Alkaloid

Alkaloids are a class of basic, naturally occurring organic compounds that contain at least one nitrogen atom. This group also includes some related compounds with neutral[2] and even weakly acidic properties.[3] Some synthetic compounds of similar structure may also be termed alkaloids.[4] In addition to carbon, hydrogen and nitrogen, alkaloids may also contain oxygen or sulfur. More rarely still, they may contain elements such as phosphorus, chlorine, and bromine.[5]
Alkaloids are produced by a large variety of organisms including
The boundary between alkaloids and other nitrogen-containing natural compounds is not clear-cut.
Naming

The name "alkaloids" (German: Alkaloide) was introduced in 1819 by German chemist Carl Friedrich Wilhelm Meissner, and is derived from late Latin root alkali and the Greek-language suffix -οειδής -('like').[nb 1] However, the term came into wide use only after the publication of a review article, by Oscar Jacobsen in the chemical dictionary of Albert Ladenburg in the 1880s.[22][23]
There is no unique method for naming alkaloids.
History

Alkaloid-containing plants have been used by humans since ancient times for therapeutic and recreational purposes. For example, medicinal plants have been known in
Extracts from plants containing toxic alkaloids, such as
Studies of alkaloids began in the 19th century. In 1804, the German chemist
A significant contribution to the chemistry of alkaloids in the early years of its development was made by the French researchers
The first complete synthesis of an alkaloid was achieved in 1886 by the German chemist

Classifications

Compared with most other classes of natural compounds, alkaloids are characterized by a great structural diversity. There is no uniform classification.[38] Initially, when knowledge of chemical structures was lacking, botanical classification of the source plants was relied on. This classification is now considered obsolete.[5][39]
More recent classifications are based on similarity of the carbon skeleton (e.g., indole-, isoquinoline-, and pyridine-like) or biochemical precursor (ornithine, lysine, tyrosine, tryptophan, etc.).[5] However, they require compromises in borderline cases;[38] for example, nicotine contains a pyridine fragment from nicotinamide and a pyrrolidine part from ornithine[40] and therefore can be assigned to both classes.[41]
Alkaloids are often divided into the following major groups:[42]
- "True alkaloids" contain heterocycle and originate from amino acids.[43] Their characteristic examples are atropine, nicotine, and morphine. This group also includes some alkaloids that besides the nitrogen heterocycle contain terpene (e.g., evonine[44]) or peptide fragments (e.g. ergotamine[45]). The piperidine alkaloids coniine and coniceine may be regarded as true alkaloids (rather than pseudoalkaloids: see below)[46] although they do not originate from amino acids.[47]
- "Protoalkaloids", which contain nitrogen (but not the nitrogen heterocycle) and also originate from amino acids.[43] Examples include mescaline, adrenaline and ephedrine.
- Polyamine alkaloids – derivatives of putrescine, spermidine, and spermine.
- Peptide and cyclopeptide alkaloids.[48]
- Pseudoalkaloids – alkaloid-like compounds that do not originate from amino acids.[49] This group includes terpene-like and steroid-like alkaloids,[50] as well as purine-like alkaloids such as caffeine, theobromine, theacrine and theophylline.[51] Some authors classify ephedrine and cathinone as pseudoalkaloids. Those originate from the amino acid phenylalanine, but acquire their nitrogen atom not from the amino acid but through transamination.[51][52]
Some alkaloids do not have the carbon skeleton characteristic of their group. So,
Main classes of monomeric alkaloids are listed in the table below:
| Class | Major groups | Main synthesis steps | Examples |
|---|---|---|---|
| Alkaloids with nitrogen heterocycles (true alkaloids) | |||
Pyrrolidine derivatives[54]
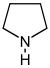 |
Ornithine or arginine → putrescine → N-methylputrescine → N-methyl-Δ1-pyrroline [55] | Cuscohygrine, hygrine, hygroline, stachydrine[54][56] | |
Tropane derivatives[57]
 |
Atropine group Substitution in positions 3, 6 or 7 |
Ornithine or arginine → putrescine → N-methylputrescine → N-methyl-Δ1-pyrroline [55] | Atropine, scopolamine, hyoscyamine[54][57][58] |
| Cocaine group Substitution in positions 2 and 3 |
Cocaine, ecgonine[57][59] | ||
Pyrrolizidine derivatives[60]
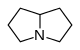 |
Non-esters | In plants: ornithine or arginine → putrescine → homospermidine → retronecine[55] | Retronecine, heliotridine, laburnine [60][61] |
| Complex esters of monocarboxylic acids
|
Indicine, lindelophin, sarracine [60] | ||
| Macrocyclic diesters | Platyphylline, trichodesmine[60] | ||
| 1-aminopyrrolizidines ( lolines )
|
In fungi: L-proline + L-homoserine → N-(3-amino-3-carboxypropyl)proline → norloline[62][63] | Loline, N-formylloline, N-acetylloline[64] | |
Piperidine derivatives[65]
 |
Lysine → cadaverine → Δ1-piperideine [66] | Sedamine, lobeline, anaferine, piperine[46][67] | |
| Octanoic acid → coniceine → coniine[47] | Coniine, coniceine [47] | ||
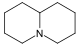
| Quinolizidine derivatives[68][69]
|
Lupinine group | Lysine → cadaverine → Δ1-piperideine [70] | Lupinine, nupharidin [68] |
| Cytisine group | Cytisine[68] | ||
| Sparteine group | Sparteine, lupanine, anahygrine[68] | ||
| Matrine group. | Matrine, oxymatrine, allomatridine[68][71][72] | ||
| Ormosanine group | Ormosanine, piptantine[68][73] | ||
Indolizidine derivatives[74]
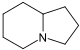 |
α-aminoadipic acid → pipecolic acid → 1 indolizidinone [75]
|
Swainsonine, castanospermine[76] | |
Pyridine derivatives[77][78]
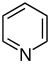 |
Simple derivatives of pyridine | Nicotinic acid → dihydronicotinic acid → 1,2-dihydropyridine [79]
|
Trigonelline, ricinine, arecoline[77][80] |
| Polycyclic noncondensing pyridine derivatives | Nicotine, nornicotine, anabasine, anatabine [77][80] | ||
| Polycyclic condensed pyridine derivatives | Actinidine, gentianine, pediculinine [81] | ||
| Sesquiterpene pyridine derivatives | Nicotinic acid, isoleucine[21]
|
Evonine, hippocrateine, triptonine [78][79] | |
Isoquinoline derivatives and related alkaloids [82]
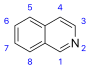 |
Simple derivatives of isoquinoline [83] | Tyrosine or phenylalanine → dopamine or tyramine (for alkaloids Amarillis) [84][85] | Salsoline, lophocerine [82][83] |
| Derivatives of 1- and 3-isoquinolines [86] | N-methylcoridaldine, noroxyhydrastinine [86] | ||
| Derivatives of 1- and 4-phenyltetrahydroisoquinolines [83] | Cryptostilin [83][87] | ||
| Derivatives of 5-naftil-isoquinoline [88] | Ancistrocladine [88] | ||
| Derivatives of 1- and 2-benzyl-izoquinolines [89] | Papaverine, laudanosine, sendaverine | ||
| Cularine group[90] | Cularine, yagonine [90] | ||
| Pavines and isopavines [91] | Argemonine, amurensine[91] | ||
| Benzopyrrocolines [92] | Cryptaustoline [83] | ||
| Protoberberines [83] | Berberine, canadine, ophiocarpine, mecambridine, corydaline [93] | ||
| Phthalidisoquinolines [83] | narcotine (Noscapine) [94]
| ||
| Spirobenzylisoquinolines [83] | Fumaricine [91] | ||
Ipecacuanha alkaloids[95]
|
Emetine, protoemetine, ipecoside [95] | ||
| Benzophenanthridines [83] | Sanguinarine, oxynitidine, corynoloxine [96] | ||
| Aporphines [83] | Glaucine, coridine, liriodenine [97] | ||
| Proaporphines [83] | Pronuciferine, glaziovine [83][92] | ||
| Homoaporphines [98] | Kreysiginine, multifloramine [98] | ||
| Homoproaporphines [98] | Bulbocodine [90] | ||
| Morphines[99] | Morphine, codeine, thebaine, sinomenine[100] | ||
| Homomorphines [101] | Kreysiginine, androcymbine [99] | ||
| Tropoloisoquinolines [83] | Imerubrine [83] | ||
| Azofluoranthenes [83] | Rufescine, imeluteine [102] | ||
| Amaryllis alkaloids[103] | Lycorine, ambelline, tazettine, galantamine, montanine [104] | ||
| Erythrina alkaloids[87] | Erysodine, erythroidine [87] | ||
| Phenanthrene derivatives [83] | Atherosperminine [83][93] | ||
| Protopines [83] | Protopine, oxomuramine, corycavidine [96] | ||
| Aristolactam [83] | Doriflavin [83] | ||
Oxazole derivatives[105]
 |
Tyrosine → tyramine[106] | Annuloline, halfordinol, texaline, texamine[107] | |
Isoxazole derivatives
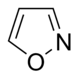 |
Ibotenic acid → Muscimol | Ibotenic acid, Muscimol | |
Thiazole derivatives[108]
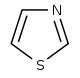 |
1-Deoxy-D-xylulose 5-phosphate (DOXP), tyrosine, cysteine[109] | Nostocyclamide, thiostreptone [108][110] | |
Quinazoline derivatives[111]
 |
3,4-Dihydro-4-quinazolone derivatives | Anthranilic acid or phenylalanine or ornithine[112] | Febrifugine[113] |
| 1,4-Dihydro-4-quinazolone derivatives | Glycorine, arborine, glycosminine[113] | ||
| Pyrrolidine and piperidine quinazoline derivatives | Vazicine (peganine) [105] | ||
| Acridine derivatives[105]
|
Anthranilic acid[114] | Rutacridone, acronicine[115][116] | |
Quinoline derivatives[117][118]
 |
Simple derivatives of quinoline derivatives of 4-quinolone
|
Anthranilic acid → 3-carboxyquinoline [119] | Cusparine, echinopsine, evocarpine[118][120][121] |
| Tricyclic terpenoids | Flindersine[118][122] | ||
| Furanoquinoline derivatives | Dictamnine, fagarine, skimmianine[118][123][124] | ||
| Quinines | Tryptophan → tryptamine → strictosidine (with secologanin) → korinanteal → cinhoninon[85][119] | Quinine, quinidine, cinchonine, cinhonidine [122] | |
Indole derivatives[100]
 |
Non-isoprene indole alkaloids | ||
| Simple indole derivatives [125] | Tryptophan → tryptamine or 5-Hydroxytryptophan[126] | ||
| Simple derivatives of β-carboline[129]
|
Harman, harmine, harmaline, eleagnine [125] | ||
| Pyrroloindole alkaloids [130] | Physostigmine (eserine), etheramine, physovenine, eptastigmine[130] | ||
| Semiterpenoid indole alkaloids | |||
| Ergot alkaloids[100] | Tryptophan → chanoclavine → agroclavine → elimoclavine → paspalic acid → lysergic acid[130] | Ergotamine, ergobasine, ergosine[131] | |
| Monoterpenoid indole alkaloids | |||
| Corynanthe type alkaloids[126] | Tryptophan → tryptamine → strictosidine (with secologanin) [126] | Ajmalicine, sarpagine, vobasine, ajmaline, yohimbine, reserpine, mitragynine,[132][133] group strychnine and (Strychnine brucine, aquamicine, vomicine[134]) | |
Iboga-type alkaloids[126]
|
Ibogamine, ibogaine, voacangine[126] | ||
| Aspidosperma-type alkaloids[126] | Vincamine, vinca alkaloids,[27][135] vincotine, aspidospermine[136][137] | ||
Imidazole derivatives[105]
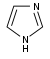 |
Directly from histidine[138] | Histamine, pilocarpine, pilosine, stevensine[105][138] | |
Purine derivatives[139]
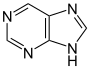 |
Caffeine, theobromine, theophylline, saxitoxin[140][141] | ||
| Alkaloids with nitrogen in the side chain (protoalkaloids) | |||
| β- Phenylethylamine derivatives[92]
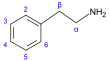 |
Tyrosine or phenylalanine → dioxyphenilalanine → dopamine → adrenaline and mescaline tyrosine → tyramine phenylalanine → 1-phenylpropane-1,2-dione → cathinone → ephedrine and pseudoephedrine[21][52][142] | ||
Colchicine alkaloids [144]
 |
Tyrosine or phenylalanine → dopamine → autumnaline → colchicine[145] | Colchicine, colchamine[144] | |
Muscarine[146]
 |
Glutamic acid → 3-ketoglutamic acid → muscarine (with pyruvic acid)[147] | Muscarine, allomuscarine, epimuscarine, epiallomuscarine[146] | |
Benzylamine[148]
 |
Phenylalanine with valine, leucine or isoleucine[149] | Capsaicin, dihydrocapsaicin, nordihydrocapsaicin, vanillylamine[148][150] | |
| Polyamines alkaloids | |||
| Putrescine derivatives[151]
|
ornithine → putrescine → spermidine → spermine[152] | Paucine [151] | |
| Spermidine derivatives[151]
|
Lunarine, codonocarpine[151] | ||
| Spermine derivatives[151]
|
Verbascenine, aphelandrine [151] | ||
| Peptide (cyclopeptide) alkaloids | |||
| Peptide alkaloids with a 13-membered cycle [48][153] | Nummularine C type | From different amino acids [48] | Nummularine C, Nummularine S [48] |
| Ziziphine type | Ziziphine A, sativanine H [48] | ||
| Peptide alkaloids with a 14-membered cycle [48][153] | Frangulanine type | Frangulanine, scutianine J [153] | |
| Scutianine A type | Scutianine A [48] | ||
| Integerrine type | Integerrine, discarine D [153] | ||
| Amphibine F type | Amphibine F, spinanine A [48] | ||
| Amfibine B type | Amphibine B, lotusine C [48] | ||
| Peptide alkaloids with a 15-membered cycle [153] | Mucronine A type | Mucronine A [45][153] | |
| Pseudoalkaloids ( steroids )
| |||
Diterpenes [45]
 |
Lycoctonine type | Mevalonic acid → Isopentenyl pyrophosphate → geranyl pyrophosphate[154][155] | Aconitine, delphinine[45][156] |
Steroidal alkaloids [157]
 |
Cholesterol, arginine[158] | Solanidine, cyclopamine, batrachotoxin[159] | |
Properties
Most alkaloids contain oxygen in their molecular structure; those compounds are usually colorless crystals at ambient conditions. Oxygen-free alkaloids, such as nicotine[160] or coniine,[36] are typically volatile, colorless, oily liquids.[161] Some alkaloids are colored, like berberine (yellow) and sanguinarine (orange).[161]
Most alkaloids are weak bases, but some, such as
Most alkaloids have a bitter taste or are poisonous when ingested. Alkaloid production in plants appeared to have evolved in response to feeding by herbivorous animals; however, some animals have evolved the ability to detoxify alkaloids.[169] Some alkaloids can produce developmental defects in the offspring of animals that consume but cannot detoxify the alkaloids. One example is the alkaloid cyclopamine, produced in the leaves of corn lily. During the 1950s, up to 25% of lambs born by sheep that had grazed on corn lily had serious facial deformations. These ranged from deformed jaws to cyclopia (see picture). After decades of research, in the 1980s, the compound responsible for these deformities was identified as the alkaloid 11-deoxyjervine, later renamed to cyclopamine.[170]
Distribution in nature
Alkaloids are generated by various living organisms, especially by higher plants – about 10 to 25% of those contain alkaloids.[171][172] Therefore, in the past the term "alkaloid" was associated with plants.[173]
The alkaloids content in plants is usually within a few percent and is inhomogeneous over the plant tissues. Depending on the type of plants, the maximum concentration is observed in the leaves (for example,
Beside plants, alkaloids are found in certain types of
Extraction

Because of the structural diversity of alkaloids, there is no single method of their extraction from natural raw materials.[179] Most methods exploit the property of most alkaloids to be soluble in organic solvents[7] but not in water, and the opposite tendency of their salts.
Most plants contain several alkaloids. Their mixture is extracted first and then individual alkaloids are separated.[180] Plants are thoroughly ground before extraction.[179][181] Most alkaloids are present in the raw plants in the form of salts of organic acids.[179] The extracted alkaloids may remain salts or change into bases.[180] Base extraction is achieved by processing the raw material with alkaline solutions and extracting the alkaloid bases with organic solvents, such as 1,2-dichloroethane, chloroform, diethyl ether or benzene. Then, the impurities are dissolved by weak acids; this converts alkaloid bases into salts that are washed away with water. If necessary, an aqueous solution of alkaloid salts is again made alkaline and treated with an organic solvent. The process is repeated until the desired purity is achieved.
In the acidic extraction, the raw plant material is processed by a weak acidic solution (e.g., acetic acid in water, ethanol, or methanol). A base is then added to convert alkaloids to basic forms that are extracted with organic solvent (if the extraction was performed with alcohol, it is removed first, and the remainder is dissolved in water). The solution is purified as described above.[179][182]
Alkaloids are separated from their mixture using their different solubility in certain solvents and different reactivity with certain reagents or by distillation.[183]
A number of alkaloids are identified from insects, among which the fire ant venom alkaloids known as solenopsins have received greater attention from researchers.[184] These insect alkaloids can be efficiently extracted by solvent immersion of live fire ants[7] or by centrifugation of live ants[185] followed by silica-gel chromatography purification.[186] Tracking and dosing the extracted solenopsin ant alkaloids has been described as possible based on their absorbance peak around 232 nanometers.[187]
Biosynthesis
Biological precursors of most alkaloids are
Synthesis of Schiff bases
Schiff bases can be obtained by reacting amines with ketones or aldehydes.[189] These reactions are a common method of producing C=N bonds.[190]
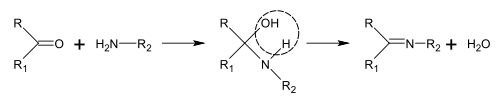
In the biosynthesis of alkaloids, such reactions may take place within a molecule,[188] such as in the synthesis of piperidine:[41]

Mannich reaction
An integral component of the Mannich reaction, in addition to an amine and a

The Mannich reaction can proceed both intermolecularly and intramolecularly:[191][192]

Dimer alkaloids
In addition to the described above monomeric alkaloids, there are also
- Mannich reaction, resulting in, e.g., voacamine
- Michael reaction(villalstonine)
- Condensation of aldehydes with amines (toxiferine)
- Oxidative addition of phenols (dauricine, tubocurarine)
- Lactonization (carpaine).
-
Tubocurarine
There are also dimeric alkaloids formed from two distinct monomers, such as the vinca alkaloids

Biological role
Alkaloids are among the most important and best-known
Most of the known functions of alkaloids are related to protection. For example, aporphine alkaloid liriodenine produced by the tulip tree protects it from parasitic mushrooms. In addition, the presence of alkaloids in the plant prevents insects and chordate animals from eating it. However, some animals are adapted to alkaloids and even use them in their own metabolism.[201] Such alkaloid-related substances as serotonin, dopamine and histamine are important neurotransmitters in animals. Alkaloids are also known to regulate plant growth.[202] One example of an organism that uses alkaloids for protection is the Utetheisa ornatrix, more commonly known as the ornate moth. Pyrrolizidine alkaloids render these larvae and adult moths unpalatable to many of their natural enemies like coccinelid beetles, green lacewings, insectivorous hemiptera and insectivorous bats.[203] Another example of alkaloids being utilized occurs in the poison hemlock moth (Agonopterix alstroemeriana). This moth feeds on its highly toxic and alkaloid-rich host plant poison hemlock (Conium maculatum) during its larval stage. A. alstroemeriana may benefit twofold from the toxicity of the naturally-occurring alkaloids, both through the unpalatability of the species to predators and through the ability of A. alstroemeriana to recognize Conium maculatum as the correct location for oviposition.[204] A fire ant venom alkaloid known as solenopsin has been demonstrated to protect queens of invasive fire ants during the foundation of new nests, thus playing a central role in the spread of this pest ant species around the world.[205]
Applications
In medicine
Medical use of alkaloid-containing plants has a long history, and, thus, when the first alkaloids were isolated in the 19th century, they immediately found application in clinical practice.[206] Many alkaloids are still used in medicine, usually in the form of salts widely used including the following:[17][207]
| Alkaloid | Action |
|---|---|
| Ajmaline | Antiarrhythmic |
| Emetine | emesis
|
| Ergot alkaloids | Vasoconstriction, hallucinogenic, Uterotonic |
| Glaucine | Antitussive
|
| Morphine | Analgesic |
| Nicotine | Stimulant, nicotinic acetylcholine receptor agonist |
| Physostigmine | Inhibitor of acetylcholinesterase |
| Quinidine | Antiarrhythmic |
| Quinine | Antipyretic, antimalarial |
| Reserpine | Antihypertensive |
Tubocurarine
|
Muscle relaxant |
| Vinblastine, vincristine | Antitumor |
| Vincamine | Vasodilating, antihypertensive |
| Yohimbine | Stimulant, aphrodisiac |
| Berberine | Antihyperglycaemic[13]
|
Many synthetic and semisynthetic drugs are structural modifications of the alkaloids, which were designed to enhance or change the primary effect of the drug and reduce unwanted side-effects.[208] For example, naloxone, an opioid receptor antagonist, is a derivative of thebaine that is present in opium.[209]
In agriculture
Prior to the development of a wide range of relatively low-toxic synthetic pesticides, some alkaloids, such as salts of nicotine and anabasine, were used as insecticides. Their use was limited by their high toxicity to humans.[210]
Use as psychoactive drugs
Preparations of plants and fungi containing alkaloids and their extracts, and later pure alkaloids, have long been used as
There are alkaloids that do not have strong psychoactive effect themselves, but are precursors for semi-synthetic psychoactive drugs. For example, ephedrine and pseudoephedrine are used to produce methcathinone and methamphetamine.[216] Thebaine is used in the synthesis of many painkillers such as oxycodone.
See also
Explanatory notes
- ^ Meissner, W. (1819). "Über Pflanzenalkalien: II. Über ein neues Pflanzenalkali (Alkaloid)" [About Plant Alkalis: II. About a New Plant Alkali (Alkaloid)]. Journal für Chemie und Physik. 25: 379–381. Archived from the original on 18 May 2023.
In the penultimate sentence of his article, Meissner wrote: "Überhaupt scheint es mir auch angemessen, die bis jetzt bekannten Pflanzenstoffe nicht mit dem Namen Alkalien, sondern Alkaloide zu belegen, da sie doch in manchen Eigenschaften von den Alkalien sehr abweichen, sie würden daher in dem Abschnitt der Pflanzenchemie vor den Pflanzensäuren ihre Stelle finden." ["In general, it seems appropriate to me to impose on the currently known plant substances not the name 'alkalis' but 'alkaloids', since they differ greatly in some properties from the alkalis; among the chapters of plant chemistry, they would therefore find their place before plant acids (since 'Alkaloid' would precede 'Säure' (acid) but follow 'Alkalien')".]
Citations
- OCLC 1056390214.
- ^
- OCLC 1090491824.
- OCLC 1026521889.
- ^ a b c d "АЛКАЛОИДЫ - Химическая энциклопедия" [Alkaloids - Chemical Encyclopedia]. www.xumuk.ru (in Russian). Retrieved 18 May 2023.
- OCLC 851770197.
- ^ PMID 23333648.
- PMID 24359196.
- PMID 23410167.
- ISBN 978-1139491983.
- S2CID 205171789.
- PMID 35673446.
- ^ PMID 36144587.
- ^ "Alkaloid". 18 December 2007.
- ISBN 978-0683085006.
- ISBN 978-0-12-597180-5.
- ^ ISBN 0-12-227411-3
- ISBN 978-1-4200-0447-2.
- ISBN 978-0-7637-0432-2.
- ISBN 978-81-224-1459-2.
- ^ a b c d Aniszewski, p. 110
- ^ Hesse, pp. 1–3
- ^ Ladenburg, Albert (1882). Handwörterbuch der chemie (in German). E. Trewendt. pp. 213–422.
- ^ a b Hesse, p. 5
- ^ The suffix "ine" is a Greek feminine patronymic suffix and means "daughter of"; hence, for example, "atropine" means "daughter of Atropa" (belladonna): "Development of Systematic Names for the Simple Alkanes". yale.edu. Archived from the original on 16 March 2012.
- ^ Hesse, p. 7
- ^ PMID 15032608.
- ISBN 9781498704304.
- ISBN 9783527326693.
- ^ a b Aniszewski, p. 182
- ^ Hesse, p. 338
- ^ Hesse, p. 304
- ^ Hesse, p. 350
- ^ Hesse, pp. 313–316
- ^ Begley, Natural Products in Plants.
- ^ a b Кониин in the Great Soviet Encyclopedia, 1969–1978 (in Russian)
- ^ Hesse, p. 204
- ^ a b Hesse, p. 11
- ^ Orekhov, p. 6
- ^ Aniszewski, p. 109
- ^ a b Dewick, p. 307
- ^ Hesse, p. 12
- ^ a b Plemenkov, p. 223
- ^ Aniszewski, p. 108
- ^ a b c d Hesse, p. 84
- ^ a b Hesse, p. 31
- ^ a b c Dewick, p. 381
- ^ PMID 9121730.
- ^ Aniszewski, p. 11
- ^ Plemenkov, p. 246
- ^ a b Aniszewski, p. 12
- ^ a b Dewick, p. 382
- ^ Hesse, pp. 44, 53
- ^ a b c Plemenkov, p. 224
- ^ a b c Aniszewski, p. 75
- ^ Orekhov, p. 33
- ^ a b c "Chemical Encyclopedia: Tropan alkaloids". xumuk.ru.
- ^ Hesse, p. 34
- ^ Aniszewski, p. 27
- ^ a b c d "Chemical Encyclopedia: Pyrrolizidine alkaloids". xumuk.ru.
- ^ Plemenkov, p. 229
- S2CID 13461396.
- S2CID 34409048.
- PMID 17346759.
- ^ Plemenkov, p. 225
- ^ Aniszewski, p. 95
- ^ Orekhov, p. 80
- ^ a b c d e f "Chemical Encyclopedia: Quinolizidine alkaloids". xumuk.ru.
- ^ Saxton, Vol. 1, p. 93
- ^ Aniszewski, p. 98
- ^ Saxton, Vol. 1, p. 91
- PMID 14620842.
- ^ Saxton, Vol. 1, p. 92
- ^ Dewick, p. 310
- ^ Aniszewski, p. 96
- ^ Aniszewski, p. 97
- ^ a b c Plemenkov, p. 227
- ^ a b "Chemical Encyclopedia: pyridine alkaloids". xumuk.ru.
- ^ a b Aniszewski, p. 107
- ^ a b Aniszewski, p. 85
- ^ Plemenkov, p. 228
- ^ a b Hesse, p. 36
- ^ a b c d e f g h i j k l m n o p q r s t "Chemical Encyclopedia: isoquinoline alkaloids". xumuk.ru.
- ^ Aniszewski, pp. 77–78
- ^ a b c d Begley, Alkaloid Biosynthesis
- ^ a b Saxton, Vol. 3, p. 122
- ^ a b c Hesse, p. 54
- ^ a b Hesse, p. 37
- ^ Hesse, p. 38
- ^ a b c Hesse, p. 46
- ^ a b c Hesse, p. 50
- ^ (PDF) from the original on 9 October 2022.
- ^ a b Hesse, p. 47
- ^ Hesse, p. 39
- ^ a b Hesse, p. 41
- ^ a b Hesse, p. 49
- ^ Hesse, p. 44
- ^ a b c Saxton, Vol. 3, p. 164
- ^ a b Hesse, p. 51
- ^ a b c Plemenkov, p. 236
- ^ Saxton, Vol. 3, p. 163
- ^ Saxton, Vol. 3, p. 168
- ^ Hesse, p. 52
- ^ Hesse, p. 53
- ^ a b c d e Plemenkov, p. 241
- ^ Brossi, Vol. 35, p. 261
- ^ Brossi, Vol. 35, pp. 260–263
- ^ a b Plemenkov, p. 242
- ^ Begley, Cofactor Biosynthesis
- PMID 10714899.
- ^ "Chemical Encyclopedia: Quinazoline alkaloids". xumuk.ru.
- ^ Aniszewski, p. 106
- ^ a b Aniszewski, p. 105
- doi:10.1039/a705734b.
- ^ Plemenkov, pp. 231, 246
- ^ Hesse, p. 58
- ^ Plemenkov, p. 231
- ^ a b c d "Chemical Encyclopedia: Quinoline alkaloids". xumuk.ru.
- ^ a b Aniszewski, p. 114
- ^ Orekhov, p. 205
- ^ Hesse, p. 55
- ^ a b Plemenkov, p. 232
- ^ Orekhov, p. 212
- ^ Aniszewski, p. 118
- ^ a b Aniszewski, p. 112
- ^ a b c d e f Aniszewski, p. 113
- ^ Hesse, p. 15
- ^ Saxton, Vol. 1, p. 467
- ^ Dewick, pp. 349–350
- ^ a b c Aniszewski, p. 119
- ^ Hesse, p. 29
- ^ Hesse, pp. 23–26
- ^ Saxton, Vol. 1, p. 169
- ^ Saxton, Vol. 5, p. 210
- ^ PMID 22609781.
- ^ Hesse, pp. 17–18
- ^ Dewick, p. 357
- ^ a b Aniszewski, p. 104
- ^ Hesse, p. 72
- ^ Hesse, p. 73
- ^ Dewick, p. 396
- ^ "PlantCyc Pathway: ephedrine biosynthesis". Archived from the original on 10 December 2011.
- ^ Hesse, p. 76
- ^ a b "Chemical Encyclopedia: colchicine alkaloids". xumuk.ru.
- ^ Aniszewski, p. 77
- ^ a b Hesse, p. 81
- ^ Brossi, Vol. 23, p. 376
- ^ a b Hesse, p. 77
- ^ Brossi, Vol. 23, p. 268
- ^ Brossi, Vol. 23, p. 231
- ^ a b c d e f Hesse, p. 82
- ^ "Spermine Biosynthesis". www.qmul.ac.uk. Archived from the original on 13 November 2003.
- ^ a b c d e f Plemenkov, p. 243
- ^ "Chemical Encyclopedia: Terpenes". xumuk.ru.
- ^ Begley, Natural Products: An Overview
- PMID 9149410.
- ^ Hesse, p. 88
- ^ Dewick, p. 388
- ^ Plemenkov, p. 247
- ^ Никотин in the Great Soviet Encyclopedia, 1969–1978 (in Russian)
- ^ a b c Grinkevich, p. 131
- ISBN 978-1-4200-5013-4.
- ^ "Caffeine". DrugBank. Retrieved 12 February 2013.
- ^ "Cocaine". DrugBank. Retrieved 12 February 2013.
- ^ "Codeine". DrugBank. Retrieved 12 February 2013.
- ^ "Nicotine". DrugBank. Retrieved 12 February 2013.
- ^ "Morphine". DrugBank. Retrieved 12 February 2013.
- ^ "Yohimbine". DrugBank. Archived from the original on 30 January 2013. Retrieved 12 February 2013.
- ^ Fattorusso, p. 53
- ISBN 978-0-85199-614-1.
- ^ Aniszewski, p. 13
- ^ Orekhov, p. 11
- ^ Hesse, p.4
- ^ Grinkevich, pp. 122–123
- ^ Orekhov, p. 12
- PMID 26805882.
- ^ Fattorusso, p. XVII
- ^ Aniszewski, pp. 110–111
- ^ a b c d Hesse, p. 116
- ^ a b Grinkevich, p. 132
- ^ Grinkevich, p. 5
- ^ Grinkevich, pp. 132–134
- ^ Grinkevich, pp. 134–136
- ISBN 978-94-007-6415-6.
- PMID 29510162.
- PMID 19326861.
- PMID 29900266.
- ^ a b c Plemenkov, p. 253
- ^ Plemenkov, p. 254
- ^ a b Dewick, p. 19
- ^ Plemenkov, p. 255
- ^ Dewick, p. 305
- ^ Hesse, pp. 91–105
- ISBN 9783540563914.
- ^ ISBN 9783540728795.
- PMID 21695100.
- PMID 19072542.
- PMID 11922805.
- hdl:10481/47295.
- ^ Aniszewski, p. 142
- ^ Hesse, pp. 283–291
- ^ Aniszewski, pp. 142–143
- ISBN 0195327373.
- S2CID 45478867– via ResearchGate.
- S2CID 54481057.
- ^ Hesse, p. 303
- ^ Hesse, pp. 303–309
- ^ Hesse, p. 309
- ^ Dewick, p. 335
- ISBN 978-0-08-087491-3.
- ^ Veselovskaya, p. 75
- ^ Hesse, p. 79
- ^ Veselovskaya, p. 136
- ISBN 978-0-12-469556-6.
- ^ Veselovskaya, p. 6
- ^ Veselovskaya, pp. 51–52
General and cited references
- Aniszewski, Tadeusz (2007). Alkaloids: secrets of life. Amsterdam: ISBN 978-0-444-52736-3.
- Begley, Tadhg P. (2009). Encyclopedia of Chemical Biology. Vol. 10. Wiley. pp. 1569–1570. ISBN 978-0-471-75477-0.
- Brossi, Arnold (1989). The Alkaloids: Chemistry and Pharmacology. Academic Press.
- Dewick, Paul M. (2002). Medicinal Natural Products: A Biosynthetic Approach (Second ed.). Wiley. ISBN 978-0-471-49640-3.
- Fattorusso, E.; Taglialatela-Scafati, O. (2008). Modern Alkaloids: Structure, Isolation, Synthesis and Biology. Wiley-VCH. ISBN 978-3-527-31521-5.
- Grinkevich NI; Safronich LN, eds. (1983). The chemical analysis of medicinal plants (in Russian). Moscow: Vysshaya Shkola.
- Hesse, Manfred (2002). Alkaloids: Nature's Curse or Blessing?. Wiley-VCH. ISBN 978-3-906390-24-6.
- Knunyants, IL (1988). Chemical Encyclopedia. Soviet Encyclopedia.
- Orekhov, AP (1955). Chemistry alkaloids (Acad. 2nd ed.). Moscow.
{{cite book}}: CS1 maint: location missing publisher (link) - Plemenkov, VV (2001). Introduction to the Chemistry of Natural Compounds. Kazan.
{{cite book}}: CS1 maint: location missing publisher (link) - Saxton, J. E. (1971). The Alkaloids: A Specialist Periodical Report. London: The Chemical Society.
- Veselovskaya, N. B.; Kovalenko, A. E. (2000). Drugs. Moscow: Triada-X.
- Wink, M (2009). "Mode of action and toxicology of plant toxins and poisonous plants". Mitt. Julius Kühn-Inst. 421: 93–112x.