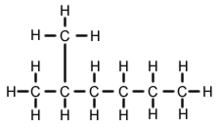
2-Methylhexane

| |
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methylhexane[1] | |
| Identifiers | |
3D model (
JSmol ) |
|
| 1696856 | |
| ChemSpider | |
ECHA InfoCard
|
100.008.847 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 3295 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C7H16 | |
| Molar mass | 100.205 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 0.679 g cm−3 |
| Melting point | −119.0 to −117.8 °C; −182.3 to −180.1 °F; 154.1 to 155.3 K |
| Boiling point | 89.6 to 90.6 °C; 193.2 to 195.0 °F; 362.7 to 363.7 K |
| Vapor pressure | 15.7 kPa (at 37.7 °C) |
Henry's law
constant (kH) |
19 nmol Pa−1 kg−1 |
| -86.24·10−6 cm3/mol | |
Refractive index (nD)
|
1.384 |
| Thermochemistry | |
Heat capacity (C)
|
222.92 J K−1 mol−1 |
Std molar
entropy (S⦵298) |
323.34 J K−1 mol−1 |
Std enthalpy of (ΔfH⦵298)formation |
−231.1–−228.5 kJ mol−1 |
Std enthalpy of (ΔcH⦵298)combustion |
−4.8127–−4.8103 MJ mol−1 |
| Hazards | |
| GHS labelling: | |
   
| |
| Danger | |
| H225, H304, H315, H336, H410 | |
| P210, P261, P273, P301+P310, P331 | |
| NFPA 704 (fire diamond) | |
| Flash point | −1 °C (30 °F; 272 K) |
| 280 °C (536 °F; 553 K) | |
Explosive limits
|
1–7% |
| Related compounds | |
Related alkanes
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2-Methylhexane (
Being an alkane, 2-methylhexane is insoluble in water, but is soluble in many organic solvents, such as alcohols and ether. However, 2-methylhexane is more commonly considered as a solvent itself. Therefore, even though it is present in many commercially available heptane products, it is not considered as a destructive impurity, as heptane is usually used as a solvent. Nevertheless, by concise processes of distillation and refining, it is possible to separate 2-methylhexane from n-heptane.
Within a group of isomers, those with more
Compared to n-heptane, 2-methylhexane also has lower melting and boiling points. A lower density of liquid is found in 2-Methylhexane than heptane.
On the NFPA 704 scale, 2-methylhexane is listed as a reactivity level-0 chemical, along with various other alkanes. In fact, most alkanes are unreactive except in extreme conditions, such as combustion or strong sunlight. At the presence of oxygen and flame, 2-methylhexane, like heptane, combusts mostly completely into water and carbon dioxide. With UV-light and mixed with halogens in solvents, usually bromine in 1,1,1-trichloroethane, a substitution reaction occurs.
See also
References
- ^ "2-METHYLHEXANE - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 26 March 2005. Identification and Related Records. Retrieved 6 March 2012.
- "2-Methylhexane". chemexper.com. Retrieved 22 November 2011.
- "Material Safety Data Sheet" (PDF). ChemADVISOR. Archived from the original (PDF) on 21 March 2006. Retrieved 22 November 2011.
- "Isoheptane". INCHEM: Chemical Safety Information from Intergovernmental Organizations. International Programme on Chemical Safety. October 2002. Retrieved 22 November 2011.

