5-Methylcytosine

| |
| Names | |
|---|---|
| Preferred IUPAC name
4-Amino-5-methylpyrimidin-2(1H)-one | |
| Identifiers | |
3D model (
JSmol ) |
|
| 3DMet | |
| 120387 | |
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.008.236 |
| EC Number |
|
| KEGG | |
| MeSH | 5-Methylcytosine |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C5H7N3O | |
| Molar mass | 125.131 g·mol−1 |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H317, H319 | |
| P261, P264, P272, P280, P302+P352, P305+P351+P338, P321, P333+P313, P337+P313, P363, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
5-Methylcytosine is a
In 5-methylcytosine, a methyl group is attached to the 5th atom in the 6-atom ring, counting counterclockwise from the NH-bonded nitrogen at the six o'clock position. This methyl group distinguishes 5-methylcytosine from cytosine.
Discovery
While trying to isolate the bacterial
In vivo
The function of this chemical varies significantly among species:[8]
- In bacteria, 5-methylcytosine can be found at a variety of sites, and is often used as a marker to protect DNA from being cut by native methylation-sensitive restriction enzymes.
- In plants, 5-methylcytosine occurs at CpG, CpHpG and CpHpH sequences (where H = A, C or T).
- In fungi and animals, 5-methylcytosine predominantly occurs at CpG dinucleotides. Most eukaryotes methylate only a small percentage of these sites, but 70-80% of CpG cytosines are methylated in vertebrates. In mammalian cells, clusters of CpG at the 5' ends of genes are termed CpG islands.[9] 1% of all mammalian DNA is 5mC.[10]
While spontaneous
In vitro
The NH2 group can be removed (deamination) from 5-methylcytosine to form thymine with use of reagents such as nitrous acid; cytosine deaminates to uracil (U) under similar conditions.[citation needed]
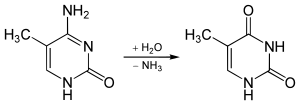
5-methylcytosine is resistant to deamination by bisulfite treatment, which deaminates cytosine residues. This property is often exploited to analyze DNA cytosine methylation patterns with bisulfite sequencing.[13]
Addition and regulation with DNMTs (Eukaryotes)
5mC marks are placed on genomic DNA via DNA methyltransferases (DNMTs). There are 5 DNMTs in humans: DNMT1, DNMT2, DNMT3A, DNMT3B, and DNMT3L, and in algae and fungi 3 more are present (DNMT4, DNMT5, and DNMT6).[14] DNMT1 contains the replication foci targeting sequence (RFTS) and the CXXC domain which catalyze the addition of 5mC marks. RFTS directs DNMT1 to loci of DNA replication to assist in the maintenance of 5mC on daughter strands during DNA replication, whereas CXXC contains a zinc finger domain for de novo addition of methylation to the DNA.[15] DNMT1 was found to be the predominant DNA methyltransferase in all human tissue.[16] Primarily, DNMT3A and DNMT3B are responsible for de novo methylation, and DNMT1 maintains the 5mC mark after replication.[1] DNMTs can interact with each other to increase methylating capability. For example, 2 DNMT3L can form a complex with 2 DNMT3A to improve interactions with the DNA, facilitating the methylation.[17] Changes in the expression of DNMT results in aberrant methylation. Overexpression produces increased methylation, whereas disruption of the enzyme decreased levels of methylation.[16]

The mechanism of the addition is as follows: first a cysteine residue on the DNMT's PCQ motif creates a nucleophillic attack at carbon 6 on the cytosine nucleotide that is to be methylated. S-Adenosylmethionine then donates a methyl group to carbon 5. A base in the DNMT enzyme deprotonates the residual hydrogen on carbon 5 restoring the double bond between carbon 5 and 6 in the ring, producing the 5-methylcytosine base pair.[15]
Demethylation
After a cytosine is methylated to 5mC, it can be reversed back to its initial state via multiple mechanisms. Passive DNA demethylation by dilution eliminates the mark gradually through replication by a lack of maintenance by DNMT. In active DNA demethylation, a series of oxidations converts it to
Role in humans
In cancer
In cancer, DNA can become both overly methylated, termed
As a biomarker of aging
"Epigenetic age" refers to the connection between chronological age and levels of DNA methylation in the genome.[26] Coupling the levels of DNA methylation, in specific sets of CpGs called "clock CpGs", with algorithms that regress the typical levels of collective genome-wide methylation at a given chronological age, allow for epigenetic age prediction. During youth (0–20 years old), changes in DNA methylation occur at a faster rate as development and growth progresses, and the changes begin to slow down at older ages. Multiple epigenetic age estimators exist. Horvath's clock measures a multi-tissue set of 353 CpGs, half of which positively correlate with age, and the other half negatively, to estimate the epigenetic age.[27] Hannum's clock utilizes adult blood samples to calculate age based on an orthogonal basis of 71 CpGs.[28] Levine's clock, known as DNAm PhenoAge, depends on 513 CpGs and surpasses the other age estimators in predicting mortality and lifespan, yet displays bias with non-blood tissues.[29] There are reports of age estimators with the methylation state of only one CpG in the gene ELOVL2.[30] Estimation of age allows for prediction lifespan through expectations of age related conditions that individuals may be subject to based on their 5mC methylation markers.[citation needed]
References
- ^ S2CID 3393814.
- ISBN 978-1130145373.
- .
- ^ Grosjean H (2009). Nucleic Acids Are Not Boring Long Polymers of Only Four Types of Nucleotides: A Guided Tour. Landes Bioscience.
- PMID 18107446.
- PMID 18873306.
- PMID 22344696.
- S2CID 10784130.
- S2CID 4236677.
- PMID 6262918.
- PMID 27588157.
- PMID 20800313.
- PMID 8065911.
- PMID 15689527.
- ^ S2CID 23370418.
- ^ PMID 10325416.
- PMID 17713477.
- PMID 23602153.
- PMID 21778364.
- PMID 25675246.
- PMID 21251613.
- ^ PMID 20495664.
- PMID 8653676.
- PMID 21376230.
- ISSN 1434-1948.
- S2CID 4709691.
- PMID 24138928.
- PMID 23177740.
- PMID 29676998.
- S2CID 8775590.
Literature
- Griffiths, Anthony J. F. (1999). An Introduction to genetic analysis. San Francisco: W.H. Freeman. Chapter 15: Gene Mutation. ISBN 0-7167-3520-2. (available online at the United States National Center for Biotechnology Information)
