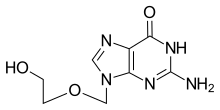
Aciclovir
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /eɪˈsaɪkloʊvɪər/ |
| Trade names | Zovirax, others[1] |
| Other names | Acycloguanosine, acyclovir (BAN UK), acyclovir (USAN US) |
| AHFS/Drugs.com | |
| MedlinePlus | a681045 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Intravenous, by mouth, topical, eye ointment |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 15–20% (by mouth)[3] |
| Protein binding | 9–33%[3] |
| Metabolism | Liver |
| Elimination half-life | 2–4 hours |
| Excretion | Kidney (62–90% as unchanged drug) |
| Identifiers | |
| |
JSmol) | |
| Melting point | 256.5 °C (493.7 °F) |
| |
| |
| (verify) | |
Aciclovir, also known as acyclovir,
Common side effects include nausea and diarrhea.[6] Potentially serious side effects include kidney problems and low platelets.[6] Greater care is recommended in those with poor liver or kidney function.[6] It is generally considered safe for use in pregnancy with no harm having been observed.[6][8] It appears to be safe during breastfeeding.[9][10] Aciclovir is a nucleoside analogue that mimics guanosine.[6] It works by decreasing the production of the virus's DNA.[6]
Aciclovir was patented in 1974 by
Medical use

Aciclovir is used for the treatment of herpes simplex virus (HSV) and varicella zoster virus infections, including:[3][16][17]
- Genital prevention)
- Neonatal herpes simplex
- Herpes simplex labialis (cold sores)
- Shingles
- Acute chickenpox in immunocompromised patients
- Herpes simplex encephalitis
- Acute mucocutaneousHSV infections in immunocompromised patients
- Herpes of the eye and herpes simplex blepharitis(a chronic (long-term) form of herpes eye infection)
- Prevention of herpes viruses in immunocompromised people (such as people undergoing cancer chemotherapy)[18]
Its effectiveness in treating Epstein–Barr virus infections is less clear.[6] It has not been found to be useful for infectious mononucleosis due to Epstein-Barr virus.[19] Valaciclovir and acyclovir act by inhibiting viral DNA replication, but as of 2016 there was little evidence that they are effective against Epstein–Barr virus, they are expensive, they risk causing resistance to antiviral agents, and (in 1% to 10% of cases) can cause unpleasant side effects.[20]
Aciclovir taken by mouth does not appear to decrease the risk of pain after shingles.[21] In those with herpes of the eye, aciclovir may be more effective and safer than idoxuridine.[22] It is not clear if aciclovir eye drops are more effective than brivudine eye drops.[22]
Pregnancy
The CDC and others have declared that during severe recurrent or first episodes of genital herpes, aciclovir may be used.[26] For severe HSV infections (especially disseminated HSV), IV aciclovir may also be used.[27]
Studies in mice, rabbits and rats (with doses more than 10 times the equivalent of that used in humans) given during organogenesis have failed to demonstrate birth defects.[28] Studies in rats in which they were given the equivalent to 63 times the standard steady-state humans concentrations of the drug[Note 1] on day 10 of gestation showed head and tail anomalies.[28]
Aciclovir is recommended by the CDC for treatment of
Aciclovir is excreted in the breast milk, therefore it is recommended that caution should be used in breast-feeding women. It has been shown in limited test studies that the nursing infant is exposed to approximately 0.3 mg/kg/day following oral administration of aciclovir to the mother. If nursing mothers have herpetic lesions near or on the breast, breast-feeding should be avoided.[30][31]
Adverse effects
Systemic therapy
Common
Intravenous aciclovir may cause reversible nephrotoxicity in up to 5% to 10% of patients because of precipitation of aciclovir crystals in the kidney. Aciclovir crystalline nephropathy is more common when aciclovir is given as a rapid infusion and in patients with dehydration and preexisting renal impairment. Adequate hydration, a slower rate of infusion, and dosing based on renal function may reduce this risk.[32][33][34]
The aciclovir metabolite 9-Carboxymethoxymethylguanine (9-CMMG) has been shown to play a role in neurological adverse events, particularly in older people and those with reduced renal function.[35][36][37]
Topical therapy
Aciclovir topical cream is commonly associated (≥1% of patients) with: dry or flaking skin or transient stinging/burning sensations. Infrequent adverse effects include
Drug interactions
Ketoconazole: In-vitro replication studies have found a synergistic, dose-dependent antiviral activity against HSV-1 and HSV-2 when given with aciclovir. However, this effect has not been clinically established and more studies need to be done to evaluate the true potential of this synergy.[38]
Probenecid: Reports of increased half life of aciclovir, as well as decreased urinary excretion and renal clearance have been shown in studies where probenecid is given simultaneously with aciclovir.[30]
Interferon: Synergistic effects when administered with aciclovir and caution should be taken when administering aciclovir to patients receiving IV interferon.[39]
Zidovudine: Although administered often with aciclovir in HIV patients, neurotoxicity has been reported in at least one patient who presented with extreme drowsiness and lethargy 30–60 days after receiving IV aciclovir; symptoms resolved when aciclovir was discontinued.[40]
Detection in biological fluids
Aciclovir may be quantitated in plasma or serum to monitor for drug accumulation in patients with renal dysfunction or to confirm a diagnosis of poisoning in acute overdose victims.[41]
Mechanism of action


Aciclovir is converted by viral
Resistance
Resistance to aciclovir is rare in people with healthy immune systems, but is more common (up to 10%) in people with immunodeficiencies on chronic antiviral prophylaxis (transplant recipients, people with
Microbiology

Aciclovir is active against most species in the
- Herpes simplex virus type I (HSV-1)
- Herpes simplex virus type II (HSV-2)
- Varicella zoster virus
- Epstein–Barr virus
- Human cytomegalovirus– least activity
Pharmacokinetics
Aciclovir is poorly water-soluble and has poor oral
Chemistry
Details of the synthesis of aciclovir were first published by scientists from the University at Buffalo.[51]
In the first step shown, 2,6-dichloro
History
Aciclovir was seen as the start of a new era in antiviral therapy, as it is extremely selective and low in
A related prodrug form, valaciclovir came into medical use in 1995. It is converted to aciclovir in the body after absorption.[61]
In 2009, acyclovir in combination with hydrocortisone cream, marketed as Xerese, was approved in the United States for the early treatment of recurrent herpes labialis (cold sores) to reduce the likelihood of ulcerative cold sores and to shorten the lesion healing time in adults and children (six years of age and older).[62][63]
Society and culture
Names
Aciclovir is the international nonproprietary name (INN) and British Approved Name (BAN) while acyclovir is the United States Adopted Name (USAN) and former British Approved Name.[citation needed]
It was originally marketed as Zovirax; patents expired in the 1990s and since then it is generic and is marketed under many brand names worldwide.[1]
Notes
- ^ Subject to the same conditions as before
References
- ^ a b c "Aciclovir". Drugs.com. Archived from the original on 23 September 2015. Retrieved 6 September 2015.
- ^ "Skin health". Health Canada. 9 May 2018. Retrieved 13 April 2024.
- ^ a b c d "Zovirax (acyclovir) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Archived from the original on 19 February 2014. Retrieved 5 February 2014.
- ^ Kevin ES. "The Aciclovir" (in German). Kevin. Retrieved 25 May 2017.
- ^ PMID 16284630.
- ^ a b c d e f g h i j "Acyclovir". The American Society of Health-System Pharmacists. Archived from the original on 2015-01-05. Retrieved Jan 1, 2015.
- PMID 20739216.
- ^ "Prescribing medicines in pregnancy database". Australian Government. 3 March 2014. Archived from the original on 8 April 2014. Retrieved 22 April 2014.
- ISBN 9781284057560.
- ^ "Acyclovir use while Breastfeeding". Mar 10, 2015. Archived from the original on 5 March 2016. Retrieved 8 March 2016.
Even with the highest maternal dosages, the dosage of acyclovir in milk is only about 1% of a typical infant dosage and would not be expected to cause any adverse effects in breastfed infants
- ISBN 9783527607495.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Acyclovir - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ ISBN 978-0-9805790-9-3.
- ISBN 978-0-85711-084-8.
- S2CID 2969472.
- PMID 16006448.
- PMID 27933614.
- PMID 24500927.
- ^ PMID 25879115.
- from the original on 2007-05-15. – Editorial
- PMID 12069980.
- PMID 20519025.
- PMID 15767977.
- PMID 19357635.
- ^ a b c d "PRODUCT INFORMATION NAME OF THE DRUG OZVIR TABLETS" (PDF). TGA eBusiness Services. Ranbaxy Australia Pty Ltd. 26 August 2011. Archived from the original on 20 August 2016. Retrieved 6 February 2014.
- ^ Centers for Disease Control and Prevention. (January 2006). Epidemiology and prevention of vaccine-preventable diseases (9th ed.). Public Health Foundation. pp. 171–192.
- ^ a b "Acyclovir label" (PDF). GSK. FDA. 2005. Archived from the original (PDF) on 2017-09-08.
- S2CID 5791615.
- PMID 21964179.
- PMID 6285711.
- PMID 3376977.
- PMID 12748346.
- PMID 25440915.
- S2CID 32983100.
- PMID 3021048.
- ^ GlaxoSmithKline. Zovirax® (acyclovir sodium) for injection prescribing information. Research Triangle Park, NC; 2003 Nov
- PMID 3468354.
- ISBN 9780962652370.
- ^ "VALTREX (valacyclovir hydrochloride) Caplets -GSKSource". gsksource.com. Retrieved 2019-08-02.
- ^ "Acyclovir (acyclovir) Capsule Acyclovir (acyclovir) Tablet [Genpharm Inc.]". DailyMed. Genpharm Inc. November 2006. Archived from the original on 21 February 2014. Retrieved 5 February 2014.
- ^ "Aciclovir Tablets BP 400mg - Summary of Product Characteristics (SPC)". electronic Medicines Compendium. Actavis UK Ltd. 20 August 2012. Archived from the original on 22 February 2014. Retrieved 5 February 2014.
- ^ Sweetman, S, ed. (7 August 2013). "Aciclovir". Martindale: The Complete Drug Reference. London, UK: Pharmaceutical Press. Retrieved 6 February 2014.
- PMID 21078929.
- S2CID 240858022.
- PMID 7510619.
- .
- ^ "Aciclovir Tablets BP 400mg - Summary of Product Characteristics (SmPC) - (emc)". www.medicines.org.uk.
- ^ PMID 5553754.
- ^ US patent 4199574, Schaeffer HJ, "Methods and compositions for treating viral infections and guanine acyclic nucleosides", published 1980-04-22, assigned to Burroughs Wellcome.
- S2CID 75449475.
- ^ Garrison T (1999). Oceanography: An Invitation to Marine Science, 3rd ed. Belmont, CA: Wadsworth Publishing Company. p. 471.
- S2CID 84041855.
- PMID 19149592.
- PMID 202961.
- ^ US 4146715, Schaeffer HJ, "2-Amido-9-(2-acyloxyethoxymethyl)hypoxanthines", issued 27 March 1979, assigned to Burrough Wellcome Co.
- ^ Vince R (2008). "A brief history of the development of Ziagen". Chemtracts. 21: 127–134.
- ^ "The Nobel Prize - Gertrude B. Elion".
- ^ "Valacyclovir Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 17 March 2019.
- ^ "Drug Approval Package: Acyclovir and Hydrocortisone NDA #022436". U.S. Food and Drug Administration (FDA). 13 December 2019. Archived from the original on 13 December 2019. Retrieved 13 December 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "Xerese- acyclovir and hydrocortisone cream". DailyMed. 12 December 2019. Archived from the original on 13 December 2019. Retrieved 12 December 2019.
Further reading
- Hazra S, Konrad M, Lavie A (August 2010). "The sugar ring of the nucleoside is required for productive substrate positioning in the active site of human deoxycytidine kinase (dCK): implications for the development of dCK-activated acyclic guanine analogues". Journal of Medicinal Chemistry. 53 (15): 5792–5800. PMID 20684612.
- Harvey SC (1990). "Drug absorption, action and disposition". In Remington JP, Gennaro AR (eds.). Remington's Pharmaceutical Sciences (18th ed.). Easton, Pa.: Mack Pub. Co. pp. 697–702. ISBN 978-0-912734-04-0.
- Huovinen P, Valtonen V (1994). Neuvonen PJ (ed.). Kliininen Farmakologia (in Finnish). Helsinki: Kandidaattikustannus Oy. ISBN 951-8951-09-8.
- Périgaud C, Gosselin G, Imbach JL (1992). "Nucleoside analogues as chemotherapeutic agents: a review". Nucleosides and Nucleotides. 11 (2–4): 903–945. .
- Rang HP, Dale MM, Ritter JM (2003). Pharmacology (5th ed.). Pearson Professional Ltd. ISBN 0-443-07145-4.

