Protein kinase B
Chr. 14 q32.32-32.33 | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
Chr. 19 q13.1-13.2 | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
Chr. 1 q43-44 | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
Protein kinase B (PKB), also known as Akt, is the collective name of a set of three
.Family members - Isoforms
There are three different genes that encode isoforms of Protein kinase B. These three genes are referred to as AKT1, AKT2, and AKT3 and encode the RAC alpha, beta, and gamma serine/threonine protein kinases respectively. The terms PKB and Akt may refer to the products of all three genes collectively, but sometimes are used to refer to PKB alpha and Akt1 alone.
The role of
Akt isoforms are overexpressed in a variety of human tumors, and, at the genomic level, are amplified in gastric adenocarcinomas (Akt1), ovarian (Akt2), pancreatic (Akt2) and breast (Akt2) cancers.[7][8]
Name
The name Akt does not refer to its function. The "Ak" in Akt refers to the AKR mouse strain that develops spontaneous thymic lymphomas. The "t" stands for 'thymoma'; the letter was added when a transforming retrovirus was isolated from the Ak mouse strain, which was termed "Akt-8". The authors state, "Stock A Strain k AKR mouse originally inbred in the laboratory of Dr. C. P. Rhoads by K. B. Rhoads at the Rockefeller Institute." When the oncogene encoded in this virus was discovered, it was termed v-Akt. Thus, the more recently identified human analogs were named accordingly.[9]
Regulation
Akt1 is involved in the PI3K/AKT/mTOR pathway and other signaling pathways.[10][citation needed]
Binding phospholipids
The Akt proteins possess a
Phosphorylation
Once correctly positioned at the membrane via binding of
Activated Akt isoforms can then go on to activate or deactivate their myriad substrates (e.g. mTOR) via their kinase activity.
Besides being a downstream effector of PI 3-kinases, Akt isoforms can also be activated in a PI 3-kinase-independent manner.[14] ACK1 or TNK2, a non-receptor tyrosine kinase, phosphorylates Akt at its tyrosine 176 residue, leading to its activation in PI 3-kinase-independent manner.[14] Studies have suggested that cAMP-elevating agents could also activate Akt through protein kinase A (PKA) in the presence of insulin.[15]
O-GlcNAcylation
Akt can be O-GlcNAcylated by OGT. O-GlcNAcylation of Akt is associated with a decrease in T308 phosphorylation.[16]
Ubiquitination
Akt1 is normally
Lipid phosphatases and PIP3
PI3K-dependent Akt1 activation can be regulated through the
Protein phosphatases
The phosphatases in the
Function
The Akt kinases regulate cellular survival
Cell survival
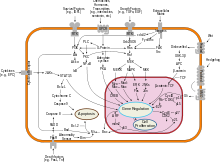
Akt kinases can promote growth factor-mediated cell survival both directly and indirectly. BAD is a pro-apoptotic protein of the Bcl-2 family. Akt1 can phosphorylate BAD on Ser136,[20] which makes BAD dissociate from the Bcl-2/Bcl-X complex and lose the pro-apoptotic function.[21] Akt1 can also activate NF-κB via regulating IκB kinase (IKK), thus result in transcription of pro-survival genes.[22]
Cell cycle
The Akt isoforms are known to play a role in the cell cycle. Under various circumstances, activation of Akt1 was shown to overcome cell cycle arrest in G1[23] and G2[24] phases. Moreover, activated Akt1 may enable proliferation and survival of cells that have sustained a potentially mutagenic impact and, therefore, may contribute to acquisition of mutations in other genes.
Metabolism
Akt2 is required for the insulin-induced translocation of glucose transporter 4 (
Lysosomal biogenesis and autophagy
Akt1 regulates TFEB, a master controller of lysosomal biogenesis,[25] by direct phosphorylation at serine 467.[26] Phosphorylated TFEB is excluded from the nucleus and less active.[26] Pharmacological inhibition of Akt promotes nuclear translocation of TFEB, lysosomal biogenesis and autophagy.[26]
Angiogenesis
Akt1 has also been implicated in angiogenesis and tumor development. Although deficiency of Akt1 in mice inhibited physiological angiogenesis, it enhanced pathological angiogenesis and tumor growth associated with matrix abnormalities in skin and blood vessels.[27][28]
Clinical relevance
Akt proteins are associated with tumor cell survival, proliferation, and invasiveness. The activation of Akt is also one of the most frequent alterations observed in human cancer and tumor cells. Tumor cells that have constantly active Akt may depend on Akt for survival.[29] Therefore, understanding the Akt proteins and their pathways is important for the creation of better therapies to treat cancer and tumor cells. A mosaic-activating mutation (c. 49G→A, p.Glu17Lys) in Akt1 is associated with the Proteus Syndrome, which causes overgrowth of skin, connective tissue, brain and other tissues.[30]
Akt inhibitors
Akt inhibitors may treat cancers such as neuroblastoma. Some Akt inhibitors have undergone clinical trials. In 2007 VQD-002 had a phase I trial.[31] In 2010 Perifosine reached phase II.[32] but it failed phase III in 2012.
Miltefosine is approved for leishmaniasis and under investigation for other indications including HIV.
Akt1 is now thought to be the "key" for cell entry by
MK-2206 reported phase 1 results for advanced solid tumors in 2011,[34] and subsequently has undergone numerous phase II studies for a wide variety of cancer types.[35]
In 2013
Ipatasertib is in phase II trials for breast cancer.[38]
Decreased Akt isoforms can cause deleterious effects
Akt isoform activation is associated with many malignancies; however, a research group from
Hyperactivation of Akt1 can cause deleterious effects
Two studies show that Akt1 is involved in Juvenile Granulosa Cell tumors (JGCT). In-frame duplications in the pleckstrin-homology domain (PHD) of the protein were found in more than 60% of JGCTs occurring in girls under 15 years of age. The JGCTs without duplications carried point mutations affecting highly conserved residues. The mutated proteins carrying the duplications displayed a non-wild-type subcellular distribution, with a marked enrichment at the plasma membrane. This led to a striking degree of Akt1 activation demonstrated by a strong phosphorylation level and corroborated by reporter assays.[40]
Analysis by RNA-Seq pinpointed a series of differentially expressed genes, involved in cytokine and hormone signaling and cell division-related processes. Further analyses pointed to a possible dedifferentiation process and suggested that most of the transcriptomic dysregulations might be mediated by a limited set of transcription factors perturbed by Akt1 activation. These results incriminate somatic mutations of Akt1 as major probably driver events in the pathogenesis of JGCTs.[41]
See also
- Akt/PKB signaling pathway
- Discovery and development of mTOR inhibitors
- PI3K/AKT/mTOR pathway
- Akt inhibitor
- PTEN
References
- PMID 20481595.
- PMID 18800763.
- PMID 11544177.
- PMID 197531.
- PMID 12843127.
- PMID 15046607.
- PMID 12191616.
- PMID 15134532.
- PMID 32850422.
- ^ Hsu, P.P. et al. The mTOR-regulated phosphoproteome reveals a mechanism of mTORC1-mediated inhibition of growth factor signaling. Science 332, 1317–1322 (2011). DOI: 10.1126/science.1199498
- S2CID 31186873.
- S2CID 45837814.
- S2CID 230319.
- ^ PMID 20333297.
- PMID 20412069.
- S2CID 18459576.
- PMID 23195959.
- ISBN 978-0-87893-106-4.
- PMID 15784165.
- ISBN 978-0-8153-3218-3.
- ISBN 978-0-7167-3136-8.
- PMID 16955703.
- PMID 10051603.
- PMID 12391152.
- S2CID 20353685.
- ^ PMID 28165011.
- PMID 16227992.
- PMID 16552185.
- ^ "Tumor Genetics; AKT Function and Oncogenic Activity" (PDF). Scientific Report. Fox Chase Cancer Center. 2005. Archived from the original (PDF) on 2010-06-04. Retrieved 2013-01-23.
- PMID 21793738.
- ^ "VioQuest Pharmaceuticals Announces Phase I/IIa Trial For Akt Inhibitor VQD-002". Apr 2007.
- PMID 20103671.
- PMID 23507869.
- "Scientists Reveal Novel Strategy for Stopping Herpes". Science News. Apr 5, 2013.
- PMID 22025163.
- ^ MK-2206 phase-2 trials
- ^ AKT inhibitor AZD5363 well tolerated, yielded partial response in patients with advanced solid tumors
- ^ "PARP/AKT Inhibitor Combination Active in Multiple Tumor Types. April 2016". Archived from the original on 2016-05-07. Retrieved 2016-04-20.
- S2CID 215793444.
- PMID 21884932.
- PMID 26137586.
- PMID 26362254.
Further reading
- Los M, Maddika S, Erb B, Schulze-Osthoff K (May 2009). "Switching Akt: from survival signaling to deadly response". BioEssays. 31 (5): 492–5. PMID 19319914.
- Quaresma AJ, Sievert R, Nickerson JA (April 2013). "Regulation of mRNA export by the PI3 kinase/AKT signal transduction pathway". Molecular Biology of the Cell. 24 (8): 1208–21. PMID 23427269.
External links
- Proto-Oncogene+Proteins+c-akt at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
