Addition reaction
- العربية
- Bosanski
- Català
- Čeština
- Dansk
- Deutsch
- Eesti
- Ελληνικά
- Español
- Euskara
- فارسی
- Français
- Gaeilge
- 한국어
- हिन्दी
- Hrvatski
- Bahasa Indonesia
- Italiano
- עברית
- Magyar
- മലയാളം
- Bahasa Melayu
- Nederlands
- 日本語
- Norsk bokmål
- Norsk nynorsk
- پښتو
- Polski
- Português
- Română
- Русский
- Simple English
- Slovenčina
- Slovenščina
- کوردی
- Српски / srpski
- Srpskohrvatski / српскохрватски
- Suomi
- Svenska
- தமிழ்
- Türkçe
- Українська
- Tiếng Việt
- 中文
In organic chemistry, an addition reaction is an organic reaction where two or more molecules combine to form a larger one (the adduct).[1][2]
Addition reactions are limited to chemical compounds that have
An addition reaction is the reverse of an elimination reaction. For instance, the hydration of an alkene to an alcohol is reversed by dehydration.
There are two main types of
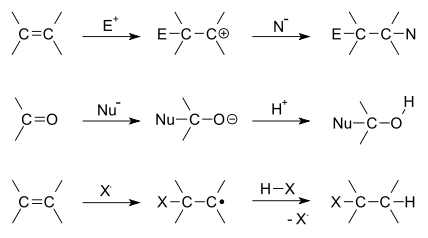
Depending on the product structure, it could promptly react further to eject a leaving group to give the addition–elimination reaction sequence.
Addition reactions are useful in
Likewise hydrogen addition often proceeds on all double-bonds of a molecule, and thus gives a count of the number of a double and triple bonds through stoichiometry:
2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/43dc6b4900e61b66f9297e7ec4c734d82bc984bd)
References
- ISBN 0-205-05838-8.
- OCLC 642506595.
External links
 Quotations related to Addition reaction at Wikiquote
Quotations related to Addition reaction at Wikiquote
Basic reaction mechanisms | |
|---|---|
| Nucleophilic substitutions | |
| Electrophilic substitutions | |
| Elimination reactions |
|
| Addition reactions | |
| Unimolecular reactions |
|
| Electron/Proton transfer reactions | |
| Medium effects | |
| Related topics | |
| Chemical kinetics | |
This organic chemistry article is a stub. You can help Wikipedia by expanding it. |

![{\displaystyle {\ce {RR'C=CR''R'''+Br2(orange-brown)->[{} \atop {\ce {CCl4}}]RR'CBr-BrCR''R'''(typically\ colorless)}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/ff968293b8be0a13e32cd53aad87367d5ff4cfda)