1,4-Dioxane
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,4-Dioxane | |||
| Systematic IUPAC name
1,4-Dioxacyclohexane | |||
| Other names
[1,4]Dioxane
p-Dioxane [6]-crown-2 Diethylene dioxide Diethylene ether Dioxane solvent | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 102551 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
ECHA InfoCard
|
100.004.239 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1165 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H8O2 | |||
| Molar mass | 88.106 g·mol−1 | ||
| Appearance | Colorless liquid[1] | ||
| Odor | Mild, diethyl ether-like[1] | ||
| Density | 1.033 g/mL | ||
| Melting point | 11.8 °C (53.2 °F; 284.9 K) | ||
| Boiling point | 101.1 °C (214.0 °F; 374.2 K) | ||
| Miscible | |||
| Vapor pressure | 29 mmHg (20 °C)[1] | ||
| −52.16·10−6 cm3/mol | |||
| Thermochemistry | |||
Std molar
entropy (S⦵298) |
196.6 J/K·mol | ||
Std enthalpy of (ΔfH⦵298)formation |
−354 kJ/mol | ||
Std enthalpy of (ΔcH⦵298)combustion |
−2363 kJ/mol | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Suspected human carcinogen[1] | ||
| GHS labelling: | |||
  
| |||
| Danger | |||
| H225, H302, H305, H315, H319, H332, H336, H351, H370, H372, H373 | |||
| P201, P202, P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P280, P281, P302+P352, P303+P361+P353, P304+P312, P304+P340, P305+P351+P338, P307+P311, P308+P313, P312, P314, P321, P332+P313, P337+P313, P362, P370+P378, P403+P233, P403+P235, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 12 °C (54 °F; 285 K) | ||
| 180 °C (356 °F; 453 K) | |||
Explosive limits
|
2.0–22%[1] | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
| ||
LC50 (median concentration)
|
| ||
LCLo (lowest published)
|
1000–3000 ppm (guinea pig, 3 hr) 12,022 ppm (cat, 7 hr) | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 100 ppm (360 mg/m3) [skin][1] | ||
REL (Recommended)
|
Ca C 1 ppm (3.6 mg/m,3) [30-minute][1] | ||
IDLH (Immediate danger) |
Ca [500 ppm][1] | ||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
1,4-Dioxane (/daɪˈɒkseɪn/) is a heterocyclic organic compound, classified as an ether. It is a colorless liquid with a faint sweet odor similar to that of diethyl ether. The compound is often called simply dioxane because the other dioxane isomers (1,2- and 1,3-) are rarely encountered.
Dioxane is used as a solvent for a variety of practical applications as well as in the laboratory, and also as a stabilizer for the transport of
Synthesis
Dioxane is produced by the acid-catalysed dehydration of diethylene glycol, which in turn is obtained from the hydrolysis of ethylene oxide.
In 1985, the global production capacity for dioxane was between 11,000 and 14,000 tons.[4] In 1990, the total U.S. production volume of dioxane was between 5,250 and 9,150 tons.[5]
Structure
The dioxane molecule is centrosymmetric, meaning that it adopts a
Uses
Trichloroethane transport
In the 1980s, most of the dioxane produced was used as a stabilizer for
As a solvent
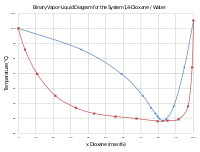
Dioxane is used in a variety of applications as a versatile
While diethyl ether is rather insoluble in water, dioxane is
The oxygen atoms are weakly
Dioxane produces coordination polymers by linking metal centers.[8] In this way, it is used to drive the Schlenk equilibrium, allowing the synthesis of dialkyl magnesium compounds.[4] Dimethylmagnesium is prepared in this manner:[9][10]
- 2 CH3MgBr + (C2H4O)2 → MgBr2(C2H4O)2 + (CH3)2Mg
Spectroscopy
Dioxane is used as an internal standard for
Toxicology
Safety
Dioxane has an
It tends to concentrate in the water and has little affinity for soil. It is resistant to abiotic degradation in the environment, and was formerly thought to also resist biodegradation. However, more recent studies since the 2000s have found that it can be biodegraded through a number of pathways, suggesting that bioremediation can be used to treat 1,4-dioxane contaminated water.[22][23]
Explosion hazard
Like some other ethers, dioxane combines with atmospheric oxygen upon prolonged exposure to air to form potentially explosive peroxides. Distillation of these mixtures is dangerous. Storage over metallic sodium could limit the risk of peroxide accumulation.
Environment
Dioxane has affected groundwater supplies in several areas. Dioxane at the level of 1 μg/L (~1 ppb) has been detected in many locations in the US.[5] In the U.S. state of New Hampshire, it had been found at 67 sites in 2010, ranging in concentration from 2 ppb to over 11,000 ppb. Thirty of these sites are solid waste landfills, most of which have been closed for years. In 2019, the Southern Environmental Law Center successfully sued Greensboro, North Carolina's Wastewater treatment after 1,4-Dioxane was found at 20 times above EPA safe levels in the Haw River.[24]
Cosmetics
As a byproduct of the ethoxylation process, a route to some ingredients found in cleansing and moisturizing products, dioxane can contaminate cosmetics and personal care products such as deodorants, perfumes, shampoos, toothpastes and mouthwashes.[25][26] The ethoxylation process makes the cleansing agents, such as sodium laureth sulfate and ammonium laureth sulfate, less abrasive and offers enhanced foaming characteristics. 1,4-Dioxane is found in small amounts in some cosmetics, a yet unregulated substance used in cosmetics in both China and the U.S.[27] Research has found the chemical in ethoxylated raw ingredients and in off-the-shelf cosmetic products. The Environmental Working Group (EWG) found that 97% of hair relaxers, 57% of baby soaps and 22 percent of all products in Skin Deep, their database for cosmetic products, are contaminated with 1,4-dioxane.[28]
Since 1979 the
On 9 December 2019, New York passed a bill to ban the sale of cosmetics with more than 10 ppm of 1,4-dioxane as of the end of 2022. The law will also prevent the sale of household cleaning and personal care products containing more than 2 ppm of 1,4-dioxane at the end of 2022.[31]
See also

References
- ^ a b c d e f g h NIOSH Pocket Guide to Chemical Hazards. "#0237". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Dioxane". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Wisconsin Department of Health Services (2013) 1,4-Dioxane Fact Sheet Archived 16 October 2020 at the Wayback Machine. Publication 00514. Accessed 2016-11-12.
- ^ ISBN 978-3527306732.
- ^ a b "1, 4-Dioxane Fact Sheet: Support Document" (PDF). OPPT Chemical Fact Sheets. United States Environmental Protection Agency. February 1995. Retrieved 14 May 2010.
- ISBN 3527305785, 9783527305780.
- .
- PMID 31328293.
- .
- .
- S2CID 34800494.
- ^ "International Chemical Safety Card". National Institute for Occupational Safety and Health. Archived from the original on 29 April 2005. Retrieved 6 February 2006.
- PMID 641607. Retrieved 26 March 2016.
- ^ "12th Report on Carcinogens". United States Department of Health and Human Services' National Toxicology Program. Archived from the original on 14 July 2014. Retrieved 11 July 2014.
- ^ IARC Monographs Volume 71 (PDF). International Agency for Research on Cancer. Retrieved 11 July 2014.
- ^ 1,4-Dioxane (1,4-Diethyleneoxide). Hazard Summary. U.S. Environmental Protection Agency. Created in April 1992; Revised in January 2000. Fact Sheet.
- ^ "Chemicals Known to the State to Cause Cancer or Reproductive Toxicity" (PDF). Office of Environmental Health Hazard Assessment. 2 April 2010. Archived from the original (PDF) on 24 May 2010. Retrieved 14 December 2013.
1,4-Dioxane CAS#123-91-1 (Listed 1 January 1988)
- PMID 18544906.
- S2CID 86811931.
- S2CID 45963495.
- ^ "Governor Cuomo Announces First in the Nation Drinking Water Standard for Emerging Contaminant 1,4-Dioxane | Governor Andrew M. Cuomo". Archived from the original on 29 October 2020. Retrieved 30 October 2020.
- .
- PMID 28625566.
- ^ "1,4-dioxane in Greensboro | Haw River Assembly". 18 November 2020. Retrieved 13 May 2022.
- ^ Tenth Report on Carcinogens Archived 1 November 2004 at the Wayback Machine. U.S. Department of Health and Human Services, Public Health Service, National Toxicology Program, December 2002.
- ^ "Chemical Encyclopedia: 1,4-dioxane". Healthy Child Healthy World. Archived from the original on 29 November 2009. Retrieved 14 December 2009.
- ^ "Watchdog issues inspection results on Johnson & Johnson". China Daily. Xinhua. 21 March 2009. Retrieved 14 May 2010.
- ^ "The Dangers of 1,4-Dioxane and How to Avoid It". Aspen Clean. Aspen Clean. 11 February 2020. Retrieved 17 December 2020.
- ^ PMID 11417628.
- ^ FDA/CFSAN--Cosmetics Handbook Part 3: Cosmetic Product-Related Regulatory Requirements and Health Hazard Issues. Prohibited Ingredients and other Hazardous Substances: 9. Dioxane Web.archive.org
- ^ "New York restricts 1,4-dioxane in cleaning and personal care products". Cen.acs.org. Retrieved 13 November 2021.



