Acute myeloid leukemia
This article needs to be updated. (July 2021) |
| Acute myeloid leukemia | |
|---|---|
| Other names | Acute myelogenous leukemia, acute nonlymphocytic leukemia (ANLL), acute myeloblastic leukemia, acute granulocytic leukemia Five-year survival ~29% (US, 2017)[2] |
| Frequency | 1 million (2015)[4] |
| Deaths | 147,100 (2015)[5] |
Acute myeloid leukemia (AML) is a
The first-line treatment of AML is usually
In 2015, AML affected about one million people, and resulted in 147,000 deaths globally.[4][5] It most commonly occurs in older adults.[2] Males are affected more often than females.[2] The five-year survival rate is about 35% in people under 60 years old and 10% in people over 60 years old.[3] Older people whose health is too poor for intensive chemotherapy have a typical survival of five to ten months.[3] It accounts for roughly 1.1% of all cancer cases, and 1.9% of cancer deaths in the United States.[2]
Signs and symptoms

Most signs and symptoms of AML are caused by the crowding out in
Some people with AML may experience swelling of the gums because of infiltration of leukemic cells into the gum tissue.[6] Involvement of other parts of the body such as the gastrointestinal tract, respiratory tract and other parts is possible but less common.[7] One area which has particular importance for treatment is whether there is involvement of the meninges around the central nervous system.[7]
Risk factors
Most cases of AML do not have exposure to any identified risk factors.[8][9] However, a number of risk factors for developing AML have been identified. These include other blood disorders, chemical exposures, ionizing radiation, and genetic risk factors.[8] Where a defined exposure to past chemotherapy, radiotherapy, toxin or hematologic malignancy is known, this is termed secondary AML.[10]
Other blood disorders
Other blood disorders, particularly
Chemical exposure
Exposure to
Other chemical exposures associated with the development of AML include benzene, chloramphenicol and phenylbutazone.[9]
Radiation
High amounts of
Genetics
Most cases of AML arise spontaneously, however there are some genetic mutations associated with an increased risk.
Other factors
Being
Pathophysiology

The malignant cell in AML is the
Specific
The
Many cells develop mutations in genes that affect
Diagnosis

A
Bone marrow is examined under
Cytochemical stains on blood and bone marrow smears are helpful in the distinction of AML from ALL, and in subclassification of AML. The combination of a
The standard classification scheme for AML is the
Because
World Health Organization
The WHO classification of AML attempts to be more clinically useful and to produce more meaningful prognostic information than the FAB criteria. The French-American-British (FAB) classification system is based on morphology to define specific immunotypes. The World Health Organization (WHO) classification reviews chromosome translocations and evidence of dysplasia.[32] SEE French-American-British (FAB) classification system.
Each of the WHO categories contains numerous descriptive subcategories of interest to the
The revised fourth edition of the WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues
| Name | Description | ICD-O
|
|---|---|---|
| Acute myeloid leukemia with recurrent genetic abnormalities | Includes:[38]
|
Multiple |
| AML with myelodysplasia-related changes | This category includes people who have had a prior documented cell lines).[39]
People who have previously received chemotherapy or radiation treatment for a non-MDS/MPD disease, and people who have genetic markers associated with AML with recurrent genetic abnormalities, are excluded from this category. This category of AML occurs most often in elderly people and often has a worse prognosis. Cytogenetic markers for AML with myelodysplasia-related changes include:[40]
|
M9895/3 |
| Therapy-related myeloid neoplasms | This category includes people who have had prior chemotherapy and/or radiation and subsequently develop AML or MDS. These leukemias may be characterized by specific chromosomal abnormalities, and often carry a worse prognosis.[41] | M9920/3 |
| Myeloid sarcoma | This category includes myeloid sarcoma.[42] | |
| Myeloid proliferations related to Down syndrome | This category includes "transient abnormal myelopoiesis" and "myeloid leukemia associated with Down syndrome". In young children, myeloid leukemia associated with Down syndrome has a much better prognosis than other types of childhood AML. The prognosis in older children is similar to conventional AML.[43] | |
| AML not otherwise categorized | Includes subtypes of AML that do not fall into the above categories:[44]
|
M9861/3 |

Acute leukemias of ambiguous lineage (also known as
French-American-British
The
The FAB system divides AML into eight subtypes, M0 through to M7, based on the type of cell from which the leukemia developed and its degree of maturity. AML of types M0 to M2 may be called acute myeloblastic leukemia. Classification is done by examining the appearance of the
Six FAB subtypes (M1 through to M6) were initially proposed in 1976,[48] although later revisions added M7 in 1985[49] and M0 in 1987.[50]
| Type | Name | Cytogenetics | Percentage of adults with AML | Immunophenotype[51] | ||||
|---|---|---|---|---|---|---|---|---|
| CD14 | CD15
|
CD33 | HLA-DR | Other | ||||
| M0 | acute myeloblastic leukemia, minimally differentiated
|
5%[52] | − better source needed ]
|
−[53] | +[53] | +[53] | MPO −[54] | |
| M1 | acute myeloblastic leukemia, without maturation
|
15%[52] | − | − | + | + | MPO +[54] | |
| M2 | acute myeloblastic leukemia, with granulocytic maturation
|
t(8;21)(q22;q22), t(6;9) | 25%[52] | − | + | + | + | |
| M3 | promyelocytic, or acute promyelocytic leukemia (APL) | t(15;17) | 10%[52] | − | + | + | − | |
| M4 | acute myelomonocytic leukemia | inv(16)(p13q22), del(16q) | 20%[52] | <45% | + | + | + | |
| M4eo | myelomonocytic together with bone marrow eosinophilia | inv(16), t(16;16) | 5%[52] | +/−[55] | +[56] | +[56] | CD2+[56] | |
| M5 | acute monoblastic leukemia (M5a) or acute monocytic leukemia (M5b)
|
del (11q), t(9;11), t(11;19) | 10%[52] | >55% | + | + | + | |
| M6 | acute erythroid leukemias, including erythroleukemia (M6a) and very rare pure erythroid leukemia (M6b) | 5%[52] | − | +/− | +/− | +/− | Glycophorin + | |
| M7 | acute megakaryoblastic leukemia | t(1;22) | 5%[52] | − | − | + | +/− | CD41/CD61+ |
The morphologic subtypes of AML also include rare types not included in the FAB system, such as acute basophilic leukemia, which was proposed as a ninth subtype, M8, in 1999.[57]
Treatment
First-line treatment of AML consists primarily of
Induction
The goal of the induction phase is to reach a complete remission. Complete remission does not mean the disease has been cured; rather, it signifies no disease can be detected with available diagnostic methods.
Consolidation
Even after complete remission is achieved, leukemic cells likely remain in numbers too small to be detected with current diagnostic techniques. If no consolidation therapy or further postremission is given, almost all people with AML will eventually relapse.[58]
The specific type of postremission therapy is individualized based on a person's prognostic factors (see above) and general health.[58] For good-prognosis leukemias (i.e. inv(16), t(8;21), and t(15;17)), people will typically undergo an additional three to five courses of intensive chemotherapy, known as consolidation chemotherapy. This generally involves cytarabine, with the doses administered being higher in younger patients, who are less likely to develop toxicity related to this treatment.[58]
Stem cell transplantation
Target therapy
Target therapy is a type of treatment that uses drugs or other substances to target specific molecules that cancer cells need to survive and spread. Targeted therapies work in different ways to treat cancer. Some stop cancer cells from growing by interrupting signals that cause them to grow and divide, stopping signals that help form blood vessels, delivering cell-killing substances to cancer cells, or starving cancer cells of hormones they need to grow. Other targeted therapies help the immune system kill cancer cells or directly cause cancer cell death. Most targeted therapies are either small-molecule drugs or monoclonal antibodies. Also called molecularly targeted therapy.[65]
Supportive treatment
Support is necessary throughout treatment because of problems associated with AML and also arising from treatment.[66] Blood transfusions, including of red blood cells and platelets, are necessary to maintain health levels, preventing complications of anemia (from low red blood cells) and bleeding (from low platelets).[66] AML leads to an increased risk of infections, particularly drug-resistant strains of bacteria and fungi.[60] Antibiotics and antifungals can be used both to treat and to prevent these infections, particularly quinolones.[60][67]
Adding aerobic physical exercises to the standard of care may result in little to no difference in the mortality, in the quality of life and in the physical functioning. These exercises may result in a slight reduction in depression. Furthermore, aerobic physical exercises probably reduce fatigue.[68]
Recent research into the role that epigenetic regulators play in hematopoietic malignancies has yielded new insights in the development of targeted epigenetic therapies as a supportive treatment for AML. The FDA has approved certain epigenetic modifying drugs like ivosidenib and enasidenib, which are used in patients that can no longer receive intensive induction chemotherapy; specifically, they are involved in the therapy of IDH1 and IDH2 mutations. Further research must be done to prove the efficacy of epigenetic treatments, but the development of new epigenetic therapies along with immunotherapies holds potential in the future treatment of AML.[69]
In pregnancy
AML is rare in pregnancy, affecting about 1 in 75,000 to 100,000 pregnant women.[70] It is diagnosed and treated similarly to AML in non pregnancy, with a recommendation that it is treated urgently.[70] However, treatment has significant implications for the pregnancy. First trimester pregnancy is considered unlikely to be viable; pregnancy during weeks 24 – 36 requires consideration of the benefits of chemotherapy to the mother against the risks to the fetus; and there is a recommendation to consider delaying chemotherapy in very late pregnancy (> 36 weeks).[70] Some elements of supportive care, such as which antibiotics to prevent or treat infections, also change in pregnancy.[70]
Medication
Olutasidenib (Rezlidhia) was approved for medical use in the United States in December 2022.[71]
Prognosis
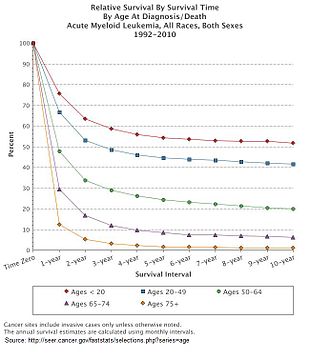

Multiple factors influence prognosis in AML, including the presence of specific mutations, and a person with AML's age. In the United States between 2011 and 2016, the median survival of a person with AML was 8.5 months, with the 5 year survival being 24%.[10] This declines with age, with the poorer prognosis being associated with an age greater than 65 years, and the poorest prognosis seen in those aged 75–84.[10]
As of 2001, cure rates in clinical trials have ranged from 20 to 45%;[72][73] although clinical trials often include only younger people and those able to tolerate aggressive therapies. The overall cure rate for all people with AML (including the elderly and those unable to tolerate aggressive therapy) is likely lower. Cure rates for APL can be as high as 98%.[74]
Subtypes
Secondary AML has a worse prognosis, as does treatment-related AML arising after chemotherapy for another previous malignancy. Both of these entities are associated with a high rate of unfavorable genetic mutations.[10]
Cytogenetics
Different genetic mutations are associated with a difference in outcomes. Certain cytogenetic abnormalities are associated with very good outcomes (for example, the (15;17) translocation in APL). About half of people with AML have "normal" cytogenetics; they fall into an intermediate risk group. A number of other cytogenetic abnormalities are known to associate with a poor prognosis and a high risk of relapse after treatment.[75][76][77]
A large number of molecular alterations are under study for their prognostic impact in AML. However, only FLT3-ITD, NPM1, CEBPA and c-KIT are currently included in validated international risk stratification schema. These are expected to increase rapidly in the near future.
Researchers are investigating the clinical significance of
Other prognostic factors
Elevated lactate dehydrogenase level were also associated with poorer outcomes.[78] Use of tobacco is associated with a person having a poorer prognosis,[10] and people who are married and live together have a better prognosis.[10] People who are treated at place with a higher volume of AML have a better prognosis than those who are treated at those in the lowest quartile.[10] As with most forms of cancer, performance status (i.e. the general physical condition and activity level of the person) plays a major role in prognosis as well.[citation needed]
For people in remission after induction chemotherapy, residual leukemic cells (minimal residual disease) are associated with higher relapse rates and decreased survival.[79]
Epidemiology
AML is a relatively rare cancer. There were 19,950 new cases in the United States in 2016.[80] In 2018, AML accounted for 1.2% of all cancer deaths in the United States.[9]
The incidence of AML increases with age and varies between countries.[10] The median age when AML is diagnosed ranges between 63 and 71 years in the UK, Canada, Australia and Sweden, compared with 40 to 45 years in India, Brazil and Algeria.[10]
AML accounts for about 90% of all acute leukemias in adults, but is rare in children.[needs update][81] The rate of therapy-related AML (AML caused by previous chemotherapy) is expected to rise with an increase in the use of chemotherapy, an ageing population and more patients surviving their initial chemotherapy treatment; therapy-related disease accounts for just under 10% of all cases of AML.[82] AML is slightly more common in men, with a male-to-female ratio of 1.3:1[83] to 1.4:1.[10] Incidence is also seen to differ by ethnicity, with caucasians having higher recorded incidences and the lowest recorded incidences being in Pacific Islanders and native Alaskans.[10]
In the UK, AML accounts for 31% of all leukemia cases, and around 3,100 people were diagnosed with the disease each year in 2016–2018.[84]
History
The first published description of a case of leukemia in medical literature dates to 1827 when French physician
The term "leukemia" was coined by
Further advances in the understanding of AML occurred rapidly with the development of new technology. In 1877,
In 2008, AML became the first cancer genome to be fully sequenced. DNA extracted from leukemic cells were compared to unaffected skin.[92] The leukemic cells contained acquired mutations in several genes that had not previously been associated with the disease.
References
- ^ a b c d e f g h i j k l m "Adult Acute Myeloid Leukemia Treatment". National Cancer Institute. 6 March 2017. Retrieved 19 December 2017.
- ^ a b c d e "Acute Myeloid Leukemia – Cancer Stat Facts". NCI. Retrieved 10 May 2017.
- ^ S2CID 40314260.
- ^ PMID 27733282.
- ^ PMID 27733281.
- ^ )
- ^ OCLC 1176325543.
- ^ S2CID 4028327.
- ^ a b c d e f g Harrison's 2018, p. 739.
- ^ S2CID 155630692.
- PMID 2752119.
- S2CID 22100511.
- PMID 5932020.
- S2CID 20643232.
- ^ a b Harrison's 2018, p. 740.
- ^ a b c d Hoffbrand's 2016, pp. 146–7.
- ^ a b Harrison's 2018, p. 742.
- ISBN 978-0-7817-3650-3.
- ^ Harrison's 2018, p. 741.
- ^ a b c d e f Harrison's 2018, p. 743.
- ^ PMID 25836588.
- PMID 24880135.
- PMID 19752007.
- S2CID 30419482.
- S2CID 9413654.
- ISBN 978-1-4963-6713-6.
- ^ ISBN 978-1-118-81733-9.
- ISBN 978-92-832-4494-3.
- ^ Foucar K. "Bone Marrow Pathology" (PDF) (3rd ed.). ASCP. Archived from the original (PDF) on 19 March 2013. Retrieved 18 March 2016.
- S2CID 11711013.
- S2CID 8271000.
- ^ "Acute Myeloid Leukemia (AML) Subtypes and Prognostic Factors".
- S2CID 18338178.
- S2CID 18338178.
- ^ Wintrobe's 2018, Chapter 74, sec. "Acute myeloid leukemia".
- PMID 35732831.
- S2CID 253063457.
- ^ Campo 2017, p. 130–145.
- ^ Campo 2017, p. 150.
- ^ Campo 2017, p. 150–152.
- ^ Campo 2017, p. 153–155.
- ^ Campo 2017, p. 167.
- ^ Campo 2017, p. 169–171.
- ^ Campo 2017, p. 156–166.
- S2CID 53523035.
- ^ Wintrobe's 2018, Chapter 74, sec. "Acute leukemias of ambiguous lineage".
- ^ Wintrobe's 2018, Chapter 74, sec. "Introduction".
- S2CID 9985915.
- PMID 4040724.
- PMID 3663939.
- OCLC 85862340.
- ^ a b c d e f g h i Seiter K, Jules EH (20 May 2011). "Acute Myeloid Leukemia Staging". Retrieved 26 August 2011.
- ^ a b c d Mihova D. "Leukemia acute – Acute myeloid leukemia with minimal differentiation (FAB AML M0)". Pathology Outlines. Topic Completed: 1 March 2013. Minor changes: 19 November 2019
- ^ PMID 23730015.
- PMID 8098967.
- ^ OCLC 432709321.
- PMID 10037024.
- ^ a b c d e f g h Harrison's 2018, p. 743-5.
- PMID 23631653.
- ^ PMID 27895058.
- ^ a b Hoffbrand's 2016, pp. 152–3.
- ^ Harrison's 2018, p. 748.
- PMID 30080246.
- ^ a b c Harrison's 2018, p. 745.
- ^ "Acute Myeloid Leukemia Treatment - NCI". www.cancer.gov. 7 June 2023. Retrieved 14 June 2023.
- ^ a b Harrison's 2018, p. 747.
- S2CID 1088077.
- PMID 30702150.
- PMID 34824880.
- ^ S2CID 11298224.
- ^ "Rigel Announces U.S. FDA Approval of Rezlidhia (olutasidenib) for the Treatment of Adult Patients with Relapsed or Refractory Acute Myeloid Leukemia with a Susceptible IDH1 Mutation" (Press release). Rigel Pharmaceuticals. 1 December 2022. Retrieved 2 December 2022 – via PR Newswire.
- PMID 9834301.
- S2CID 34233226.
- PMID 10942364. Archived from the originalon 27 May 2010. Retrieved 17 March 2008.
- S2CID 27266593.
- PMID 11110676.
- S2CID 16003833.
- PMID 12525517.
- PMID 29601269.
- ISBN 9781259644030.
- S2CID 5659023.
- S2CID 246443548.
- S2CID 22441565.
- ^ "Acute myeloid leukaemia (AML) statistics". Cancer Research UK. 14 May 2015. Retrieved 3 April 2022.
- ISBN 978-0-443-06629-0.
- ^ Bennett JH (1845). "Two cases of hypertrophy of the spleen and liver, in which death took place from suppuration of blood". Edinburgh Med Surg J. 64: 413.
- ^ Virchow R (1856). "Die Leukämie". In Virchow R (ed.). Gesammelte Abhandlungen zur Wissenschaftlichen Medizin (in German). Frankfurt: Meidinger. p. 190.
- ^ Ebstein W (1889). "Über die acute Leukämie und Pseudoleukämie". Deutsch Arch Klin Med. 44: 343.
- ^ Mosler F (1876). "Klinische Symptome und Therapie der medullären Leukämie". Berl Klin Wochenschr. 13: 702.
- S2CID 71572772.
- S2CID 2788589.
- PMID 18987736.
