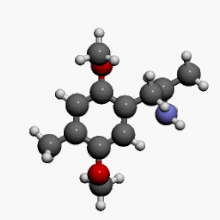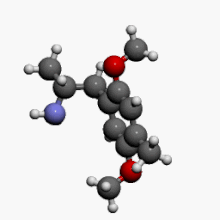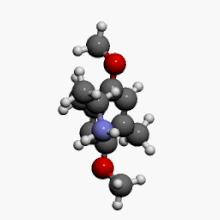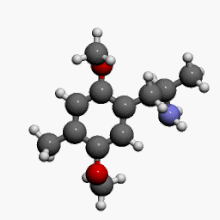
2,5-Dimethoxy-4-methylamphetamine

| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
1-(2,5-Dimethoxy-4-methylphenyl)propan-2-amine | |
| Other names
2,5-Dimethoxy-4-methylamphetamine
| |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C12H19NO2 | |
| Molar mass | 209.289 g·mol−1 |
| Melting point | 61 °C (142 °F; 334 K) |
| Pharmacology | |
| Legal status |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2,5-Dimethoxy-4-methylamphetamine (DOM; known as STP, standing for "Serenity, Tranquility and Peace") is a psychedelic and a substituted amphetamine. It was first synthesized by Alexander Shulgin, and later reported in his book PiHKAL: A Chemical Love Story. DOM is classified as a Schedule I substance in the United States, and is similarly controlled in other parts of the world. Internationally, it is a Schedule I drug under the Convention on Psychotropic Substances.[2] It is generally taken orally.
History
STP was first synthesized and tested in 1963 by Alexander Shulgin, who was investigating the effect of 4-position substitutions on psychedelic amphetamines.[3]
In mid-1967, tablets containing 20 mg (later 10 mg) of STP were widely distributed in the Haight-Ashbury District of San Francisco under the name of STP, having been manufactured by underground chemists Owsley Stanley and Tim Scully. This short-lived appearance of STP on the black market proved disastrous for several reasons. First, the tablets contained an excessively high dose of the chemical. This, combined with DOM's slow onset of action (which encouraged some users, familiar with drugs that have quicker onsets, such as LSD, to re-dose) and its remarkably long duration, caused many users to panic and sent some to the emergency room. Second, treatment of such overdoses was complicated by the fact that no one at the time knew that the tablets called STP were, in fact, DOM.
Effects
Effects of this drug include substantial perceptual changes such as
Pharmacology
DOM is a selective
Analogues and derivatives

The 2,6-dimethoxy positional isomer of DOM, known as
Toxicity
Very little is known about the toxicity of DOM. According to Alexander Shulgin, the effects of DOM typically last 14 to 20 hours, though other clinical trials indicate a duration of 7 to 8 hours.[4]
Legal status
Canada
Listed as a Schedule 1, as it is an analogue of amphetamine.
United States
DOM is Schedule I in the United States. This means it is illegal to manufacture, buy, possess, or distribute (make, trade, own or give) without a DEA license.
Australia
DOM is schedule 9 under the Australia Poisons standard.[8] A schedule 9 substance is a "Substances which may be abused or misused, the manufacture, possession, sale or use of which should be prohibited by law except when required for medical or scientific research, or for analytical, teaching or training purposes with approval of Commonwealth and/or State or Territory Health Authorities."[8]
United Kingdom
DOM is a
See also
- 2,5-Dimethoxy-4-Substituted Amphetamines
References
- ^ Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- ^ "Green List: List of Psychotropic Substances Under International Control" (PDF) (23rd ed.). International Narcotics Control Board. August 2003. p. 4. Archived from the original (PDF) on 19 December 2013. Retrieved 22 February 2014.
- ISBN 978-0-9630096-0-9.
- ^ S2CID 24065654.
- PMID 2843634.
- S2CID 36463979.
- OCLC 703618147. Retrieved 28 February 2012.
- ^ a b Poison Standard https://www.comlaw.gov.au/Details/F2015L01534/Html/Text#_Toc420496379 Archived 2015-12-22 at the Wayback Machine
