Acid salt
Acid salts are a class of
Acidic solution and examples of acid salts
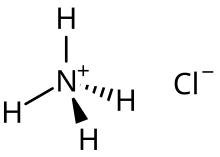
For example, the acid salt ammonium chloride is the main species formed upon the half neutralization of ammonia in aqueous solution of hydrogen chloride:[2]
- NH3(aq) + HCl(aq) → [NH4]+Cl−(aq)
| Name | Sodium bisulfate | Monosodium phosphate | Disodium phosphate |
|---|---|---|---|
| Structural formula |  |
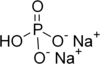 | |
| Chemical formula | NaHSO4 | NaH2PO4 | Na2HPO4 |
IUPAC name
|
Sodium hydrogen sulfate | Sodium dihydrogen phosphate | Disodium hydrogen phosphate |
| Other name |
|
|
|
Molecular weight
|
120.054 g/mol | 119.976 g/mol | 141.957 g/mol |
| Formal charge | zero | zero | zero |
Odour
|
Odourless | Odourless | Odourless[3] |
| Appearance | White crystals or granules | White crystalline powder[4] | White, hygroscopic powder[5] |
| Structure |
|
Monoclinic crystals[6] | Monoclinic crystals (anhydrous)[7] |
| Solubility |
|
|
|
| Density |
|
0.5–1.2 g/cm3 | 1.7 g/cm3 |
| Decomposition (through heating) |
Emits toxic fumes of phosphoxides and sodium oxide[8] | Emits toxic fumes of phosphorus- and sodium oxides[8] | |
| Uses |
|
|
|
Use in food
Acidic salts are often used in foods as part of leavening agents. In this context, the acid salts are referred to as "leavening acids."[9] Common leavening acids include cream of tartar and monocalcium phosphate.
An acidic salt can be mixed with an alkali salt (such as sodium bicarbonate or baking soda) to create baking powders which release carbon dioxide.[10] Leavening agents can be slow-acting (e.g. sodium aluminum phosphate) which react when heated, or fast-acting (e.g., cream of tartar) which react immediately at low temperatures. Double-acting baking powders contain both slow- and fast-acting leavening agents and react at low and high temperatures to provide leavening rising throughout the baking process.[11]
Disodium phosphate, Na2HPO4, is used in foods and monosodium phosphate, NaH2PO4, is used in animal feed, toothpaste and evaporated milk.
Intensity of acid
An acid with higher
- The solution is acidic if Ka > Kb. It contains a greater concentration of H+ ions than concentration of OH− ions due more extensive of cation hydrolysis compared to that of anion hydrolysis.
- The solution is alkali if Ka < Kb. Anions hydrolyze more than cations, causing an exceeding concentration of OH− ions.
- The solution is expected to be neutral only when Ka = Kb.[12]
Other possible factors that could vary
For example, in ammonium chloride solution, NH+4 is the main influence for acidic solution. It has greater Ka value compared to that of water molecules; Ka of NH+4 is 5.6×10−10, and Kw of H2O is 1.0×10−14. This ensures its deprotonation when reacting with water, and is responsible for the pH below 7 at room temperature. Cl− will have no affinity for H+ nor tendency to hydrolyze, as its Kb value is very low (Kb of Cl− is 7.7×10−21).[13]
Hydrolysis of ammonium at room temperature produces:
See also
- Alkali salt
- Salt (chemistry)
- Oxoacid
- Sodium bicarbonate
- Sodium bisulfate
- Disodium phosphate
- Monosodium phosphate
References
- .
- ISBN 978-0-935702-61-3. Retrieved 8 February 2018.
- ^ U.S. Coast Guard, Department of Transportation. CHRIS - Hazardous Chemical Data. Volume II. Washington, D.C.: U.S. Government Printing Office, 1984-5.
- ^ Lewis, R.J. Sr.; Hawley's Condensed Chemical Dictionary 15th Edition. John Wiley & Sons, Inc. New York, NY 2007., p. 1153
- ^ Lide, D.R. CRC Handbook of Chemistry and Physics 88TH Edition 2007-2008. CRC Press, Taylor & Francis, Boca Raton, Florida 2007, p. 4-90
- ^ Haynes, W.M. (ed.). CRC Handbook of Chemistry and Physics. 95th Edition. CRC Press LLC, Boca Raton: FL 2014-2015, p. 4-89
- S2CID 102468247.
- ^ ISBN 978-0-471-47662-7.
- ^ Wallace, David (10 June 2015). "Leavening Acids – Baking Ingredients". BAKERpedia. Retrieved 17 October 2019.
- ^ "The Many Uses of Cream of Tartar". Office for Science and Society. Retrieved 17 October 2019.
- S2CID 54052197.
- ISBN 978-0077274313. Retrieved 9 February 2018.
- ^ Lower, S.K., (1999). Introduction to acid-base chemistry. Chem1 General Chemistry Text. Retrieved from http://www.chem1.com/acad/pdf/c1xacid1.pdf



![{\displaystyle K_{\mathrm {a} }={\frac {\mathrm {[NH_{3}][H_{3}O^{+}]} }{\mathrm {[NH_{4}^{+}]} }}={\frac {K_{\mathrm {w} }}{K_{\mathrm {b} }}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/5eac34ff25fa376268b6f116d33bf3d00ca7dfab)
![{\displaystyle K_{\mathrm {a} }={\frac {[1.0\times 10^{-14}]}{[1.8\times 10^{-5}]}}={5.6}\times 10^{-10}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/43f0025153c13e4b649bc0ef2c4ea94caefc4c28)