Acene

In
molecular formula
C4n+2H2n+4.
The larger representatives have potential interest in
optoelectronic applications and are actively researched in chemistry and electrical engineering. Pentacene has been incorporated into organic field-effect transistors, reaching charge carrier
mobilities as high as 5 cm2/Vs.
The first 5 unsubstituted members are listed in the following table:
| Name | Number of rings | Molecular formula
|
Structural formula |
|---|---|---|---|
| Anthracene | 3 | C14H10 | 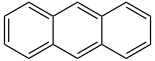
|
| Tetracene | 4 | C18H12 | 
|
| Pentacene | 5 | C22H14 | 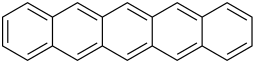
|
| Hexacene | 6 | C26H16 | 
|
| Heptacene | 7 | C30H18 | 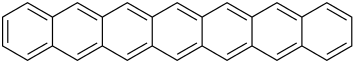
|
Hexacene is not stable in air, and
dimerises upon isolation. Heptacene (and larger acenes) is very reactive and has only been isolated in a matrix. However, bis(trialkylsilylethynylated) versions of heptacene have been isolated as crystalline solids.[3]
Larger acenes
Due to their increased
HOMO-LUMO gap reported for any acene,[8] an observation in violation of Kasha's rule. Subsequent work by others on different derivatives included crystal structures, with no such violations.[9] The on-surface synthesis and characterization of unsubstituted, parent nonacene (n=9)[10] and decacene (n=10)[11] have been reported. In 2020, scientists reported about the creation of dodecacene (n=12)[12] for the first time. Four years later, in the beginning of 2024, Ruan et al. succeeded in synthesizing unsubstitued tridecacene (n=13) on a (111)-gold surface. The acene was characterized by STM- and STS-measurements. [13]
Related compounds
The acene series have the consecutive rings linked in a linear chain, but other chain linkages are possible. The phenacenes have a zig-zag structure and the helicenes have a helical structure.
- Macromolecular forms consisting of seven fused benzene rings
-
Heptacene
-
[7]Phenacene
-
M-heptahelicene
Benz[a]anthracene, an isomer of tetracene, has three rings connected in a line and one ring connected at an angle.
References
- ^ Electronic structure of higher acenes and polyacene: The perspective developed by theoretical analyses Holger F. Bettinger Pure Appl. Chem., Vol. 82, No. 4, pp. 905–915, 2010.
- PMID 18046697.
- PMID 20468014.
- PMID 26894252.
- PMID 27609626.
- PMID 20432492.
- PMID 20055388.
- PMID 21717552.
- S2CID 209341741.
- PMC 10870776.

