Activation-induced cell death
AICD (activation-induced cell death) is
The AICD effector cell is one that expresses FasL, and
Signaling
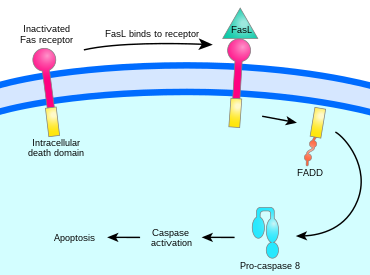
The binding of Fas ligand to Fas receptor triggers trimerization of Fas, whose cytoplasmic domain is then able to bind the death domain of the adaptor protein FADD (Fas-associated protein with death domain). Procaspase 8 binds to FADD's death effector domain (DED) and proteolytically self-activates as caspase 8. Fas, FADD, and procaspase 8 together form a death-inducing signaling complex (DISC). Activated caspase 8 is released into the cytosol, where it activates the caspase cascade that initiates apoptosis.[5][1]
Regulation of Fas-FasL and AICD
FasL is primarily regulated at the transcriptional level. (The other option is regulation of the signal emanating from the
Not much is known about the regulation of Fas and other death receptors. However, overexpression of the protein CFLAR (caspase and FADD-like apoptosis regulator) inhibits Fas-mediated apoptosis.[6]
See also
References
- ^ a b c d e f g Zhang J, Xu X, Liu Y. (2004), Activation-Induced Cell Death in T Cells and Autoimmunity. Cell Mol Immunol. 1(3):186-92
- ^ Kabelitz D, Janssen O. (1997), Antigen-induced death of T-lymphocytes. Front Biosci. 2:d61-77
- ^ a b Green DR, Droin N, Pinkoski M. (2003), Activation-induced cell death in T cells. Immunol Rev. 193:70-81
- ^ Donjerković D, Scott DW. (2000), Activation-induced cell death in B lymphocytes. Cell Res. 10(3):179-92
- ^ Nagata S. (1997), Apoptosis by death factor. Cell. 88(3):355-65
- ^ Scaffidi C, Schmitz I, Krammer PH, Peter ME. (1999é, The role of c-FLIP in modulation of CD95-induced apoptosis. J Biol Chem 274:1541–1548
