Infective endocarditis
| Infective endocarditis | |
|---|---|
| Other names | Bacterial endocarditis |
heart surgery[1] | |
| Prognosis | 25% risk of death[6] |
| Frequency | 5 per 100,000 per year[6] |
Infective endocarditis is an
The cause is typically a
The usefulness of
Classification
Infective endocarditis is divided into the three categories of acute, subacute, and chronic based on the duration of symptoms.[12] Acute infective endocarditis refers to the presence of signs and symptoms of infective endocarditis that are present for days up to six weeks.[12] If these signs and symptoms persist for more than six weeks but less than three months, this is subacute infective endocarditis.[12] Chronic infective endocarditis refers to the presence of such signs and symptoms when they persist for more than three months.[12]
- streptococci of low virulence (mainly viridans streptococci) and mild to moderate illness which progresses slowly over weeks and months (>2 weeks) and has low propensity to hematogenously seed extracardiac sites.
- Acute bacterial endocarditis (ABE) is a fulminant illness over days to weeks (<2 weeks), and is more likely due to Staphylococcus aureus, which has much greater virulence or disease-producing capacity and frequently causes metastatic infection.[13]
This classification is now discouraged, because the ascribed associations (in terms of organism and prognosis) were not strong enough to be relied upon clinically. The terms short incubation (meaning less than about six weeks) and long incubation (greater than about six weeks) are preferred.[14]
Culture results
Infective endocarditis may also be classified as culture-positive or culture-negative. By far the most common cause of "culture-negative" endocarditis is prior administration of antibiotics and can occur in up to 31% of cases.[15][16]
Sometimes
Heart side
Endocarditis can also be classified by the side of the heart affected:
- People who intravenously inject opioids such as heroin or methamphetamine may introduce infection which can travel to the right side of the heart, classically affecting the tricuspid valve, and most often caused by S. aureus.[13]
- Regardless of cause, left-sided endocarditis is the most common, while right-sided endocarditis accounts for 5-10% of cases and is more common in people who inject IV drugs and in patients with congenital heart disease.[13][19]
Infection setting
Another form of endocarditis is
Valve type
Finally, the distinction between native-valve endocarditis and prosthetic-valve endocarditis is clinically important. Prosthetic valve endocarditis can be early (within 1 year of surgery) or late (> 1 year following valvular surgery).[21]
- Early prosthetic valve endocarditis is usually due to intraoperative contamination or postoperative bacterial contamination which is usually nosocomialin nature.
- Late prosthetic valve endocarditis is usually due to community-acquired microorganisms.[20]
Prosthetic valve endocarditis is commonly caused by Staphylococcus epidermidis as it is capable of growing as a biofilm on plastic surfaces.[22] Cutibacterium acnes almost exclusively causes endocarditis on prosthetic heart valves.[17]
Signs and symptoms
- fatigue in 90% of people.[23]
- A new or changing coughing occurs in 35% of people.[23]
- Vascular phenomena: septic embolism (a piece of infected debris or tissue breaking off and traveling through the bloodstream to a distant site) (causing thromboembolic problems such as a stroke or gangrene of the fingers), Janeway lesions (painless hemorrhagic cutaneous lesions on the palms and soles),[24] bleeding in the brain, conjunctival hemorrhage, splinter hemorrhages, kidney infarcts, and splenic infarcts.[25] Infective endocarditis can also lead to the formation of mycotic aneurysms.[12][8]
- Immunologic phenomena: glomerulonephritis which allows for blood and albumin to enter the urine,[13] Osler's nodes ("ephemeral spots of a painful nodular erythema, chiefly in the skin of the hands and feet"), Roth's spots on the retina, positive serum rheumatoid factor
- Other signs may include night sweats, rigors, anemia, spleen enlargement[26]
Cause
Many microorganisms can cause infective endocarditis. These are generally isolated by blood culture, where the patient's blood is drawn and any growth is noted and identified. The term bacterial endocarditis (BE) commonly is used, reflecting the fact that most cases of IE are due to bacteria; however, infective endocarditis (IE) has become the preferred term.[27]
Bacterial
The viridans group includes S. oralis, S. mitis, S. sanguis, S. gordonii and S. parasanguis. The primary habitats for these organisms are the oral cavity and upper respiratory tract.[30] These bacteria are present in the normal oral flora and enter the bloodstream due to disruption of tissues in the mouth when dental surgical procedures are performed (tooth extractions) or genitourinary manipulation. Similarly, HACEK organisms are a group of bacteria that live on the dental gums and can be seen with people who inject drugs who contaminate their needles with saliva. Patients may also have a history of poor dental hygiene or pre-existing valvular disease.[31]
Enterococcus can enter the bloodstream as a consequence of abnormalities in the gastrointestinal or genitourinary tracts.[citation needed]
Some organisms, when isolated, give valuable clues to the cause, as they tend to be specific.
- Pseudomonas species, which are very resilient organisms that thrive in water, may contaminate street drugs that have been contaminated with drinking water. P. aeruginosa can infect a child through foot punctures, and can cause both endocarditis and septic arthritis.[32]
- colon cancers. When they present as the causative agent in endocarditis, it usually prompts a colonoscopy to be done immediately due to concerns regarding spread of bacteria from the colon through the bloodstream due to the cancer breaking down the barrier between the inside of the colon (lumen) and the blood vessels which drain the bowel.[33][34]
- Less commonly reported bacteria responsible for so called "culture negative endocarditis" include Bartonella, Chlamydia psittaci, and Coxiella.[35] Such bacteria can be identified by serology, culture of the excised valve tissue, sputum, pleural fluid, and emboli, and by polymerase chain reaction or sequencing of bacterial 16S ribosomal RNA.
Multiple case reports of infective endocarditis caused by unusual organisms have been published.
Dental operations
One in eight cases of infective endocarditis is thought to be caused by S. viridans infection associated with dental procedures such as cleaning or tooth
Fungal
Fungal endocarditis (FE) is often fatal and one of the most serious forms of infective endocarditis. The types of fungi most seen associated with this disease are:
Other fungi demonstrated to cause endocarditis are Histoplasma capsulatum and Aspergillus.[35] Aspergillus contributes to roughly 25% of FE cases.[41] Endocarditis with Tricosporon asahii has also been reported in a case report.[42]
Risk factors
Risk factors for infective endocarditis are based on the premise that in a healthy individual,
- Artificial heart valves
- Intracardiac devices, such as implantable cardioverter-defibrillators
- Unrepaired cyanotic congenital heart defects
- History of infective endocarditis
- Neoplastic disease
- Chronic rheumatic heart disease, which is an autoimmune response to repeated Streptococcus pyogenesinfection (mostly in the developing world)
- Age-related degenerative valvular lesions
- Congenital heart valve abnormalities
- Hemodialysis, a medical procedure that filters the blood of individuals with kidney failure
- Poor oral hygiene
- Co-existing immunosuppressing conditions, such as alcohol use disorder, chronic liver disease, and HIV/AIDS
Pathogenesis

Damaged valves and
Damage to the valves and endocardium can be caused by:[43]
- Altered, turbulent blood flow. The areas that fibrose, clot, or roughen as a result of this altered flow are known as jet lesions. Altered blood flow is more likely in high pressure areas, so ventricular septal defects or patent ductus arteriosus can create more susceptibility than atrial septal defects.
- Catheters, electrodes, and other intracardiac prosthetic devices.
- Solid particles from repeated intravenous injections.
- Chronic inflammation. Examples include auto-immunemechanisms and degenerative valvular lesions.
The risk factors for infective endocarditis provide a more extensive list of conditions that can damage the heart.
Diagnosis
In general, the Duke criteria should be fulfilled in order to establish the diagnosis of endocarditis.[12][44] Although the Duke criteria are widely used, they have significant limitations.[12] For example, the sensitivity of the Duke criteria for detecting infective endocarditis decreases when prosthetic heart valves are present.[12]
As the Duke criteria rely heavily on the results of echocardiography, research has addressed when to order an
The blood tests
Ultrasound
Echocardiography is the main type of diagnostic imaging used to establish the diagnosis of infective endocarditis.[12] There are two main types of echocardiography used to assist with the diagnosis of IE: transthoracic echocardiography (TTE) and transesophageal echocardiography (TEE).[12]
The transthoracic echocardiogram has a sensitivity and specificity of approximately 65% and 95% if the echocardiographer believes there is 'probable' or 'almost certain' evidence of endocarditis.[51][52] However, in endocarditis involving a prosthetic valve, TTE has a sensitivity of approximately 50%, whereas TEE has a sensitivity exceeding 90%.[12] The TEE also has an important diagnostic role when the TTE does not reveal IE but diagnostic suspicion remains high, since TEE is more sensitive for infective endocarditis and is better able to characterize infection-related damage to the heart valves and surrounding tissues.[12]
Guidelines support the initial use of TTE over TEE in people with abnormal blood cultures, a new heart murmur, and suspected infective endocarditis.
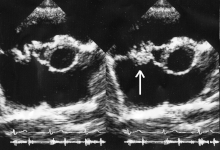
-
Ultrasound showing infectious endocarditis[53]
-
Ultrasound showing infectious endocarditis[53]
-
Ultrasound showing infectious endocarditis[53]
-
Ultrasound showing another case of infectious endocarditis[54]
Modified Duke criteria
Established in 1994 by the Duke Endocarditis Service and revised in 2000, the Duke criteria are a collection of major and minor criteria used to establish a diagnosis of infective endocarditis.[44][55] According to the Duke criteria, diagnosis of infective endocarditis can be definite, possible, or rejected.[43] A diagnosis of infective endocarditis is definite if either the following pathological or clinical criteria are met:
One of these pathological criteria:
- Histology or culture of cardiac vegetation, embolized vegetation, or intracardiac abscess from the heart finds microorganisms
- Active endocarditis
One of these combinations of clinical criteria
- Two major clinical criteria
- One major and three minor criteria
- Five minor criteria
Diagnosis of infective endocarditis is possible if one of the following combinations of clinical criteria is met:
- One major and one minor criteria
- Three minor criteria are fulfilled
Major criteria
Positive blood culture with typical IE microorganism, defined as one of the following:[43]
- Typical microorganism consistent with IE from two separate blood cultures, as noted below:
- Viridans-group streptococci, or
- Streptococcus bovis including nutritional variant strains, or
- HACEKgroup, or
- Staphylococcus aureus, or
- Community-acquired enterococci, in the absence of a primary focus
- Microorganisms consistent with IE from persistently positive blood cultures defined as:
- Two positive cultures of blood samples drawn >12 hours apart, or
- Three or a majority of ≥four separate blood cultures (with first and last sample drawn at least one hour apart)
- IgG antibody titer for Q feverphase 1 antigen >1:800. This was previously a minor criterion
Evidence of endocardial involvement with positive echocardiogram is defined as
- Oscillating intracardiac mass on valve or supporting structures, in the path of regurgitant jets, or on implanted material in the absence of an alternative anatomic explanation, or
- Abscess, or
- New partial dehiscence of prosthetic valve or new valvular regurgitation (worsening or changing of preexisting murmur not sufficient)
Minor criteria
- Predisposing factor: known cardiac lesion, recreational drug injection
- Fever >38 °C
- Janeway lesions, conjunctivalhemorrhage
- Immunological phenomena: Roth's spots, Rheumatoid factor
- Microbiologic evidence: Positive blood culture (that doesn't meet a major criterion) or serologic evidence of infection with organism consistent with IE but not satisfying major criterion
Updated (2023) Modified Duke Criteria for Infective Endocarditis: Infective endocarditis (IE) is a life-threatening condition and the Duke criteria (established in 1994 and revised in 2000) has been fundamental for the diagnosis of the disease. However, the landscape of micro-biology, diagnostics, epidemiology, and treatment for lE has evolved significantly over the years. The 2023 modified Duke criteria address these changes: https://medicalstudyhub.com/updated-2023-modified-duke-criteria-for-infective-endocarditis/
Risk
Among people who do not
Among people who do use intravenous drugs and have a fever in the emergency department, there is about a 10% to 15% prevalence of endocarditis. This estimate is not substantially changed by whether the doctor believes the patient has a trivial explanation for their fever.[47] Weisse found that 13% of 121 patients had endocarditis.[45] Marantz also found a prevalence of endocarditis of 13% among such patients in the emergency department with fever.[47] Samet found a 6% incidence among 283 such patients, but after excluding patients with initially apparent major illness to explain the fever (including 11 cases of manifest endocarditis), there was a 7% prevalence of endocarditis.[46] During the Opioid epidemic in the United States, hospitals observed an increase in stroke associated with infective endocarditis.[56]
Among people with staphylococcal bacteremia (SAB), one study found a 29% prevalence of endocarditis in community-acquired SAB versus 5% in nosocomial SAB.[57] However, only 2% of strains were resistant to methicillin and so these numbers may be low in areas of higher resistance.[citation needed]
Prevention
Not all people with heart disease require antibiotics to prevent infective endocarditis. Heart diseases have been classified into high, medium and low risk of developing IE. Those falling into high risk category require IE prophylaxis before endoscopies and urinary tract procedures. Diseases listed under high risk include:[58]
- Prior endocarditis
- Unrepaired cyanotic congenital heart diseases
- Completely repaired congenital heart disease in their first 6 months
- Prosthetic heart valves or valves repaired with any prosthetic material
- Incompletely repaired congenital heart diseases
- Cardiac transplant valvulopathy
Following are the antibiotic regimens recommended by the American Heart Association for antibiotic prophylaxis:[40]
- Oral amoxicillin one hour before the procedure
- Intravenous or intramuscular ampicillin one hour before the procedure
- In patients allergic to penicillins
- Azithromycin or clarithromycin orally one hour before the procedure
- Cephalexin orally one hour before the procedure
- Clindamycin orally one hour before the procedure
In the UK, NICE clinical guidelines no longer advise prophylaxis because there is no clinical evidence that it reduces the incidence of IE and there are negative effects (e.g. allergy and increased bacterial resistance) of taking antibiotics that may outweigh the benefits.[59]
Antibiotics were historically commonly recommended to prevent IE in those with heart problems undergoing dental procedures (known as dental antibiotic prophylaxis). There is, however, insufficient evidence to support whether antibiotics are effective or ineffective at preventing IE when given prior to a dental procedures in people at high risk.[60] They are less commonly recommended for this procedure.[61]
In some countries e.g. the US, high risk patients may be given prophylactic antibiotics such as
Treatment
High-dose antibiotics are the cornerstone of treatment for infective endocarditis. These antibiotics are administered by the intravenous (IV) route to maximize diffusion of antibiotic molecules into vegetation(s) from the blood filling the chambers of the heart. This is necessary because neither the heart valves nor the vegetations adhering to them are supplied by blood vessels. Antibiotics are typically continued for two to six weeks depending on the characteristics of the infection and the causative microorganisms. Antibiotic treatment lowers the risk of embolic complications in people with infective endocarditis.[12]
In acute endocarditis, due to the fulminant inflammation, empirical antibiotic therapy is started immediately after the blood has been drawn for
Viridans group
Some people may be treated with a relatively shorter course of treatment[64] (two weeks) with benzyl penicillin IV if infection is caused by viridans group streptococci or Streptococcus bovis as long as the following conditions are met:
- Endocarditis of a native valve, not of a prosthetic valve
- A MIC ≤ 0.12 mg/l
- No complication such as arrhythmia, or pulmonary embolism occurs
- No evidence of extracardiac complication like septic thromboembolism
- No vegetations > 5 mm in diameter conduction defects
- Rapid clinical response and clearance of bloodstream infection
Additionally, oxacillin-susceptible Staphylococcus aureus native valve endocarditis of the right side can also be treated with a short 2-week course of a

The main indication for surgical treatment is regurgitation or stenosis. In active infective endocarditis, the surgery should remove enough leaflet tissue to ensure eradication of the infectious process.[66] Subsequent valve repair can be performed in limited disease.[66] Replacement of the valve with a mechanical or bioprosthetic artificial heart valve is necessary in certain situations:[67]
- Patients with significant valve stenosis or regurgitation causing heart failure
- Evidence of hemodynamic compromise in the form of elevated end-diastolic left ventricular or left atrial pressure or moderate to severe pulmonary hypertension
- Presence of intracardiac complications like paravalvular abscess, conduction defects or destructive penetrating lesions
- Recurrent septic embolidespite appropriate antibiotic treatment
- Large vegetations (> 10 mm)
- Persistently positive blood cultures despite appropriate antibiotic treatment
- Prosthetic valve dehiscence
- Relapsing infection in the presence of a prosthetic valve
- Abscess formation
- Early closure of mitral valve
- Infection caused by fungi or resistant Gram-negative bacteria.
The guidelines were recently updated by both the American College of Cardiology and the European Society of Cardiology. There was a recent meta-analysis published that showed surgical intervention at seven days or less is associated with lower mortality.[68]
Prognosis
Infective endocarditis is associated with 18% in-hospital mortality.[29] However, adult patients with congenital heart disease can have relatively lower mortality down to 5% due to younger age, right-sided endocarditis and management by multidisciplinary teams. As many as 50% of people with infective endocarditis may experience embolic complications.[12]
Epidemiology
In developed countries, the annual incidence of infective endocarditis is 3 to 9 cases per 100,000 persons.[43] Infective endocarditis occurs more often in men than in women.[12] There is an increased incidence of infective endocarditis in persons 65 years of age and older, which is probably because people in this age group have a larger number of risk factors for infective endocarditis. In recent years, over one-third of infective endocarditis cases in the United States was healthcare-associated.[43] Another trend observed in developed countries is that chronic rheumatic heart disease accounts for less than 10% of cases. Although a history of valve disease has a significant association with infective endocarditis, 50% of all cases develop in people with no known history of valvular disease.[citation needed]
History
Few diseases present greater difficulties in the way of diagnosis than malignant endocarditis, difficulties which in many cases are practically insurmountable. It is no disparagement to the many skilled physicians who have put their cases upon record to say that, in fully one-half the diagnosis was made post mortem.
— William Osler, 1885
Lazare Riviére first described infective endocarditis affecting the
References
- ^ a b c d e f g h i j k l m n o p q r "Infective Endocarditis – Cardiovascular Disorders". Merck Manuals Professional Edition. September 2017. Retrieved 11 December 2017.
- ^ PMID 27886786.
- ^ "Endocarditis". Mayo Clinic. Retrieved June 4, 2022.
- ^ "Endocarditis". Cleveland Clinic. Retrieved 2022-06-09.
- ^ PMID 36946284.
- ^ S2CID 24935834.
- PMID 26320109.
- ^ PMID 31312561.
- PMID 26163428.
- PMID 26320109.
- S2CID 25918810.
- ^ PMID 32299668.
- ^ ISBN 978-1-4160-2973-1.
- S2CID 10450799.
- ^ "Endocarditis". The Lecturio Medical Concept Library. Retrieved 18 July 2021.
- PMID 26320109.
- ^ PMID 19260876.
- PMID 27582414.
- PMID 26320109.
- ^ OCLC 54501403.
- PMID 26320109.
- PMID 19609257.
- ^ ISBN 978-1-4051-4166-6.
- PMID 28765422.
- PMID 24365290.
- ^ "Heart Valves and Infective Endocarditis". American Heart Association. Retrieved 18 July 2021.
- PMID 26320109.
- ISBN 978-3-319-27784-4.
- ^ PMID 19273776.
- ^ OCLC 61756542.
- ^ HACEK Group Infections at eMedicine
- ^ "Pseudomonas aeruginosa". Topics in Infectious Diseases Newsletter. August 2001. Archived from the original on July 24, 2008.
- PMID 15249410.
- S2CID 7571197.
- ^ PMID 12591823.
- PMID 16849716.
- PMID 15472298.
- PMID 19812281.
- PMID 19386832.
- ^ PMID 17446442.
- ^ PMID 27737409.
- PMID 19901894.
- ^ PMID 23574121.
- ^ PMID 8154507.
- ^ PMID 8452151.
- ^ PMID 2368794.
- ^ PMID 3579068.
- ^ PMID 2353860.
- ^ PMID 3827454.
- PMID 23601504.
- PMID 1856406.
- PMID 3345769.
- ^ a b c "UOTW #27 – Ultrasound of the Week". Ultrasound of the Week. 26 November 2014. Archived from the original on 9 May 2017. Retrieved 27 May 2017.
- ^ "UOTW #60 – Ultrasound of the Week". Ultrasound of the Week. 5 October 2015. Archived from the original on 9 May 2017. Retrieved 27 May 2017.
- PMID 10770721.
- PMID 30699043.
- PMID 16524411.
- PMID 26320109.
- ^ "Prophylaxis against infective endocarditis: Antimicrobial prophylaxis against infective endocarditis in adults and children undergoing interventional procedures". NICE Clinical Guidelines. National Institute for Health Care and Excellence (UK). March 2008. Archived from the original on 2013-01-26. Retrieved 2013-04-30.
- PMID 35536541.
- ^ "Prophylaxis against infective endocarditis: antimicrobial prophylaxis against infective endocarditis in adults and children undergoing interventional procedures". National Institute for Health and Care Excellence (NICE). 2008.
- ^ "Drug Prescribing". SDCEP. Retrieved 2018-01-25.
- PMID 19207079.
- ^ PMID 15956145.
- PMID 21404910.
- ^ PMID 28521809.
- PMID 18848134.
- S2CID 12007082.
