Adenine

| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
9H-Purin-6-amine | |||
| Other names
6-Aminopurine
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 608603 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
ECHA InfoCard
|
100.000.724 | ||
| EC Number |
| ||
| 3903 | |||
IUPHAR/BPS |
|||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C5H5N5 | |||
| Molar mass | 135.13 g/mol | ||
| Appearance | white to light yellow, crystalline | ||
| Density | 1.6 g/cm3 (calculated) | ||
| Melting point | 360 to 365 °C (680 to 689 °F; 633 to 638 K) decomposes | ||
| 0.103 g/100 mL | |||
| Solubility | negligible in aqua ammonia
| ||
| Acidity (pKa) | 4.15 (secondary), 9.80 (primary)[1] | ||
| Thermochemistry | |||
Heat capacity (C)
|
147.0 J/(K·mol) | ||
Std enthalpy of (ΔfH⦵298)formation |
96.9 kJ/mol | ||
| Hazards | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
227 mg/kg (rat, oral) | ||
| Safety data sheet (SDS) | MSDS | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Adenine (/ˈædɪnɪn/) (symbol A or Ade) is a purine nucleobase. It is one of the four nucleobases in the nucleic acids of DNA, the other three being guanine (G), cytosine (C), and thymine (T). Adenine derivatives have various roles in biochemistry including cellular respiration, in the form of both the energy-rich adenosine triphosphate (ATP) and the cofactors nicotinamide adenine dinucleotide (NAD), flavin adenine dinucleotide (FAD) and Coenzyme A. It also has functions in protein synthesis and as a chemical component of DNA and RNA.[2] The shape of adenine is complementary to either thymine in DNA or uracil in RNA.
The adjacent image shows pure adenine, as an independent molecule. When connected into DNA, a covalent bond is formed between deoxyribose sugar and the bottom left nitrogen (thereby removing the existing hydrogen atom). The remaining structure is called an adenine residue, as part of a larger molecule. Adenosine is adenine reacted with ribose, as used in RNA and ATP; Deoxyadenosine is adenine attached to deoxyribose, as used to form DNA.
Structure

Adenine forms several tautomers, compounds that can be rapidly interconverted and are often considered equivalent. However, in isolated conditions, i.e. in an inert gas matrix and in the gas phase, mainly the 9H-adenine tautomer is found.[3][4]
Biosynthesis
Manufacturing method
Patented Aug. 20, 1968, the current recognized method of industrial-scale production of adenine is a modified form of the formamide method. This method heats up formamide under 120 degree Celsius conditions within a sealed flask for 5 hours to form adenine. The reaction is heavily increased in quantity by using a phosphorus oxychloride (phosphoryl chloride) or phosphorus pentachloride as an acid catalyst and sunlight or ultraviolet conditions. After the 5 hours have passed and the formamide-phosphorus oxychloride-adenine solution cools down, water is put into the flask containing the formamide and now-formed adenine. The water-formamide-adenine solution is then poured through a filtering column of activated charcoal. The water and formamide molecules, being small molecules, will pass through the charcoal and into the waste flask; the large adenine molecules, however, will attach or "adsorb" to the charcoal due to the van der Waals forces that interact between the adenine and the carbon in the charcoal. Because charcoal has a large surface area, it's able to capture the majority of molecules that pass a certain size (greater than water and formamide) through it. To extract the adenine from the charcoal-adsorbed adenine, ammonia gas dissolved in water (aqua ammonia) is poured onto the activated charcoal-adenine structure to liberate the adenine into the ammonia-water solution. The solution containing water, ammonia, and adenine is then left to air dry, with the adenine losing solubility due to the loss of ammonia gas that previously made the solution basic and capable of dissolving adenine, thus causing it to crystallize into a pure white powder that can be stored.[5]
Function
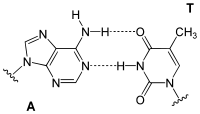
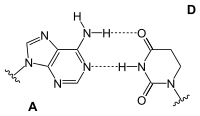
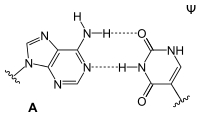
Adenine is one of the two purine nucleobases (the other being guanine) used in forming nucleotides of the nucleic acids. In DNA, adenine binds to thymine via two hydrogen bonds to assist in stabilizing the nucleic acid structures. In RNA, which is used for protein synthesis, adenine binds to uracil.
|

|
|
|
| A-T-Base-pair (DNA) | A-U-Base-pair (RNA) | A-D-Base-pair (RNA) | A-Ψ-Base-pair (RNA) |
Adenine forms
History

In older literature, adenine was sometimes called Vitamin B4.
It was named in 1885 by Albrecht Kossel after Greek ἀδήν aden "gland", in reference to the pancreas, from which Kossel's sample had been extracted.[7][8]
Experiments performed in 1961 by
On August 8, 2011, a report, based on
References
- ^ Dawson, R.M.C., et al., Data for Biochemical Research, Oxford, Clarendon Press, 1959.
- ^ "MedlinePlus: Genetics". medlineplus.gov. Retrieved 2022-12-23.
- doi:10.1039/b204595h.
- ISSN 0584-8539.
- ^ [1], "Process for preparing adenine", issued 1966-11-10
- PMID 16744538.
- ^ texte, Deutsche chemische Gesellschaft Auteur du (1885-01-01). "Berichte der Deutschen chemischen Gesellschaft zu Berlin". Gallica. Retrieved 2022-12-23.
- ^ "adenine | Etymology, origin and meaning of adenine by etymonline". www.etymonline.com. Retrieved 2022-12-23.
- PMID 13731263.
- S2CID 21941930.
- PMID 21836052.
- ^ Steigerwald, John (8 August 2011). "NASA Researchers: DNA Building Blocks Can Be Made in Space". NASA. Archived from the original on 2015-06-23. Retrieved 2011-08-10.
- ^ ScienceDaily Staff (9 August 2011). "DNA Building Blocks Can Be Made in Space, NASA Evidence Suggests". ScienceDaily. Retrieved 2011-08-09.
- ^ Williams P (August 18, 2011). "Physicists Uncover New Data On Adenine, a Crucial Building Block of Life". Science Daily. Retrieved 2011-09-01.
- PMID 21804965.




