Alaproclate
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Alaproclate (developmental code name GEA-654) is a
liver complications in rodent studies. In addition to its SSRI properties, alaproclate has been found to act as a non-competitive NMDA receptor antagonist, but does not have discriminative stimulus properties similar to phencyclidine.[1][2]
Synthesis
Method for Treatment of Senile Dementia:[3]
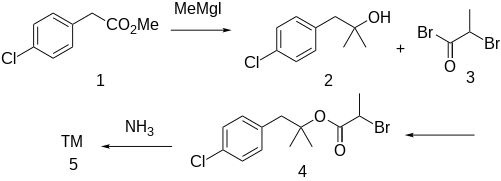
The
methylmagnesium iodide gives 1-(4-chlorophenyl)-2-methyl-2-propanol [5468-97-3] (2). Acylation with 2-bromopropionyl bromide [563-76-8] (3) gives [1-(4-Chlorophenyl)-2-methylpropan-2-yl] 2-bromopropanoate, CID:13695101
(3). Displacement of halogen with ammonia leads to alaproclate (4).
See also
References
- PMID 7996440.
- S2CID 30803162.
- ^ Ulf H. A. Lindberg, Sven-Ove gren, U.S. patent 4,469,707 (1984 to Astra Lakemedel Aktiebolag).
- ^ Lindberg, Ulf Henrik (1978). "Inhibitors of neuronal monoamine uptake. 2. Selective inhibition of 5-hydroxytryptamine uptake by .alpha.-amino acid esters of phenethyl alcohols". Journal of Medicinal Chemistry 21 (5): 448–456. doi:10.1021/jm00203a008.
- ^ Ulf M. A. Lindberg, Svante B. Ross, Seth-Olov Thorberg, Sven O. / gren, U.S. patent 4,237,311 & U.S. patent 4,331,684 (1980 & 1982 both to Astra Lakemedel Aktiebolag).
- ^ Gawell, Lars (1986). "Synthesis of [14C]alaproclate". Journal of Labelled Compounds and Radiopharmaceuticals. 23 (9): 947–949. doi:10.1002/jlcr.2580230905.
- ^ Bengtsson, Stefan; Gawell, Lars; Högberg, Thomas; Sahlberg, Christer (1985). "Synthesis and 3H NMR of 3H-alaproclate of high specific activity". Journal of Labelled Compounds and Radiopharmaceuticals. 22 (5): 427–435. doi:10.1002/jlcr.2580220503.
