Alkyl group


In organic chemistry, an alkyl group is an alkane missing one hydrogen.[1] The term alkyl is intentionally unspecific to include many possible substitutions. An acyclic alkyl has the general formula of −CnH2n+1. A cycloalkyl group is derived from a cycloalkane by removal of a hydrogen atom from a ring and has the general formula −CnH2n−1.[2] Typically an alkyl is a part of a larger molecule. In
Related concepts
Alkylation is the addition of alkyl groups to molecules, often by alkylating agents such as alkyl halides.
Alkylating antineoplastic agents are a class of compounds that are used to treat cancer. In such case, the term alkyl is used loosely. For example, nitrogen mustards are well-known alkylating agents, but they are not simple hydrocarbons.
In chemistry, alkyl is a group, a substituent, that is attached to other molecular fragments. For example,
In medicinal chemistry
In
Alkyl cations, anions, and radicals
Usually, alkyl groups are attached to other atoms or groups of atoms. Free alkyls occur as neutral radicals, as anions, or as cations. The cations are called
Nomenclature
Alkyl groups form
The naming convention is taken from IUPAC nomenclature:
| Number of carbon atoms | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Prefix | meth- | eth- | prop- | but- | pent- | hex- | hept- | oct- | non- | dec- | undec- | dodec- | tridec- | tetradec- |
| Group name | Methyl | Ethyl | Propyl | Butyl | Pentyl | Hexyl | Heptyl | Octyl | Nonyl | Decyl | Undecyl | Dodecyl | Tridecyl | Tetradecyl |
The prefixes taken from IUPAC nomenclature are used to name branched chained structures by their

The structure of
If there is more than one of the same alkyl group attached to a chain, then the prefixes are used on the alkyl groups to indicate multiples (i.e., di, tri, tetra, etc.)
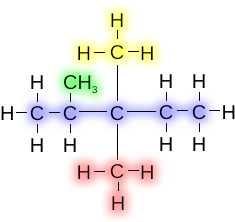
This compound is known as
In addition, each position on an alkyl chain can be described according to how many other carbon atoms are attached to it. The terms primary, secondary, tertiary, and quaternary refer to a carbon attached to one, two, three, or four other carbons respectively.
| Cn | Trivial name | Symbol | Condensed formula | IUPAC status | Preferred IUPAC name | Skeletal formula |
|---|---|---|---|---|---|---|
| С1 | methyl | Ме | −CH3 | methyl | ||
| С2 | ethyl | Et | −CH2−CH3 | ethyl | ||
| С3 | propyl , n-propyl
|
Pr, nPr, n-Pr | −CH2−CH2−CH3 | propyl | ||
| isopropyl | iPr, i-Pr,iPr | −CH(−CH3)2 | 2-propyl | |||
| С4 | n-butyl | Bu, n-Bu, nBu | −CH2−CH2−CH2−CH3 | butyl | ||
| isobutyl | iBu, i-Bu, iBu | −CH2−CH(−CH3)2 | 2-methylpropyl | |||
| sec-butyl | sBu, s-Bu, sBu | −CH(−CH3)−CH2−CH3 | 2-butyl | 
| ||
| tert-butyl | tBu, t-Bu, tBu | −C(−CH3)3 | tert-butyl | 
| ||
| С5 | n-pentyl, amyl | Pe, Am, nPe, n-Pe, nPe, nAm | −CH2−CH2−CH2−CH2−CH3 | pentyl | ||
| tert-pentyl | tPe, t-Pe, tPe | −C(−CH3)2−CH2−CH3 | No longer recommended | 2-methylbutan-2-yl (aka 1,1-dimethylpropyl) | ||
| neopentyl | −CH2−C(−CH3)3 | No longer recommended | 2,2-dimethylpropyl | |||
| isopentyl, isoamyl | −CH2−CH2−CH(−CH3)2 | No longer recommended | 3-methylbutyl | |||
| sec-pentyl | sPe, s-Pe, sPe | −CH(−CH3)−CH2−CH2−CH3 | pentan-2-yl(or (1-Methylbutyl)) | |||
| 3-pentyl | −CH(−CH2−CH3)2 | pentan-3-yl (also known as (1-Ethylpropyl)) | ||||
| sec-isopentyl, sec-isoamyl, siamyl | Sia | −CH(−CH3)−CH(−CH3)2 | 3-methylbutan-2-yl (or (1,2-Dimethylpropyl)) | |||
| active pentyl | −CH2−CH(−CH3)−CH2−CH3 | 2-methylbutyl |
Etymology
The first named alkyl radical was ethyl, named so by
See also
References
- ^ Virtual Textbook of Organic Chemistry Naming Organic Compounds Archived 2016-05-21 at the Portuguese Web Archive
- S2CID 5159000.
- ISSN 0001-4842.
- PMID 32786419.
- ISBN 978-3-642-27415-2.
Ethyl radicals (named by Liebig in 1833 from the Greek and German word "aether" plus Greek "hyle"
- ISBN 978-3-642-27415-2.
The methyl radical ... named from Greek roots, by Dumas and Peligot in 1834: methyl = methy + hyle ("spirit" + "wood")
- ISBN 978-3-642-27415-2.
Amyl radicals ("amilène" was coined by Auguste Cahours in 1840, to designate a substance from potato starch after fermentation and distillation
- ISBN 978-3-642-27415-2.
"Alkyl" was coined without fanfare by Johannes Wislicenus, professor at Würzburg; an early use (perhaps not the first) is in his 1882 article [22, 244]. The word was derived from the first three letters of "Alkoholradicale" combined with the suffix -yl; it was (and is) a generic term for any of those radicals who bear the "first names" methyl, ethyl, propyl, butyl, amyl, etc.
- .
