Adipose tissue
| Adipose tissue | |
|---|---|
 Pig belly fat (white) | |
 Adipose tissue is one of the main types of connective tissue. | |
| Pronunciation | /ˈædɪˌpoʊs/ ⓘ |
| Identifiers | |
| MeSH | D000273 |
| FMA | 20110 |
| Anatomical terminology | |
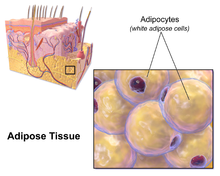
Adipose tissue (also known as body fat or simply fat) is a loose
Previously treated as being hormonally inert, in recent years adipose tissue has been recognized as a major
Adipose tissue is derived from preadipocytes and its formation appears to be controlled in part by the
Anatomical features

In humans, adipose tissue is located: beneath the
Adipose tissue contains many small blood vessels. In the integumentary system, which includes the skin, it accumulates in the deepest level, the subcutaneous layer, providing insulation from heat and cold. Around organs, it provides protective padding. However, its main function is to be a reserve of lipids, which can be oxidised to meet the energy needs of the body and to protect it from excess glucose by storing triglycerides produced by the liver from sugars, although some evidence suggests that most lipid synthesis from carbohydrates occurs in the adipose tissue itself.[6] Adipose depots in different parts of the body have different biochemical profiles. Under normal conditions, it provides feedback for hunger and diet to the brain.
Mice
Mice have eight major adipose depots, four of which are within the
The two superficial depots are the paired inguinal depots, which are found anterior to the upper segment of the hind limbs (underneath the skin) and the subscapular depots, paired medial mixtures of brown adipose tissue adjacent to regions of white adipose tissue, which are found under the skin between the dorsal crests of the scapulae. The layer of brown adipose tissue in this depot is often covered by a "frosting" of white adipose tissue; sometimes these two types of fat (brown and white) are hard to distinguish. The inguinal depots enclose the inguinal group of lymph nodes. Minor depots include the pericardial, which surrounds the heart, and the paired popliteal depots, between the major muscles behind the knees, each containing one large lymph node.[7] Of all the depots in the mouse, the gonadal depots are the largest and the most easily dissected,[8] comprising about 30% of dissectible fat.[9]
Obesity
In an
Visceral fat
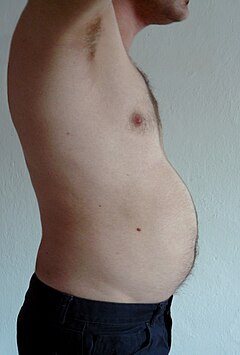
Visceral fat or abdominal fat
An excess of visceral fat is known as
Men are more likely to have fat stored in the abdomen due to sex hormone differences. Estrogen (female sex hormone) causes fat to be stored in the buttocks, thighs, and hips in women.[21][22] When women reach menopause and the estrogen produced by the ovaries declines, fat migrates from the buttocks, hips and thighs to the waist;[23] later fat is stored in the abdomen.[12]
Visceral fat can be caused by excess cortisol levels.
Epicardial fat
Subcutaneous fat
Most of the remaining nonvisceral fat is found just below the skin in a region called the
Like all other fat organs, subcutaneous fat is an active part of the endocrine system, secreting the hormones leptin and resistin.[35]
The relationship between the subcutaneous adipose layer and total body fat in a person is often modelled by using regression equations. The most popular of these equations was formed by Durnin and Wormersley, who rigorously tested many types of skinfold, and, as a result, created two formulae to calculate the body density of both men and women. These equations present an inverse correlation between skinfolds and body density—as the sum of skinfolds increases, the body density decreases.[39]
Factors such as sex, age, population size or other variables may make the equations invalid and unusable, and, as of 2012[update], Durnin and Wormersley's equations remain only estimates of a person's true level of fatness. New formulae are still being created.[39]
Marrow fat
Marrow fat, also known as
Ectopic fat
Ectopic fat is the storage of
The specific cause for the accumulation of ectopic fat is unknown. The cause is likely a combination of genetic, environmental, and behavioral factors that are involved in excess energy intake and decreased physical activity. Substantial weight loss can reduce ectopic fat stores in all organs and this is associated with an improvement of the function of those organs.[46]
In the latter case, non-invasive weight loss interventions like diet or exercise can decrease ectopic fat (particularly in heart and liver) in overweight or obese children and adults.[47][48]
Physiology
There is a constant flux of FFAs entering and leaving adipose tissue.[2] The net direction of this flux is controlled by insulin and leptin—if insulin is elevated, then there is a net inward flux of FFA, and only when insulin is low can FFA leave adipose tissue. Insulin secretion is stimulated by high blood sugar, which results from consuming carbohydrates.[50]
In humans, lipolysis (hydrolysis of triglycerides into free fatty acids) is controlled through the balanced control of lipolytic
Fat cells have an important
Recent advances in biotechnology have allowed for the harvesting of
Adipose tissue is a major peripheral source of aromatase in both males and females, contributing to the production of estradiol.[58]
- Adiponectin
- Resistin
- Plasminogen activator inhibitor-1 (PAI-1)
- TNFα
- IL-6
- Leptin
- Estradiol (E2)
Adipose tissues also secrete a type of cytokines (cell-to-cell signalling proteins) called adipokines (adipose cytokines), which play a role in obesity-associated complications. Perivascular adipose tissue releases adipokines such as adiponectin that affect the contractile function of the vessels that they surround.[1][59]
Brown fat

Brown fat or
BAT activation may also occur in response to overfeeding.[62] UCP1 activity is stimulated by long chain fatty acids that are produced subsequent to β-adrenergic receptor activation.[60] UCP1 is proposed to function as a fatty acid proton symporter, although the exact mechanism has yet to be elucidated.[63] In contrast, UCP1 is inhibited by ATP, ADP, and GTP.[64]
Attempts to simulate this process pharmacologically have so far been unsuccessful. Techniques to manipulate the differentiation of "brown fat" could become a mechanism for weight loss therapy in the future, encouraging the growth of tissue with this specialized metabolism without inducing it in other organs. A review on the eventual therapeutic targeting of brown fat to treat human obesity was published by Samuelson and Vidal-Puig in 2020.[65]
Until recently, brown adipose tissue in humans was thought to be primarily limited to infants, but new evidence has overturned that belief. Metabolically active tissue with temperature responses similar to brown adipose was first reported in the neck and trunk of some human adults in 2007,
Beige fat and WAT browning
Browning of WAT, also referred to as "beiging", occurs when adipocytes within WAT depots develop features of BAT. Beige adipocytes take on a multilocular appearance (containing several lipid droplets) and increase expression of uncoupling protein 1 (UCP1).[70] In doing so, these normally energy-storing adipocytes become energy-releasing adipocytes.
The calorie-burning capacity of brown and beige fat has been extensively studied as research efforts focus on therapies targeted to treat obesity and diabetes. The drug
Cold is a primary regulator of BAT processes and induces WAT browning. Browning in response to chronic cold exposure has been well documented and is a reversible process. A study in mice demonstrated that cold-induced browning can be completely reversed in 21 days, with measurable decreases in UCP1 seen within a 24-hour period.[72] A study by Rosenwald et al. revealed that when the animals are re-exposed to a cold environment, the same adipocytes will adopt a beige phenotype, suggesting that beige adipocytes are retained.[73]
Transcriptional regulators, as well as a growing number of other factors, regulate the induction of beige fat. Four regulators of transcription are central to WAT browning and serve as targets for many of the molecules known to influence this process.[74] These include peroxisome proliferator-activated receptor gamma (PPARγ), PRDM16,[75] peroxisome proliferator-activated receptor gamma coactivator 1 alpha (PGC-1α), and Early B-Cell Factor-2 (EBF2).[76][77][78]
The list of molecules that influence browning has grown in direct proportion to the popularity of this topic and is constantly evolving as more knowledge is acquired. Among these molecules are
Genomics and bioinformatics tools to study browning
Due to the complex nature of adipose tissue and a growing list of browning regulatory molecules, great potential exists for the use of bioinformatics tools to improve study within this field. Studies of WAT browning have greatly benefited from advances in these techniques, as beige fat is rapidly gaining popularity as a therapeutic target for the treatment of obesity and diabetes.
DNA microarray is a bioinformatics tool used to quantify expression levels of various genes simultaneously, and has been used extensively in the study of adipose tissue. One such study used microarray analysis in conjunction with Ingenuity IPA software to look at changes in WAT and BAT gene expression when mice were exposed to temperatures of 28 and 6 °C.[84] The most significantly up- and downregulated genes were then identified and used for analysis of differentially expressed pathways. It was discovered that many of the pathways upregulated in WAT after cold exposure are also highly expressed in BAT, such as oxidative phosphorylation, fatty acid metabolism, and pyruvate metabolism.[84] This suggests that some of the adipocytes switched to a beige phenotype at 6 °C. Mössenböck et al. also used microarray analysis to demonstrate that insulin deficiency inhibits the differentiation of beige adipocytes but does not disturb their capacity for browning.[85] These two studies demonstrate the potential for the use of microarray in the study of WAT browning.
RNA sequencing (RNA-Seq) is a powerful computational tool that allows for the quantification of RNA expression for all genes within a sample. Incorporating RNA-Seq into browning studies is of great value, as it offers better specificity, sensitivity, and a more comprehensive overview of gene expression than other methods. RNA-Seq has been used in both human and mouse studies in an attempt characterize beige adipocytes according to their gene expression profiles and to identify potential therapeutic molecules that may induce the beige phenotype. One such study used RNA-Seq to compare gene expression profiles of WAT from wild-type (WT) mice and those overexpressing Early B-Cell Factor-2 (EBF2). WAT from the transgenic animals exhibited a brown fat gene program and had decreased WAT specific gene expression compared to the WT mice.[86] Thus, EBF2 has been identified as a potential therapeutic molecule to induce beiging.
Chromatin immunoprecipitation with sequencing
Genetics
The thrifty gene hypothesis (also called the famine hypothesis) states that in some populations the body would be more efficient at retaining fat in times of plenty, thereby endowing greater resistance to starvation in times of food scarcity. This hypothesis, originally advanced in the context of glucose metabolism and insulin resistance, has been discredited by physical anthropologists, physiologists, and the original proponent of the idea himself with respect to that context, although according to its developer it remains "as viable as when [it was] first advanced" in other contexts.[87][88]
In 1995,
Leptin, however, plays a different role in diet-induced obesity in rodents and humans. Because adipocytes produce leptin, leptin levels are elevated in the obese. However, hunger remains, and—when leptin levels drop due to weight loss—hunger increases. The drop of leptin is better viewed as a starvation signal than the rise of leptin as a satiety signal.[92] However, elevated leptin in obesity is known as leptin resistance. The changes that occur in the hypothalamus to result in leptin resistance in obesity are currently the focus of obesity research.[93]
Gene defects in the leptin gene (ob) are rare in human obesity.[94] As of July 2010[update], only 14 individuals from five families have been identified worldwide who carry a mutated ob gene (one of which was the first ever identified cause of genetic obesity in humans)—two families of Pakistani origin living in the UK, one family living in Turkey, one in Egypt, and one in Austria[95][96][97][98][99]—and two other families have been found that carry a mutated ob receptor.[100][101] Others have been identified as genetically partially deficient in leptin, and, in these individuals, leptin levels on the low end of the normal range can predict obesity.[102]
Several mutations of genes involving the melanocortins (used in brain signaling associated with appetite) and their receptors have also been identified as causing obesity in a larger portion of the population than leptin mutations.[103]
Physical properties
Adipose tissue has a density of ~0.9 g/ml.
Body fat meter
A body fat meter is a tool used to measure the body fat to weight ratio in the human body. Different meters use various methods to determine the ratio. They tend to under-read body fat percentage.
In contrast with clinical tools like
Animal studies
Within the fat (adipose) tissue of CCR2 deficient mice, there is an increased number of eosinophils, greater alternative Macrophage activation, and a propensity towards type 2 cytokine expression. Furthermore, this effect was exaggerated when the mice became obese from a high fat diet.[106]
Gallery
-
Diagrammatic sectional view of the skin (magnified)
-
White adipose tissue in paraffin section
-
Electronic instrument of body fat meter
See also
- Adipose differentiation-related protein
- Adipocytes
- Apelin
- Bioelectrical impedance analysis – a method to measure body fat percentage.
- Blubber – an extra thick form of adipose tissue found in some marine mammals.
- Body fat percentage
- Cellulite
- Lipolysis
- Lipodystrophy
- Human fat used as pharmaceutical in traditional medicine
- Obesity
- Starvation
- Steatosis (also called fatty change, fatty degeneration or adipose degeneration)
- Stem cells
- Subcutaneous fat
- Bariatrics
- Classification of obesity
- Classification of childhood obesity
- EPODE International Network, the world's largest obesity-prevention network
- World Fit A program of the United States Olympic Committee (USOC), and the United States Olympians and Paralympians Association (USOP)
- Obesity and walking
- Social stigma of obesity
References
- ^ PMID 23517218.
- ^ PMID 34100954.
- PMID 15181022.
- PMID 27529061.
- S2CID 205040511.
- PMID 9174472.
- ISBN 978-0-521-63577-6.
- S2CID 24434046.
- PMID 11282146.
- PMID 25385482.
- ^ Fat on the Inside: Looking Thin is Not Enough Archived 2016-11-17 at the Wayback Machine, By Fiona Haynes, About.com
- ^ a b "Abdominal fat and what to do about it". President & Fellows of Harvard College. September 2005.
Visceral fat more of a health concern than subcutaneous fat
- PMID 20150289.
- PMID 10866038.
- S2CID 24306481.
- S2CID 30693649.
- PMID 12503980.
- PMID 26559286.
- PMID 27614626.
- PMID 27427402.
- ^ "Reduce Abdominal Fat". Archived from the original on 2011-09-28. Retrieved 2009-04-10.
Estrogen causes fat to be stored around the pelvic region, hips, butt and thighs (pelvic region)
- ^ "Waistline Worries: Turning Apples Back Into Pears". healthywomen.org. Archived from the original on 2009-06-09.
- ^ Researchers think that the lack of estrogen at menopause plays a role in driving our fat northward. See: Andrews M (2006-12-01). "A Matter of Fat". Yahoo Health. Women's Health. Archived from the original on 2007-03-15.
- ISBN 978-0-19-935827-4. Retrieved August 3, 2021.
- PMID 17637702.
- S2CID 234747534.
- S2CID 206228752.
- PMID 18845966.
- PMID 19196080.
- PMID 7942575.
- PMID 14581396.
- PMID 30344493.
- PMID 29391548.
- PMID 19422139.
- ^ ISBN 978-0-8053-0094-9.
- PMID 19244087.
- ^ "Belly fat in women: Taking – and keeping – it off". MayoClinic.com. 2013-06-08. Retrieved 2013-12-02.
- S2CID 21052919.
- ^ PMID 9583375.
- PMID 20229598.
- PMID 26696121.
- PMID 19318450.
- ^ PMID 28436105.
- PMID 26052898.
- PMID 24709686.
- ^ PMID 22675355.
- S2CID 10876113.
- PMID 26694194.
- PMID 13920823.
- PMID 23579596.
- PMID 19427436.
- Time. Archivedfrom the original on December 20, 2013. Retrieved 2013-12-19.
- PMID 20133714.
- ^ S2CID 33960130.
- PMID 22574183.
- PMID 24361924.
- PMID 31077538.
- PMID 22108547.
- S2CID 902537.
- ^ S2CID 14289041.
- ^ "Eavesdropping on Communication Between Fat and Brain". August 31, 2022.
- PMID 25713540.
- PMID 23063128.
- PMID 20006514.
- PMID 33042008.
- S2CID 230947.
- PMID 19357407.
- S2CID 477352.
- PMID 19357406.
- ^ PMID 24100998.
- PMID 20160646.
- PMID 25645913.
- S2CID 2842953.
- PMID 23895241.
- PMID 24703692.
- PMID 25197048.
- PMID 27368105.
- PMID 28428261.
- PMID 22237023.
- PMID 24605108.
- S2CID 35008082.
- PMID 25133427.
- PMID 25533952.
- ^ PMID 24549398.
- PMID 27251423.
- ^ PMID 26844207.
- ISBN 978-1-58829-721-1.
- PMID 2699601.
- ISBN 978-1-4223-5243-4.
- ISBN 978-1-4223-5243-4.
- PMID 7624776.
- ISBN 978-0-323-22154-2.[page needed]
- PMID 19724019.
- PMID 9061713.
- S2CID 205032762.
- S2CID 36920931.
- PMID 15472169.
- PMID 19427251.
- PMID 20382689.
- S2CID 4400661.
- PMID 10395972.
- S2CID 4344492.
- S2CID 13946212.
- S2CID 46736884.(registration required)
- PMID 11320099.
- PMID 25934927. Archived from the originalon 2017-05-09. Retrieved 2016-09-08.
Further reading
- Stock MJ, Cinti S (2003). "Adipose Tissue / Structure and Function of Brown Adipose Tissue". Encyclopedia of Food Sciences and Nutrition. pp. 29–34. ISBN 978-0-12-227055-0.
- Vernon RG, Flint DJ (2003). "Adipose Tissue / Structure and Function of White Adipose Tissue". Encyclopedia of Food Sciences and Nutrition. pp. 23–29. ISBN 978-0-12-227055-0.
External links
- Adipose tissue at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Adipose tissue photomicrographs



