Alcohol oxidation
Alcohol oxidation is a collection of oxidation reactions in organic chemistry that convert alcohols to aldehydes, ketones, carboxylic acids, and esters where the carbon carries a higher oxidation state. The reaction mainly applies to primary and secondary alcohols. Secondary alcohols form ketones, while primary alcohols form aldehydes or carboxylic acids.[1]
The reaction can occur using a variety of oxidants.
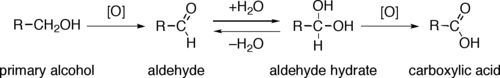
In principle, a simple way to oxidize an alcohols uses an oxygen atom and produce water: In practice, oxygen atoms are unavailable, so the above equation is only conceptual. Instead, most oxidations use oxides-based reagents, such as
Through a variety of mechanisms, the removal of a hydride equivalent converts a primary or secondary alcohol to an aldehyde or ketone, respectively. The oxidation of primary alcohols to carboxylic acids normally proceeds via the corresponding aldehyde, which is transformed via an aldehyde hydrate (gem-diol, R-CH(OH)2) by reaction with water. Thus, the oxidation of a primary alcohol at the aldehyde level without further oxidation to the carboxylic acid is possible by performing the reaction in absence of water, so that no aldehyde hydrate can be formed.
Ketone and aldehyde formation

Many reagents oxidize secondary alcohols to ketones and primary alcohols to aldehydes.
Chromium(VI) reagents

Chromium(VI) reagents are commonly used for these oxidations. One family of Cr(VI) reagents employs the complex CrO3(pyridine)2.[2]
- Sarett's reagent: a solution of CrO3(pyridine)2 in pyridine. It was popularized for selective oxidation of primary and secondary alcohols to carbonyl compounds.
- Collins reagent is a solution of the same CrO3(pyridine)2 but in dichloromethane. The Ratcliffe variant of Collins reagent relates to details of the preparation of this solution, i.e., the addition of chromium trioxide to a solution of pyridine in methylene chloride.[3]
A second family of Cr(VI) reagents are salts, featuring the pyridinium cation (C5H5NH+).
- pyridinium dichromate(PDC) is the pyridium salt of dichromate, [Cr2O7]2-.
- pyridinium chlorochromate (PCC) is the pyridinium salt of [CrO3Cl]−.
These salts are less reactive, more easily handled, and more selective than Collins reagent in oxidations of alcohols.
The above reagents represent improvements over the older
The Dess–Martin periodinane is a mild oxidant for the conversion of alcohols to aldehydes or ketones.[4] The reaction is performed under standard conditions, at room temperature, most often in dichloromethane. The reaction takes between half an hour and two hours to complete. The product is then separated from the spent periodinane.[5] Many iodosyl-based oxidants have been developed, e.g. IBX.
Swern oxidation
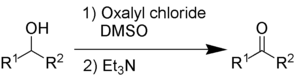
The by-products are dimethyl sulfide (Me2S), carbon monoxide (CO), carbon dioxide (CO2) and – when triethylamine is used as base – triethylammonium chloride (C6H15NHCl).
Oppenauer oxidation
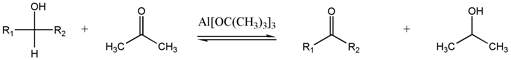
This seldom-used method interconverts alcohols and carbonyls.
Niche methods
Fétizon oxidation, also a seldom-used method, uses silver carbonate supported on Celite. This reagent operates through single electron oxidation by the silver cations.
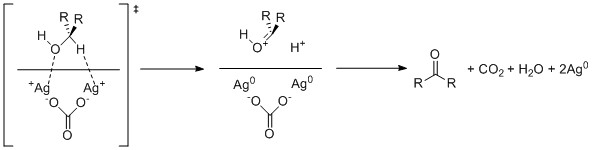
Another method is the oxoammonium-catalyzed oxidation.
Additionally, sodium hypochlorite (or household bleach) in acetone has been reported for efficient conversion of secondary alcohols in the presence of primary alcohols (Stevens oxidation).[6]
soluble transition metal complexes catalyze the oxidation of alcohols by presence of dioxygen or another terminal oxidant.[7]
Oxidation of diols

Compounds possessing two
Oxidation of primary alcohols to carboxylic acids

When a primary alcohol is converted to a carboxylic acid, the terminal carbon atom increases its oxidation state by four. The direct oxidation of primary alcohols to carboxylic acids can be carried out using a variety of reagents.
Aldehydes are readily oxidized using Cr(VI) and Mn(VII) reagents to carboxylic acids.
Potassium permanganate

Jones oxidation
The so-called

Two-step oxidation of alcohols to acids via isolated aldehydes
As a lot of the aforementioned conditions for the oxidations of primary alcohols to acids are harsh and not compatible with common protection groups, organic chemists often use a two-step procedure for the oxidation to acids. The alcohol is oxidized to an aldehyde using one of the many procedures above. This sequence is often used in natural product synthesis as in their synthesis of platencin.[15]
Niche methods and reagents
Ruthenium tetroxide is an aggressive, seldom-used agent that allows mild reaction conditions.
Heyns oxidation.[16]
The use of chlorites as terminal oxidants in conjunction with both hypochlorites and TEMPO gives carboxylic acids without chlorination side products.[17] The reaction is usually carried out in two steps in the same pot: partial oxidation is effected with TEMPO and hypochlorite, then chlorite is added to complete the oxidation. Only primary alcohol oxidation is observed. In conjunction with Sharpless dihydroxylation, this method can be used to generate enantiopure α-hydroxy acids.[18]
The Pinnick oxidation uses sodium chlorite.[19]
References
- ISBN 0-435-63120-9
- ISBN 0-387-23607-4.
- .
- J. Am. Chem. Soc.1991, 113, 7277–87.
- ^ J. S. Yadav, et al. "Recyclable 2nd generation ionic liquids as green solvents for the oxidation of alcohols with hypervalent iodine reagents", Tetrahedron, 2004, 60, 2131–35
- .
- ISSN 1463-9270.
- PMID 20192259.
- C. R. Acad. Sci.: 331.
- Bull. Soc. Chim. Fr.: 920.
- .
- ISBN 0-387-23607-4.
- .
- PMID 16608311.
- .
- ISBN 0-387-35431-X.
- ^ Song, Z. J.; Zhao, M.; Desmond, R.; Devine, P.; Tschaen, D. M.; Tillyer, R.; Frey, L.; Heid, R.; Xu, F.; Foster, B.; Li, J.; Reamer, R.; Volante, R.; Grabowski, E. J. J.; Dolling, U. H.; Reider, P. J.; Okada, S.; Kato, Y.; Mano, E. J. Org. Chem. 1999, 64, 9658.
- ^ Sharpless, K. B.; Amberg, W.; Bennani, Y. L.; Crispino, G. A.; Hartung, J.; Jeong, K. S.; Kwong, H. L.; Morikawa, K.; Wang, Z. M.; Xu, D.; Zhang, X. L. J. Org. Chem. 1992, 57, 2768.
- .
