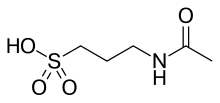
Acamprosate
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /əˈkæmproʊseɪt/ |
| Trade names | Campral EC |
| Other names | N-Acetyl homotaurine, acamprosate calcium (JAN JP), acamprosate calcium (USAN US) |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Routes of administration | Oral[1] |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 11%[1] |
| Protein binding | Negligible[1] |
| Metabolism | Nil[1] |
| Elimination half-life | 20 h to 33 h[1] |
| Excretion | Kidney[1] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Acamprosate, sold under the brand name Campral, is a medication which reduces
Serious side effects include
It is on the World Health Organization's List of Essential Medicines.[12]
Medical uses
Acamprosate is useful when used along with counseling in the treatment of
Contraindications
Acamprosate is primarily removed by the kidneys. A dose reduction is suggested in those with moderately impaired kidneys (
Adverse effects
The US label carries warnings about increases of suicidal behavior, major depressive disorder, and kidney failure.[1]
Adverse effects that caused people to stop taking the drug in clinical trials included diarrhea, nausea, depression, and anxiety.[1]
Potential adverse effects include headache, stomach pain, back pain, muscle pain, joint pain, chest pain, infections, flu-like symptoms, chills, heart palpitations, high blood pressure, fainting, vomiting, upset stomach, constipation, increased appetite, weight gain, edema, sleepiness, decreased sex drive, impotence, forgetfulness, abnormal thinking, abnormal vision, distorted sense of taste, tremors, runny nose, coughing, difficulty breathing, sore throat, bronchitis, and rashes.[1]
Pharmacology

Pharmacodynamics
The
Its activity on those receptors is indirect, unlike that of most other agents used in this context.[20] An inhibition of the GABA-B system is believed to cause indirect enhancement of GABAA receptors.[20] The effects on the NMDA complex are dose-dependent; the product appears to enhance receptor activation at low concentrations, while inhibiting it when consumed in higher amounts, which counters the excessive activation of NMDA receptors in the context of alcohol withdrawal.[21]
The product also increases the endogenous production of taurine.[21]
In addition, alcohol also inhibits the activity of
The substance also helps re-establish a standard sleep architecture by normalizing stage 3 and REM sleep phases, which is believed to be an important aspect of its pharmacological activity.[21]
Pharmacokinetics
Acamprosate is not
Its absorption and elimination are very slow, with a
History
Acamprosate was
In October 2001 Forest Laboratories acquired the rights to market the drug in the US.[30][31]
It was approved by the FDA in July 2004.[32]
The first generic versions of acamprosate were launched in the US in 2013.[33]
As of 2015, acamprosate was in development by Confluence Pharmaceuticals as a potential treatment for
Society and culture
Acamprosate is the
. It is also technically known as N-acetylhomotaurine or as calcium acetylhomotaurinate.It is sold under the brand name Campral.[1]
Research
In addition to its apparent ability to help patients refrain from drinking, some evidence suggests that acamprosate is neuroprotective (that is, it protects neurons from damage and death caused by the effects of alcohol withdrawal, and possibly other causes of neurotoxicity).[26][35]
Some struggle to take the full course of medication (2 tablets, 3 times a day), which makes acamprosate less effective. Standard support to help people take their medication involves monthly check-ins with addiction services or a GP. Research found that compared to standard support alone, extra telephone support by a pharmacist, plus financial incentives, increased the numbers who took medication as prescribed and was cost-effective. The same telephone support without financial incentives did not significantly increase the numbers taking their medication as prescribed and was less cost-effective.[36][37]
References
- ^ a b c d e f g h i j k l "Campral label" (PDF). FDA. January 2012. Retrieved 27 November 2017. For label updates see FDA index page for NDA 021431
- ^ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 30 March 2024.
- ^ S2CID 19119078.
- PMID 16300039. Archived from the originalon 2007-09-29. Retrieved 2006-11-29.
- ^ ISBN 978-0-07-182770-6.
It has been hypothesized that long-term ethanol exposure alters the expression or activity of specific GABAA receptor subunits in discrete brain regions. Regardless of the underlying mechanism, ethanol-induced decreases in GABAA receptor sensitivity are believed to contribute to ethanol tolerance, and also may mediate some aspects of physical dependence on ethanol. ... Detoxification from ethanol typically involves the administration of benzodiazepines such as chlordiazepoxide, which exhibit cross-dependence with ethanol at GABAA receptors (Chapters 5 and 15). A dose that will prevent the physical symptoms associated with withdrawal from ethanol, including tachycardia, hypertension, tremor, agitation, and seizures, is given and is slowly tapered. Benzodiazepines are used because they are less reinforcing than ethanol among alcoholics. Moreover, the tapered use of a benzodiazepine with a long half-life makes the emergence of withdrawal symptoms less likely than direct withdrawal from ethanol. ... Unfortunately, acamprosate is not adequately effective for most alcoholics.
- PMID 11584875.
- S2CID 36860967.
- ^ "Acamprosate". drugs.com. 2005-03-25. Archived from the original on 22 December 2006. Retrieved 2007-01-08.
- S2CID 195691152.
- ^ "Acamprosate (Campral) Use During Pregnancy". Drugs.com.
- ^ Haber P, Lintzeris N, Proude E, Lopatko O. "Guidelines for the Treatment of Alcohol Problems" (PDF). Australian Government Department of Health and Ageing. Retrieved 20 February 2023.
- hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- PMID 30167705.
- PMID 23075288.
- PMID 25664494.
- ^ S2CID 34047050.
- ^ "Acamprosate: Biological activity". IUPHAR/BPS Guide to Pharmacology. International Union of Basic and Clinical Pharmacology. Retrieved 26 November 2017.
Due to the complex nature of this drug's MMOA, and a paucity of well defined target affinity data, we do not map to a primary drug target in this instance.
- ^ a b c d "Acamprosate: Summary". IUPHAR/BPS Guide to Pharmacology. International Union of Basic and Clinical Pharmacology. Retrieved 26 November 2017.
Acamprosate is a NMDA glutamate receptor antagonist and a positive allosteric modulator of GABAA receptors.
Marketed formulations contain acamprosate calcium - ^ a b c d e f "Acamprosate". DrugBank. University of Alberta. 19 November 2017. Retrieved 26 November 2017.
Acamprosate is thought to stabilize the chemical balance in the brain that would otherwise be disrupted by alcoholism, possibly by blocking glutaminergic N-methyl-D-aspartate receptors, while gamma-aminobutyric acid type A receptors are activated. ... The mechanism of action of acamprosate in the maintenance of alcohol abstinence is not completely understood. Chronic alcohol exposure is hypothesized to alter the normal balance between neuronal excitation and inhibition. in vitro and in vivo studies in animals have provided evidence to suggest acamprosate may interact with glutamate and GABA neurotransmitter systems centrally, and has led to the hypothesis that acamprosate restores this balance. It seems to inhibit NMDA receptors while activating GABA receptors.
- ^ PMID 23278595.
- ^ PMID 20201812.
- ISBN 978-0-07-148127-4.
- PMID 22429661.
- PMID 9509257.
- PMID 9619143.
- ^ S2CID 11563216.
- PMID 12394279.
- PMID 11584146.
- OCLC 1015240036.
- ^ a b Berfield S (27 May 2002). "A CEO and His Son". Bloomberg Businessweek.
- ^ "Press release: Forest Laboratories Announces Agreement For Alcohol Addiction Treatment". Forest Labs via Evaluate Group. October 23, 2001.
- Food and Drug Administration. 2004-07-29. Archived from the originalon 2008-01-17. Retrieved 2009-08-15.
- ^ "Acamprosate generics". DrugPatentWatch. Archived from the original on 1 December 2017. Retrieved 27 November 2017.
- ^ "Acamprosate - Confluence Pharmaceuticals". AdisInsight. Springer Nature Switzerland AG. Retrieved 27 November 2017.
- PMID 18540918.
- PMID 37924307.
- .
