ATP synthase
| ATP Synthase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
ATP synthase is an enzyme that catalyzes the formation of the energy storage molecule adenosine triphosphate (ATP) using adenosine diphosphate (ADP) and inorganic phosphate (Pi). ATP synthase is a molecular machine. The overall reaction catalyzed by ATP synthase is:
- ADP + Pi + 2H+out ⇌ ATP + H2O + 2H+in
ATP synthase lies across a cellular membrane and forms an aperture that
.Eukaryotic ATP synthases are F-ATPases, running "in reverse" for an ATPase. This article deals mainly with this type. An F-ATPase consists of two main subunits, FO and F1, which has a rotational motor mechanism allowing for ATP production.[1][2]
Nomenclature
The F1 fraction derives its name from the term "Fraction 1" and FO (written as a subscript letter "o", not "zero") derives its name from being the binding fraction for oligomycin, a type of naturally derived antibiotic that is able to inhibit the FO unit of ATP synthase.[3][4] These functional regions consist of different protein subunits — refer to tables. This enzyme is used in synthesis of ATP through aerobic respiration.
Structure and function


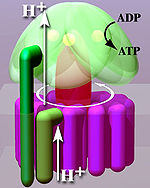
Located within the
F1 region
The F1 portion of ATP synthase is
Three other subunits catalyze the ATP synthesis. The other F1 subunits γ, δ, and ε are a part of a rotational motor mechanism (rotor/axle). The γ subunit allows β to go through conformational changes (i.e., closed, half open, and open states) that allow for ATP to be bound and released once synthesized. The F1 particle is large and can be seen in the transmission electron microscope by negative staining.[8] These are particles of 9 nm diameter that pepper the inner mitochondrial membrane.
| Subunit | Human Gene | Note |
|---|---|---|
| alpha | ATP5A1, ATPAF2 |
|
| beta | ATP5B, ATPAF1 | |
| gamma | ATP5C1 | |
| delta | ATP5D | Mitochondrial "delta" is bacterial/chloroplastic epsilon. |
| epsilon | ATP5E | Unique to mitochondria. |
| OSCP | ATP5O |
Called "delta" in bacterial and chloroplastic versions. |
FO region

FO is a water
In eukaryotes, mitochondrial FO forms membrane-bending dimers. These dimers self-arrange into long rows at the end of the
| Subunit | Human Gene |
|---|---|
| a | MT-ATP6 |
| b | ATP5F1 |
| c | ATP5G1, ATP5G2, ATP5G3 |
Binding model
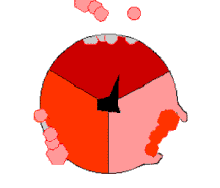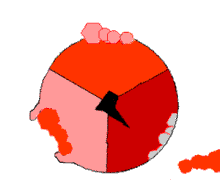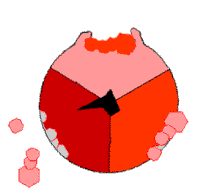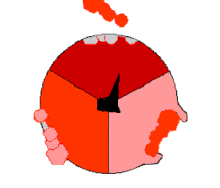

In the 1960s through the 1970s,
The crystal structure of the F1 showed alternating alpha and beta
The binding change mechanism involves the active site of a β subunit's cycling between three states.[14] In the "loose" state, ADP and phosphate enter the active site; in the adjacent diagram, this is shown in pink. The enzyme then undergoes a change in shape and forces these molecules together, with the active site in the resulting "tight" state (shown in red) binding the newly produced ATP molecule with very high affinity. Finally, the active site cycles back to the open state (orange), releasing ATP and binding more ADP and phosphate, ready for the next cycle of ATP production.[15]
Physiological role
Like other enzymes, the activity of F1FO ATP synthase is reversible. Large-enough quantities of ATP cause it to create a transmembrane
In respiring
Evolution
The evolution of ATP synthase is thought to have been modular whereby two functionally independent subunits became associated and gained new functionality.[16][17] This association appears to have occurred early in evolutionary history, because essentially the same structure and activity of ATP synthase enzymes are present in all kingdoms of life.[16] The F-ATP synthase displays high functional and mechanistic similarity to the V-ATPase.[18] However, whereas the F-ATP synthase generates ATP by utilising a proton gradient, the V-ATPase generates a proton gradient at the expense of ATP, generating pH values of as low as 1.[19]
The F1 region also shows significant similarity to hexameric
The H+
motor of the FO particle shows great functional similarity to the H+
motors that drive flagella.[18] Both feature a ring of many small alpha-helical proteins that rotate relative to nearby stationary proteins, using a H+
potential gradient as an energy source. This link is tenuous, however, as the overall structure of flagellar motors is far more complex than that of the FO particle and the ring with about 30 rotating proteins is far larger than the 10, 11, or 14 helical proteins in the FO complex. More recent structural data do however show that the ring and the stalk are structurally similar to the F1 particle.[21]
The modular evolution theory for the origin of ATP synthase suggests that two subunits with independent function, a DNA helicase with ATPase activity and a H+
motor, were able to bind, and the rotation of the motor drove the ATPase activity of the helicase in reverse.[16][22] This complex then evolved greater efficiency and eventually developed into today's intricate ATP synthases. Alternatively, the DNA helicase/H+
motor complex may have had H+
pump activity with the ATPase activity of the helicase driving the H+
motor in reverse.[16] This may have evolved to carry out the reverse reaction and act as an ATP synthase.[17][23][24]
Inhibitors
A variety of natural and synthetic inhibitors of ATP synthase have been discovered.[25] These have been used to probe the structure and mechanism of ATP synthase. Some may be of therapeutic use. There are several classes of ATP synthase inhibitors, including peptide inhibitors, polyphenolic phytochemicals, polyketides, organotin compounds, polyenic α-pyrone derivatives, cationic inhibitors, substrate analogs, amino acid modifiers, and other miscellaneous chemicals.[25] Some of the most commonly used ATP synthase inhibitors are oligomycin and DCCD.
In different organisms
Bacteria
E. coli ATP synthase is the simplest known form of ATP synthase, with 8 different subunit types.[11]
Bacterial F-ATPases can occasionally operate in reverse, turning them into an ATPase.[26] Some bacteria have no F-ATPase, using an A/V-type ATPase bidirectionally.[9]
Yeast
Yeast ATP synthase is one of the best-studied eukaryotic ATP synthases; and five F1, eight FO subunits, and seven associated proteins have been identified.[7] Most of these proteins have homologues in other eukaryotes.[27][28][29][30]
Plant
In plants, ATP synthase is also present in
Mammal
The ATP synthase isolated from bovine (Bos taurus) heart mitochondria is, in terms of biochemistry and structure, the best-characterized ATP synthase. Beef heart is used as a source for the enzyme because of the high concentration of mitochondria in cardiac muscle. Their genes have close homology to human ATP synthases.[32][33][34]
Human genes that encode components of ATP synthases:
Other eukaryotes
Eukaryotes belonging to some divergent lineages have very special organizations of the ATP synthase. A euglenozoa ATP synthase forms a dimer with a boomerang-shaped F1 head like other mitochondrial ATP synthases, but the FO subcomplex has many unique subunits. It uses cardiolipin. The inhibitory IF1 also binds differently, in a way shared with trypanosomatida.[35]
Archaea
Archaea do not generally have an F-ATPase. Instead, they synthesize ATP using the A-ATPase/synthase, a rotary machine structurally similar to the V-ATPase but mainly functioning as an ATP synthase.[26] Like the bacteria F-ATPase, it is believed to also function as an ATPase.[9]
LUCA and earlier
F-ATPase gene linkage and gene order are widely conserved across ancient prokaryote lineages, implying that this system already existed at a date before the last universal common ancestor, the LUCA.[36]
See also
- ATP10 protein required for the assembly of the FO sector of the mitochondrial ATPase complex.
- Chloroplast
- Electron transfer chain
- Flavoprotein
- Mitochondrion
- Oxidative phosphorylation
- P-ATPase
- Proton pump
- Rotating locomotion in living systems
- Transmembrane ATPase
- V-ATPase
References
- PMID 21524994.
- PMID 25839341.
- PMID 4223640.
- PMID 9874753.
- PMID 26439008.
- .
- ^ PMID 10838057.
- PMID 14195622.
- ^ PMID 24878343.
- PMID 15327958.
- ^ PMID 22312470.
- PMID 30760595.
- PMID 29074581.
- PMID 6214554.
- PMID 18515057.
- ^ PMID 8599633.
- ^ University of Illinois at Urbana–Champaign.
- ^ a b c "ATP Synthase". InterPro Database.
- PMID 16449553.
- S2CID 5765103.
- ^ PMID 26984495.
- ^ S2CID 4333137.
- S2CID 32559858.
- S2CID 25800744.
- ^ PMID 19052322.
- ^ PMID 26671611.
- PMID 10838056.
- PMID 17082766.
- PMID 10576729.
- PMID 25759169.
- PMID 29748256.
- S2CID 4275221.
- S2CID 23229994.
- S2CID 1266814.
- PMID 31738165.
- "Different from the rest". eLife. December 24, 2019.
- S2CID 234747849.
Further reading
- Nick Lane: The Vital Question: Energy, Evolution, and the Origins of Complex Life, Ww Norton, 2015-07-20, ISBN 978-0393088816(Link points to Figure 10 showing model of ATP synthase)
External links
- Boris A. Feniouk: "ATP synthase — a splendid molecular machine"
- Well illustrated ATP synthase lecture by Antony Crofts of the University of Illinois at Urbana–Champaign.
- Proton and Sodium translocating F-type, V-type and A-type ATPases in OPM database
- The Nobel Prize in Chemistry 1997 to Paul D. Boyer and John E. Walker for the enzymatic mechanism of synthesis of ATP; and to Jens C. Skou, for discovery of an ion-transporting enzyme, Na+
, K+
-ATPase. - Harvard Multimedia Production Site — Videos – ATP synthesis animation
- David Goodsell: "ATP Synthase- Molecule of the Month" Archived 2015-09-05 at the Wayback Machine
