2C-Se
Source: Wikipedia, the free encyclopedia.
(Redirected from
2C-SE
)
| |
| Names | |
|---|---|
| Preferred IUPAC name
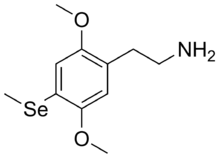
2-[2,5-Dimethoxy-4-(methylselanyl)phenyl]ethan-1-amine | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C11H17NO2Se | |
| Molar mass | 274.218 g/mol |
| Melting point | 240–241 °C (464–466 °F; 513–514 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chemical compound
2C-Se is a lesser-known
psychedelic drug. It was originally named by Alexander Shulgin as described in his book PiHKAL (Phenethylamines i Have Known And Loved). Shulgin considered 2C-Se to be around three times the potency of mescaline,[1][2][3][4] but was too concerned about toxicity to test it extensively, though he considered it noteworthy as the only psychedelic drug to contain a selenium atom.[5]
See also
- Phenethylamine
- Psychedelics, dissociatives and deliriants
References
- ISBN 978-0-9630096-3-0.
- ISBN 978-0124339514
- ISBN 978-3-03788-700-4.
- ^ Jacob P, Shulgin AT. Structure-activity relationships of the classic hallucinogens and their analogs. In Hallucinogens: An update. pp 74–91. NIDA Research Monograph 146. NIH Publication 94-3872, 1994.
- OCLC 25627628.
External links
(NMDAR
antagonists)
| Arylcyclo‐ hexylamines |
| ||||||
|---|---|---|---|---|---|---|---|
| Adamantanes | |||||||
| Diarylethylamines | |||||||
| Morphinans |
| ||||||
| Others |
(mAChR
antagonists)
- Atropine
- Benactyzine
- Benzatropine
- Benzydamine
- Biperiden
- BRN-1484501
- Brompheniramine
- BZ
- CAR-226,086
- CAR-301,060
- CAR-302,196
- CAR-302,282
- CAR-302,368
- CAR-302,537
- CAR-302,668
- Chloropyramine
- Chlorphenamine
- Clemastine
- CS-27349
- Cyclizine
- Cyproheptadine
- Dicycloverine
- Dimenhydrinate
- Diphenhydramine
- Ditran
- Doxylamine
- EA-3167
- EA-3443
- EA-3580
- EA-3834
- Flavoxate
- Hyoscyamine
- JB-318
- JB-336
- Meclozine
- Mepyramine
- Orphenadrine
- Oxybutynin
- Pheniramine
- Phenyltoloxamine
- Procyclidine
- Promethazine
- Scopolamine
- Tolterodine
- Trihexyphenidyl
- Tripelennamine
- Triprolidine
- WIN-2299
Natural | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Synthetic |
|
enhancers
(Mixed MOA)
- Aliphatic hydrocarbons
- Aromatic hydrocarbons
- Ethers
- Haloalkanes
- 2-EMSB
- Alazocine
- Bremazocine
- Butorphan
- Butorphanol
- Cyclazocine
- Cyclorphan
- Cyprenorphine
- Diprenorphine
- Enadoline
- Herkinorin
- Heroin
- HZ-2
- Ibogaine
- Ketazocine
- Levallorphan
- Levomethorphan
- Levorphanol
- LPK-26
- Metazocine
- Morphine
- Nalbuphine
- Nalmefene
- Nalorphine
- Noribogaine
- Oxilorphan
- Pentazocine
- Phenazocine
- Proxorphan
- Racemethorphan
- Racemorphan
- Salvinorin A
- Spiradoline
- Tifluadom
- U-50488
- U-69,593
- Xorphanol
This psychoactive drug-related article is a stub. You can help Wikipedia by expanding it. |
Retrieved from "https://en.wikipedia.org/w/index.php?title=2C-Se&oldid=1018386626"
