Adrenoleukodystrophy
| Adrenoleukodystrophy | |
|---|---|
| Other names | X-linked adrenoleukodystrophy, ALD, X-ALD, Siemerling–Creutzfeldt disease, bronze Schilder disease |
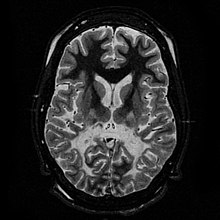 | |
| White matter, with reduced volume and increased signal intensity. The anterior white matter is spared. Features are consistent with X-linked adrenoleukodystrophy. | |
| Pronunciation | |
| Specialty | Medical genetics |
| Types | X-Linked ALD |
Adrenoleukodystrophy (ALD) is a
and hyperactivity. Other symptoms include problems in speaking, listening, and understanding verbal instructions.Clinically, ALD presents as a heterogeneous disorder, showing several distinct
ALD is caused by mutations in
Signs and symptoms
ALD can present in different ways. The different presentations are complicated by the pattern of
Male adrenoleukodystrophy phenotypes
| Phenotype | Description | Onset | Approximate relative frequency |
|---|---|---|---|
| Childhood cerebral | Progressive neurodegenerative decline, leading to a vegetative state without treatment | 3–10 years | 31–35% |
| Adolescent | Similar to childhood cerebral, with a slower progression | 11–21 years | 4–7% |
| Adrenomyeloneuropathy (AMN) | Progressive paraparesis ; approximately 40% progress to cerebral involvement
|
21–37 years | 40–46% |
| Adult cerebral | Dementia, behavioral disturbances, similar progression to childhood cerebral form, but without preceding AMN phenotype | Adulthood | 2–5% |
| Olivo-ponto-cerebellar | Cerebral and brain stem involvement
|
Adolescence to adulthood | 1–2% |
| " Addison disease only"
|
Adrenal insufficiency | Before 7.5 years | Up to 50% in childhood, varies with age |
| Asymptomatic | No clinical presentation, further studies can reveal subclinical adrenal insufficiency or mild AMN phenotype | Most common phenotype in boys under four years of age | Proportion of asymptomatic patients decreases with age |
Female adrenoleukodystrophy phenotypes
| Phenotype | Description | Onset | Approximate relative frequency |
|---|---|---|---|
| Asymptomatic | No neurologic or adrenal involvement | Most women under 30 do not have any neurologic involvement | Diminishes with age |
| Mild myelopathy | Increased deep tendon reflexes, sensory changes in lower extremities | Adulthood | Approximately 50% of women over 40 years of age |
| Moderate to severe myeloneuropathy | Similar to male AMN phenotype, but later onset and milder presentation | Adulthood | Approximately 15% of women over 40 years of age |
| Cerebral involvement | Progressive dementia and decline | Rare in childhood, more common in adults | ~2% |
| Adrenal involvement | Primary adrenal insufficiency | Any age | ~1% |
Genetics

ALD is caused by mutations in ABCD1, located at
Males with an ABCD1 mutation are
Pathogenesis
The exact cause for the varied collection of symptoms found in the different ALD phenotypes is not clear. The
Diagnosis
The clinical presentation of ALD can vary greatly, making diagnosis difficult. With the variety of phenotypes, clinical suspicion of ALD can result from a variety of different presentations. Symptoms vary based on the disease phenotype, and even within families or between twins.
Because the characteristic elevations associated with ALD are present at birth, well before any symptoms are apparent, there have been methods developed
The Loes score is a rating of the severity of abnormalities in the brain found on MRI. It ranges from 0 to 34, based on a point system derived from the location and extent of disease and the presence of atrophy in the brain, either localized to specific points or generally throughout the brain. A Loes score of 0.5 or less is classified as normal, while a Loes score of 14 or greater is considered severe. It was developed by neuroradiologist Daniel J. Loes MD and is an important tool in assessing disease progression and the effectiveness of therapy.[12]
Treatments
Dietary therapy
Initial attempts at dietary therapy in ALD involved restricting the intake of very-long chain fatty acids (VLCFA). Dietary intake is not the only source for VLCFA in the body, as they are also synthesized endogenously. This dietary restriction did not impact the levels of VLCFA in plasma and other body tissues.
Adrenomix®: In 2009, a second-generation mixture was created, adding to the glycerol trioleate (GTO) and trierucate glycerol (GTE), conjugated linoleic acid (CLA) a group of linoleic acid isomers capable of overcoming the blood-brain barrier. CLA, through the activation of peroxisome beta oxidation, increases the catabolism of pro-inflammatory molecules and ROS, acting as an anti-inflammatory and antioxidant. The use of CLA was initially considered in relation to the ability to inhibit fatty acid synthase together with a hypolipidic diet. A group of Italian researchers of the Bambino Gesù Pediatric Hospital in Rome showed that the administration of Adrenomix (GTO, GTE and CLA), in addition to decreasing levels of VLCFA throughout the body, reduces neuro inflammation and improves somatosensory evoked potential, found unchanged or worsened with only administration of GTO and GTE.[15]
Aldixyl®: In 2016, based on studies developed in recent years in the field of adrenoleukodystrophy and adrenomyelouropathy, a mixture was developed that adds to GTO, GTE and CLA, a mixture of powerful antioxidants at high dosages containing alpha lipoic acid (ALA), reduced L- glutathione and Vitamin E (α- tocopherol). Researchers at the IDIBELL- Hospital Duran i Reynals in Barcelona have shown that the early administration of a cocktail of powerful antioxidants, able to overcome the blood-brain barrier and thus carry out its activity at the CNS level, prevents the oxidative stress typical of the disease, intervenes on the initial axonal dysfunctions and therefore on locomotor damage.[16] This new mixture, unlike what happened with the administration of GTO and GTE alone, poorly accumulated at the level of the nervous system, enhances the anti-inflammatory activity and reduces the levels of VLCFA in the CNS by combining synergistically the activity of its components. In particular, CLA, in addition to overcoming the blood-brain barrier and regulating at the CNS level the metabolism of VLCFA is able to influence the catabolism of pro-inflammatory eicosanoids and lipid peroxidation products.[17][18][19][20][21] In this sense, the anti-inflammatory activity of ALA, reduced L-glutathione and Vitamin E is enhanced at the level of the whole body, and not only at the peripheral level as was the case in the past.[22]
Transplant
While dietary therapy has been shown to be effective to normalize the very-long chain fatty acid concentrations in the plasma of individuals with ALD,
Gene therapy
For patients where an appropriate match for a transplant cannot be found, there have been investigations into the use of
Elivaldogene autotemcel is pending authorization by the European Commission as of May 2021[update]. While this treatment is effective with 90% of patients being free of major functional disabilities after treatment, it costs $3.0 million per treatment and it comes with several adverse effects including mucositis and alopecia. [24][25][26][27]
Adrenal insufficiency
Treatment of the adrenal insufficiency that can accompany any of the common male phenotypes of ALD does not resolve any of the neurological symptoms. Hormone replacement is standard for ALD patients demonstrating adrenal insufficiency.[28] Adrenal insufficiency does not resolve with successful transplant; most patients still require hormone replacement.[23]
Epidemiology
ALD has not been shown to have an increased incidence in any specific country or ethnic group. In the United States, the incidence of affected males is estimated at 1:21,000. Overall incidence of
Asymptomology
There are documented asymptomatic males who present no ALD symptoms well into their 60s and 70s. It's not understood how they can have an ABCD1 gene variant and possess elevated VLCFAs and not exhibit either Cerebral ALD, Adrenal Insufficiency, or Adrenomyeloneuropathy symptoms. Daughters of asymptomatic males become obligate carriers, who may themselves be asymptomatic and who can pass the variant onto their children, which then silently perpetuates ALD. Sons of asymptomatic males only receive their father's Y chromosome and therefore can't inherit ALD.[29]
References
- ^ ISBN 978-0-07-136320-4.
- ^ PMID 16949688.
- ^ PMID 20301491. National Library of Medicine Bookshelf ID NBK1315.
- ^ a b "#300100 - Adrenoleukodystrophy". Johns Hopkins University. Archived from the original on 2015-09-24. Retrieved 2012-06-27.
- PMID 23835273.
- S2CID 38326524.
- ^ S2CID 24182017.
- ^ S2CID 24516921.
- PMID 22197596.
- PMID 19423374.
- ^ S2CID 21323198.
- from the original on September 1, 2015. Retrieved January 17, 2013.
- S2CID 21333247.
- (PDF) from the original on 13 April 2021. Retrieved 4 January 2020.
- PMID 22189598.
- PMID 21786300.
- PMID 2324649.
- PMID 8155738.
- PMID 12054595.
- S2CID 20240456.
- PMID 19403295.
- PMID 9236627.
- ^ PMID 22388279.
- ^ Liu, Angus (September 19, 2022). "A $3M gene therapy: Bluebird bio breaks its own pricing record with FDA approval of Skysona". Fierce Pharma. Retrieved November 1, 2023.
- ^ "Skysona: Pending EC decision". European Medicines Agency (EMA). 21 May 2021. Archived from the original on 2 June 2021. Retrieved 1 June 2021.
- ^ "Bluebird Bio Receives Positive CHMP Opinion for Skysona (elivaldogene autotemcel, Lenti-D) Gene Therapy for Patients Less Than 18 Years of Age with Early Cerebral Adrenoleukodystrophy (CALD)" (Press release). Bluebird Bio. 21 May 2021. Archived from the original on 2 June 2021. Retrieved 1 June 2021 – via Business Wire.
- ^ "Elivaldogene autotemcel". SPS - Specialist Pharmacy Service. 28 May 2021. Archived from the original on 2 June 2021. Retrieved 1 June 2021.
- S2CID 39152217.
- ^ "Asymptomatic adrenoleukodystrophy in elderly males | Request PDF". Archived from the original on 2021-04-18. Retrieved 2021-02-05.
