3C-like protease
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
| Peptidase C30, Coronavirus endopeptidase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | Peptidase_C30 | ||||||||
SCOP2 | d1q2wb1 / SCOPe / SUPFAM | ||||||||
| |||||||||
The 3C-like protease (3CLpro) or main protease (Mpro), formally known as C30 endopeptidase or 3-chymotrypsin-like protease,
The
Function
The 3C-like protease is able to
TSAVLQ-SGFRK-NH2 and SGVTFQ-GKFKK are the two peptides corresponding to the two self-cleavage sites of the SARS 3C-like proteinase
The protease is important in the processing of the coronavirus
| Position | Substrate preference |
|---|---|
| P5 | No strong preference |
| P4 | Small hydrophobic residues
|
| P3 | Positively charged residue |
| P2 | High hydrophobicity and absence of beta-branch |
| P1 | Glutamine |
| P1' | Small residues |
| P2' | Small residues |
| P3' | No strong preference |
Nomenclature
Alternative names provided by the EC include 3CLpro, 3C-like protease, coronavirus 3C-like protease, Mpro, SARS 3C-like protease, SARS coronavirus 3CL protease, SARS coronavirus main peptidase, SARS coronavirus main protease, SARS-CoV 3CLpro enzyme, SARS-CoV main protease, SARS-CoV Mpro and severe acute respiratory syndrome coronavirus main protease.
As a treatment target

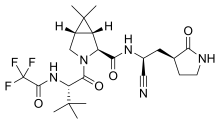
The protease 3CLpro is used as a
A number of
After clinical trials, in December 2021, the oral medication
The 3C-like protease inhibitor ensitrelvir received authorization to treat COVID-19 in Japan in 2022.[27][28]
In 2022, an ultralarge virtual screening campaign of 235 million molecules was able to identify a novel broad-spectrum inhibitor targeting the main protease of several coronaviruses. It is unusually not a peptidomimetic.[29]
Other 3C(-like) proteases
3C-like proteases (3C(L)pro) are widely found in
- This entry is the coronavirus 3CLpro.
- Picornaviridae have a picornavirus 3Cpro (EC 3.4.22.28; InterPro: IPR000199; MEROPS C03). This is the earliest-studied family. Examples include the ones found in poliovirus and in rhinovirus (both are members of genus Enterovirus).
- Norwalk virus.
Additional members are known from Potyviridae and non-Coronaviridae Nidovirales.[31]
See also
- 3CLpro-1
- Carmofur
- COVID Moonshot
- Ebselen
- EDP-235
- GC376
- GRL-0617
- Nirmatrelvir
- Olgotrelvir
- RAY1216
- Rupintrivir
- SIM0417
- Theaflavin digallate
- Tollovir
- Y180
- Tetrahydrocannabutol
References
- ^ PMID 32321856.
- PMID 34502033.
- PMID 17605471.
- ^ PMID 14561748.
- PMID 18845442.
- PMID 25720466.
See section: Virion Structure.
- PMID 20167482.
- PMID 22073294.
- PMID 34029993.
- ^ "Pfizer begins dosing in Phase II/III trial of antiviral drug for Covid-19". Clinical Trials Arena. 2 September 2021.
- PMID 25039866.
- S2CID 220304661.
- PMID 1738140.
- PMID 12746549.
- PMID 31312413.
- PMID 26149721.
- PMID 32045235.
- PMID 32198291.
- PMID 33556871.
- PMID 32022370.
- PMID 32226821.
- PMID 21936817.
- ^ "First-In-Human Study To Evaluate Safety, Tolerability, And Pharmacokinetics Following Single Ascending And Multiple Ascending Doses of PF-07304814 In Hospitalized Participants With COVID-19". Clinical Trials. 24 June 2021. Retrieved 3 July 2021.
- ^ Fact sheet for healthcare providers: Emergency Use Authorization for Paxlovid (PDF) (Technical report). Pfizer. 22 December 2021. LAB-1492-0.8. Archived from the original on 23 December 2021.
- ^ "Pfizer Receives U.S. FDA Emergency Use Authorization for Novel COVID-19 Oral Antiviral Treatment" (Press release). Pfizer. 22 December 2021. Archived from the original on 22 December 2021. Retrieved 22 December 2021 – via Business Wire.
- ^ "FDA Approves First Oral Antiviral for Treatment of COVID-19 in Adults". U.S. Food and Drug Administration (FDA) (Press release). 26 May 2023. Retrieved 26 May 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "Xocova (Ensitrelvir Fumaric Acid) Tablets 125mg Approved in Japan for the Treatment of SARS-CoV-2 Infection, under the Emergency Regulatory Approval System". Shionogi (Press release). 22 November 2022. Retrieved 28 November 2022.
- ^ Lenharo, Mariana (18 October 2023). "New Pill Helps COVID Smell and Taste Loss Fade Quickly". Scientific American. Retrieved 28 October 2023.
- PMID 35142215.
- PMID 22915796.
- PMID 12502857.
Further reading
- Chuck CP, Chow HF, Wan DC, Wong KB (2 November 2011). "Profiling of substrate specificities of 3C-like proteases from group 1, 2a, 2b, and 3 coronaviruses". PLOS ONE. 6 (11): e27228. PMID 22073294.
External links
- SARS+coronavirus+main+proteinase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Peptidase C30/C16 in coronavirus, InterPro: IPR013016. The MEROPS C16 one is the "papain-like" PL-PRO.

