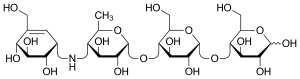
Acarbose
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Glucobay, Precose, Prandase |
| Other names | (2R,3R,4R,5S,6R)-5-{[(2R,3R,4R,5S,6R)-5- {[(2R,3R,4S,5S,6R)-3,4-dihydroxy-6-methyl- 5-{[(1S,4R,5S,6S)-4,5,6-trihydroxy-3- (hydroxymethyl)cyclohex-2-en-1-yl]amino} tetrahydro-2H-pyran-2-yl]oxy}-3,4-dihydroxy- 6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl]oxy}- 6-(hydroxymethyl)tetrahydro-2H-pyran-2,3,4-triol |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a696015 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Extremely low |
| Metabolism | Gastrointestinal tract |
| Elimination half-life | 2 hours |
| Excretion | Kidney (less than 2%) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Acarbose (
Acarbose is a starch blocker. It works by
Acarbose is cheap and popular in China, but not in the U.S. One physician explains that use in the U.S. is limited because it is not potent enough to justify the side effects of diarrhea and flatulence.[4] However, a large study concluded in 2013 that "acarbose is effective, safe and well tolerated in a large cohort of Asian patients with type 2 diabetes."[5] A possible explanation for the differing opinions is an observation that acarbose is significantly more effective in patients eating a relatively high carbohydrate Eastern diet.[6]
Medical uses
Dosing
Since acarbose prevents the digestion of complex carbohydrates, the drug should be taken at the start of main meals (taken with first bite of meal).
Efficacy
In type II diabetic patients, acarbose averages an absolute decrease of 0.8 percentage points in HbA1c, which is a decrease of about 10% in typical HbA1c values in diabetes studies.[8] Individuals with higher baseline levels show higher reductions, about an 0.12% additional decrease for each point of baseline HbA1c.[8] Its effect on postprandial glucose, but not on HbA1c, scales with dose.[8] Among diabetic patients, acarbose may help reduce the damage done to blood vessels and kidneys by reducing glucose levels.[8]
A Cochrane systematic review assessed the effect of alpha-glucosidase inhibitors in people with prediabetes, defined as impaired glucose tolerance, impaired fasting blood glucose, elevated glycated hemoglobin A1c (HbA1c).[9] It was found that acarbose reduced the incidence of diabetes mellitus type 2 when compared to placebo, however there was no conclusive evidence that acarbose, when compared to diet and exercise, metformin, placebo, or no intervention, improved all-cause mortality, reduced or increased risk of cardiovascular mortality, serious or non-serious adverse events, non-fatal stroke, congestive heart failure, or non-fatal myocardial infarction.[9]
Combination therapy
The combination of acarbose with metformin results in greater reductions of HbA1c, fasting blood glucose and post-prandial glucose than either agent alone.[10]
Adverse effects
Since acarbose prevents the degradation of complex carbohydrates into glucose, some carbohydrate will remain in the intestine and be delivered to the
If a patient using acarbose has a bout of hypoglycemia, the patient must eat something containing monosaccharides, such as glucose tablets or gel (GlucoBurst, Insta-Glucose, Glutose, Level One) and a doctor should be called. Because acarbose blocks the breakdown of table sugar and other complex sugars, fruit juice or starchy foods will not effectively reverse a hypoglycemic episode in a patient taking acarbose.[12] Acarbose by itself carries minimal risk of hypoglycemia.[8]
Acarbose is associated with very rare elevated transaminases (19 out of 500,000).[8] Even rarer cases of hepatitis has been reported with acarbose use. It usually goes away when the medicine is stopped. Liver enzymes should be checked before and during use of this medicine as a precaution.[13] A 2016 meta-analysis confirms that alpha-glucosidase inhibitors, including acarbose, have a statistically significant link to elevated transaminase levels.[14]
Pharmacology
Mechanism of action
Acarbose inhibits enzymes (
Metabolism
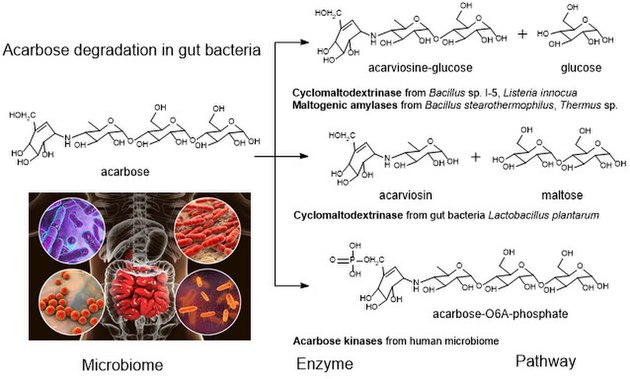
Acarbose degradation is the unique feature of glycoside hydrolases in gut microbiota, acarbose degrading glucosidase, which hydrolyze acarbose into an acarviosine-glucose and glucose.[18] Human enzymes do transform acarbose: the pancreatic alpha-amylase is able to perform a rearrangement reaction, moving the glucose unit in the "tail" maltose to the "head" of the molecule. Analog drugs with the "tail" glucose removed or flipped to an α(1-6) linkage resist this transformation.[15]
It has been reported that the maltogenic alpha-amylase from Thermus sp. IM6501 (ThMA) and a cyclodextrinase (CDase) from Streptococcus pyogenes could hydrolyse acarbose to glucose and acarviosine-glucose, ThMA can further hydrolyze acarviosine-glucose into acarviosin and glucose.[20][21] A cyclomaltodextrinase (CDase) from gut bacteria Lactobacillus plantarum degraded acarbose via two different modes of action to produce maltose and acarviosin, as well as glucose and acarviosine-glucose, suggest that acarbose resistance is caused by the human microbiome.[3] The microbiome-derived acarbose kinases are also specific to phosphorylate and inactivate acarbose.[22] The molecular modeling showed the interaction between gut bacterial acarbose degrading glucosidase and human α-amylase.[23]

Natural distribution
In nature, acarbose is synthesized by
In molecular biology
Acarbose is described chemically as a pseudo
Research
In human T2DM patients, acarbose reduces total triglyceride levels.[27] Acarbose has a similar effect in non-T2DM patients with isolated familial hypertriglyceridemia.[8]
In smaller samples of healthy human volunteers, acarbose increases postprandial
In studies conducted by three independent laboratories by the US National Institute on Aging's intervention testing programme, acarbose was shown to extend the lifespan of female mice by 5% and of male mice by 22%.[28][29]
References
- ISBN 978-1-4757-2085-3.
- ^ "International Nonproprietary Names for Pharmaceutical Substances. Recommended International Nonproprietary Names (Rec. INN): List 19" (PDF). World Health Organization. 1979. Retrieved 9 November 2016.
- ^ S2CID 2660911.
- ^ Kresge N (21 November 2011). "China's Thirst for New Diabetes Drugs Threatens Bayer's Lead". Bloomberg Business Week. Archived from the original on 21 November 2011. Retrieved 15 April 2016.
- S2CID 207483590.
- PMID 23602502.
- S2CID 36582205.
- ^ PMID 26512331.
- ^ PMID 30592787.
- S2CID 203985605.
- PMID 9428831.
- ^ "Acarbose". MedlinePlus Drug Information.
- ^ "Acarbose: hepatitis: France, Spain". WHO Pharmaceuticals Newsletter. 1999. Archived from the original on October 15, 2009.
- PMID 27596383.
- ^ PMID 15736945.
- PMID 10209866.
- PMID 18756100.
- ^ PMID 10103262.
- ^ Drug Therapy in Nursing, 2nd Edition.
- ISSN 0440-2413.
- S2CID 2813481.
- S2CID 244644880.
- S2CID 86894203.
- PMID 35705566.
- S2CID 25592455.
- PMID 32381508.
- PMID 37990256.
- PMID 24245565.
- PMID 29291036.
External links
- "Probing the Pancreas" - by Craig D. Reid, Ph.D. (US FDA Consumer Article)
