2C-T-7
| |
| |

| |
| Names | |
|---|---|
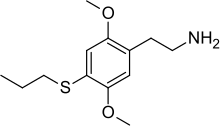
| Preferred IUPAC name
2-[2,5-Dimethoxy-4-(propylsulfanyl)phenyl]ethan-1-amine | |
| Other names
2,5-Dimethoxy-4-(propylsulfanyl)phenethylamine, 2,5-Dimethoxy-4-(propylthio)phenethylamine, Blue Mystic, Tweety-Bird Mescaline
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C13H21NO2S | |
| Molar mass | 255.38 g/mol |
| Melting point | 206 to 207 °C (403 to 405 °F; 479 to 480 K) |
| Pharmacology | |
| Legal status |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2C-T-7 is a
There has been little real research done on this chemical other than Shulgin's comments in PiHKAL and a few small animal studies mostly aimed at detecting metabolites.Pharmacology
The mechanism that produces the psychedelic and entactogenic effects of 2C-T-7 is most likely to result from action as a 5-HT2A serotonin receptor agonist in the brain, a mechanism of action shared by most currently-known hallucinogenic tryptamines and phenethylamines.[4] 2C-T-7 has structural and pharmacodynamic properties similar to those of 2C-T-2.
Effects
2C-T-7 is psychedelic.[5][6] In PiHKAL, Shulgin records that the hallucinations are unique, and that the chemical may cause muscle tension and an altered vocal quality.[7] Shulgin rated it as one of the "magical half-dozen" most important psychedelic phenethylamine compounds, together with Mescaline, 2C-B and 2C-T-2.[8]
Deaths
The
All of these known deaths of individuals under the influence of 2C-T-7, therefore, occurred in those known either to be intoxicated with potentially deadly[16] stimulants such as ephedrine or MDMA or after the individual insufflated an excessive amount of 2C-T-7—excessive being an amount greater than necessary to induce the full range of the drug's effects, such as the reported 35 mg insufflated dose taken by the individual who died in the fall of 2000. This reported dose was characterized as "excessive" by the US DEA.
Legal status
Around the year 2000, 2C-T-7 began to change from an obscure chemical to a drug used at parties and clubs in North America and Europe as it became available through a number of grey-market commercial vendors. This aroused the attention of the authorities, and many countries have since scheduled the chemical.
Germany
2C-T-7 is scheduled in Germany. (BTMG)
Australia
In Australia,
Canada
As of October 31, 2016, 2C-T-7 is a controlled substance (Schedule III) in Canada.[17]
China
As of October 2015 2C-T-7 is a controlled substance in China.[18]
The Netherlands
The Netherlands was the first country in the world to ban 2C-T-7, after being sold in smartshops for a short period. After 2C-T-2 was first banned, 2C-T-7 quickly appeared on the market, but was soon banned as well. 2C-T-7 is a list I drug of the Opium Law.
Sweden
Schedule I in Sweden.[19] 2C-T-7 was first classified as "health hazard" under the act Lagen om förbud mot vissa hälsofarliga varor (translated Act on the Prohibition of Certain Goods Dangerous to Health) as of April 1, 1999, under SFS 1999:58[20] that made it illegal to sell or possess.
United Kingdom
In 1999,
United States
On September 20, 2002, 2C-T-7 was classified as a Schedule I substance in the United States by an emergency ruling by the DEA. On March 18, 2004, the DEA published a Final Rule in the Federal Register permanently placing 2C-T-7 in Schedule I. (69 FR 12794).[21][22][23]
References
- ^ Alexander Shulgin. "PIHKAL #43".
- ^ Platoni, Kara. (May 1, 2002). "2C-T-7's Bad Trip". East Bay Express.
In 1999 it made its first commercial appearance in the Netherlands' drug-dealing smart shops in both tablet and powder form. It was given the street name "Blue Mystic," perhaps in order to differentiate it from its chemical cousin, another Shulgin creation named 2C-T-2
- ^ O'Connell, Chris (August 19, 2002). "A psychedelic summer". Newsweek.
- S2CID 8108926.
- ^ Hardison, Casey (2000). "An Amateur Qualitative Study of 48 2C-T-7 Subjective Bioassays". maps.org. Bulletin of the Multidisciplinary Association for Psychedelic Studies MAPS. Retrieved October 30, 2023.
- ^ "Erowid 2C-T-7 Vault: Sulfurous Samadhi: Stolaroff's & Well's Study". erowid.org. February 6, 2001. Retrieved October 30, 2023.
- ^ Shulgin, Alexander (June 28, 2001). "2C-T-7". Ask Dr Shulgin. Centre for Cognitive Liberty and Ethics (COLE). Retrieved August 28, 2009.
- ^ Shulgin (1990). ""PIHKAL" - The Chemical Story". www.erowid.org. Retrieved October 30, 2023.
- Partnership for a Drug-Free America. "2C-B, 2C-T-7". Archived from the originalon October 19, 2006. Retrieved October 4, 2006.
- ^ Curtis, B. "Postmortem Identification and Quantitation of 2,5-Dimethoxy-4-n-propylthiophenethylamine Using GC-MSD and GC-NPD". Journal of Analytical Toxicology 27.7 (2003): 493–98: "This compound was initially identified from a routine screening procedure in postmortem urine from a 20-year-old male that died in a local emergency room after reportedly insufflating 35 mg."
- ^ Platoni, Chris. "A psychedelic summer". East Bay Express, May 1, 2002: "In the same month, Joshua Robbins, a seventeen-year-old from Cordova, Tennessee, died after snorting between thirty and thirty-five milligrams of 2C-T-7, not long after taking several other stimulant drugs. According to Rolling Stone, which ran an article on Robbins' death, in the twelve hours before he died Robbins also had consumed Ecstasy, nitrous oxide, and a 'mini-thin' containing ephedrine and guaifenisen."
- ^ "In the fall of 2000, a young healthy male died following snorting an excessive amount of 2C-T-7. Since this initial 2C-T-7-related death, two additional deaths reported in April 2001 have been linked to 2C-T-7. These two deaths resulted from the co-abuse of 2C-T-7 with MDMA." http://www.deadiversion.usdoj.gov/drugs_concern/2ct7.htm Archived October 20, 2008, at the Wayback Machine
- ^ "The New (Legal) Killer Drug". Rolling Stone, January 10, 2002, issue 888: 44–49.
- ^ 21 USC §813
- ^ Ken Lilly (2002). "The new (hip) drug hysteria: a journey into rolling stone's abandonment of journalistic ethics". Disinformation. Archived from the original on September 8, 2008. Retrieved November 10, 2008.
- S2CID 245067669.
- ^ Gazette, Government of Canada, Public Works and Government Services Canada, Public Services and Procurement Canada, Integrated Services Branch, Canada (May 4, 2016). "Canada Gazette – Regulations Amending the Food and Drug Regulations (Part J — 2C-phenethylamines)". gazette.gc.ca.
- ^ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). China Food and Drug Administration. September 27, 2015. Archived from the original on October 1, 2015. Retrieved October 1, 2015.
- ^ "Läkemedelsverkets författningssamling" (PDF).
- ^ "Förordning (1999:58) om förbud mot vissa hälsofarliga varor - Karnov Open". notisum.se. Archived from the original on October 4, 2013. Retrieved September 15, 2013.
- ^ DEA. "Micgrogram Bulletin Jan 2004". Archived from the original on February 12, 2007. Retrieved December 19, 2006.
- ^ U.S. Department of Justice. "2C-T-7 Fast Facts" (PDF).
- ^ "List of Schedule 1 drugs on the DEA Office of Diversion Control website". Archived from the original on August 27, 2009. Retrieved July 7, 2008.
