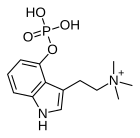
Aeruginascin
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Aeruginascin or N,N,N-trimethyl-4-phosphoryloxytryptamine is an
analogue of psilocybin. It is closely related to the frog skin toxin bufotenidine (5-HTQ), a potent 5-HT3 receptor agonist, but the aeruginascin metabolite 4-HO-TMT shows strong binding at the 5-HT2 receptors similar to psilocin.[8][9] The first scientific literature about the pharmacological effects of aeruginascin is from a study published by Gartz in 1989.[10] Across 23 analyzed cases of accidental hallucinogenic mushroom poisonings, people who had ingested the mushroom Inocybe aeruginascens reported only euphoric experiences.[11]
This is in contrast to the slight and in some cases extremely dysphoric experiences reported from the accidental ingestion of non aeruginascin containing mushrooms (containing solely psilocybin and psilocin).
References
- ^ Gartz J (1995). "Inocybe aeruginascens Babos". Eleusis, Journal of Psychoactive Plants & Compounds. 3. Museo Civico di Rovereto: 31–4.
- S2CID 260281286. Archived from the original(PDF) on 2011-05-24.
- S2CID 211214973.
- PMID 22957740.
- PMID 30875889.
- ^ PMID 36430546.
- ^ "CaaMTech Publishes Fundamental Research on Aeruginascin Derivatives". 14 September 2022.
- PMID 32685863.
- ^ Bauer BE (2020-07-07). "Study Finds Aeruginascin Metabolite 4-HO-TMT is Active at the Serotonin 5-HT2A Receptor". Psychedelic Science Review. Archived from the original on 2020-08-05. Retrieved 2021-09-07.
- ISSN 0167-7314.
- ^ "Aeruginascin". Psychedelic Science Review. 2018-11-19. Retrieved 2021-09-07.

