
See also
- 1,3-dioxetanedione
References
- ISBN 978-0-470-18720-3John Wiley & Sons.
- .
- S2CID 94708095.
- PMID 29493234.
- .
- PMID 19206238.
- .
External links
 Media related to 1,2-Dioxetanedione at Wikimedia Commons
Media related to 1,2-Dioxetanedione at Wikimedia Commons
