25CN-NBOH
 | |
| Clinical data | |
|---|---|
| Other names | NBOH-2C-CN |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
25CN-NBOH (sometimes also referred to as NBOH-2C-CN)[1] is a compound indirectly derived from the phenethylamine series of hallucinogens, which was discovered in 2014 at the University of Copenhagen.[2] This compound is notable as one of the most selective agonist ligands for the 5-HT2A receptor yet discovered, with a pKi of 8.88 at the human 5-HT2A receptor and with 100x selectivity for 5-HT2A over 5-HT2C, and 46x selectivity for 5-HT2A over 5-HT2B.[3][4][5][6] A tritiated version of 25CN-NBOH has also been accessed and used for more detailed investigations of the binding to 5-HT2 receptors and autoradiography.[7]
Structure
The structure of 25CN-NBOH in complex with an engineered Gαq heterotrimer of the 5-HT2AR has been determined by cryoelectron microscopy (cryo-EM), showing a distinct binding mode when compared to LSD.[8]
Synthesis
25CN-NBOH is readily available from 2C-H in 57% over 4 steps.[9]
Animal studies
25CN-NBOH was found to partially substitute for DOI but was considerably weaker at inducing a head-twitch response in mice.[10][11] Another in vivo evaluation of 25CN-NBOH concluded that "Given its distinct in vitro selectivity for 5-HT2A over non 5-HT2 receptors and its behavioral dynamics, 25CN-NBOH appears to be a powerful tool for dissection of receptor-specific cortical circuit dynamics, including 5-HT2A related psychoactivity."[12]
25CN-NBOH induces the Head Twitch Response (HTR) also refererred to as "wet dog shakes" in rodents and the cortical fingerprint of serotonin-2A-receptor-mediated shaking behavior has been investigated in detail.[13]
Additional in vivo investigations with this ligand has emerged.[14][15][16][17][18][19][20][21] Chronic administration in mice lead to desensitization of the 5-HT2AR (measured via HTR) and increased startle amplitude[22] whereas it does not effect reversal learning in mice.[23] 25CN-NBOH was shown to increase the production of CTGF in chondrocytes.[24] In rats, 25CN-NBOH induce a reduction in conditioned fear that was countered by pretreatment with 5-HT2AR inverse agonist MDL100907.[25]
A bioanalytical method for the detection of 25CN-NBOH has been developed.[26]
Literature
A review covering the literature up to 2020 was published in 2021.[27]
Related compounds
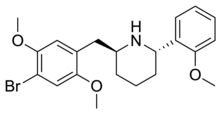
The tendency of the 4-cyano substitution to confer high 5-HT2A selectivity had previously been observed with DOCN,
Legality
Hungary
25CN-NBOH is illegal in Hungary.[30]
United Kingdom
This substance is a
See also
References
- ^ "25CN-NBOH". Chemical Probes.
- ^ "CNS Medicinal Chemistry in the Kristensen Group". Department of Drug Design and Pharmacology. University of Copenhagen. 25 March 2019.
- PMID 24397362.
- PMID 28360333.
- ^ Hansen M (2011). Design and Synthesis of Selective Serotonin Receptor Agonists for Positron Emission Tomography Imaging of the Brain (Ph.D. thesis). University of Copenhagen.
- PMID 27020041.
- S2CID 215802376.
- PMID 32946782.
- ISSN 2509-9396.
- PMID 25224567.
- PMID 30448990.
- PMID 29467649.
- PMID 37298417.
- PMID 26787771.
- S2CID 207831116.
- PMID 31540405.
- PMID 32814148.
- S2CID 224775595.
- PMID 33343373.
- S2CID 246548173.
- PMID 35954229.
- S2CID 225052555.
- S2CID 231953516.
- PMID 29145495.
- PMID 34228806.
- S2CID 232431316.
- S2CID 236157499.
- S2CID 20150858.
- PMID 23336049.
- ^ "A Magyarországon megjelent, a Kábítószer és Kábítószer-függőség Európai Megfigyelő Központjának Korai Jelzőrendszerébe (EMCDDA EWS) 2005 óta bejelentett ellenőrzött anyagok büntetőjogi vonatkozású besorolása" [The criminal law-related classification of controlled substances announced in 2005 in the Early Warning System of the European Monitoring Center for Drugs and Drug Addiction (EMCDDA EWS) in Hungary] (PDF) (in Hungarian). September 2015.
- ^ "The Misuse of Drugs Act 1971 (Ketamine etc.) (Amendment) Order 2014". UK Statutory Instruments 2014 No. 1106. www.legislation.gov.uk.
