Alkyne

In
Structure and bonding
In acetylene, the H–C≡C
- 2-butyne, e, the naturally occurring 1-phenylhepta-1,3,5-triyne, and f, the strained cycloheptyne. Triple bonds are highlighted blue.
The
Terminal and internal alkynes
Internal alkynes feature carbon substituents on each acetylenic carbon. Symmetrical examples include
Terminal alkynes have the formula RC2H. An example is
Naming alkynes
In systematic chemical nomenclature, alkynes are named with the Greek prefix system without any additional letters. Examples include ethyne or octyne. In parent chains with four or more carbons, it is necessary to say where the triple bond is located. For octyne, one can either write 3-octyne or oct-3-yne when the bond starts at the third carbon. The lowest number possible is given to the triple bond. When no superior functional groups are present, the parent chain must include the triple bond even if it is not the longest possible carbon chain in the molecule. Ethyne is commonly called by its trivial name acetylene.
In chemistry, the suffix -yne is used to denote the presence of a triple bond. In organic chemistry, the suffix often follows IUPAC nomenclature. However, inorganic compounds featuring unsaturation in the form of triple bonds may be denoted by substitutive nomenclature with the same methods used with alkynes (i.e. the name of the corresponding saturated compound is modified by replacing the "-ane" ending with "-yne"). "-diyne" is used when there are two triple bonds, and so on. The position of unsaturation is indicated by a numerical locant immediately preceding the "-yne" suffix, or 'locants' in the case of multiple triple bonds. Locants are chosen so that the numbers are low as possible. "-yne" is also used as a suffix to name substituent groups that are triply bound to the parent compound.
Sometimes a number between hyphens is inserted before it to state which atoms the triple bond is between. This suffix arose as a collapsed form of the end of the word "acetylene". The final "-e" disappears if it is followed by another suffix that starts with a vowel.[6]
Structural isomerism
Alkynes having four or more carbon atoms can form different structural isomers by having the triple bond in different positions or having some of the carbon atoms be substituents rather than part of the parent chain. Other non-alkyne structural isomers are also possible.
- C2H2: acetylene only
- C3H4: propyne only
- C4H6: 2 isomers: 2-butyne
- C5H8: 3 isomers: 2-pentyne, and 3-methyl-1-butyne
- C6H10: 7 isomers:
Synthesis
Cracking
Commercially, the dominant alkyne is acetylene itself, which is used as a fuel and a precursor to other compounds, e.g., acrylates. Hundreds of millions of kilograms are produced annually by partial oxidation of natural gas:[7]
Propyne, also industrially useful, is also prepared by
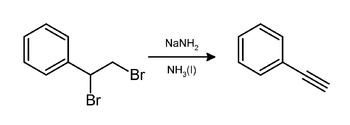
Alkynes are prepared from 1,1- and 1,2-
Via the Fritsch–Buttenberg–Wiechell rearrangement, alkynes are prepared from vinyl bromides. Alkynes can be prepared from aldehydes using the Corey–Fuchs reaction and from aldehydes or ketones by the Seyferth–Gilbert homologation.
Vinyl chlorides are susceptible to dehydrochlorination. Vinyl chlorides are available from aldehydes using the reagent (chloromethylene)triphenylphosphorane.
Reactions, including applications
Featuring a reactive functional group, alkynes participate in many organic reactions. Such use was pioneered by Ralph Raphael, who in 1955 wrote the first book describing their versatility as intermediates in synthesis.[10]
Hydrogenation
Being more unsaturated than alkenes, alkynes characteristically undergo reactions that show that they are "doubly unsaturated". Alkynes are capable of adding two equivalents of H2, whereas an alkene adds only one equivalent.[11] Depending on catalysts and conditions, alkynes add one or two equivalents of hydrogen. Partial hydrogenation, stopping after the addition of only one equivalent to give the alkene, is usually more desirable since alkanes are less useful:

The largest scale application of this technology is the conversion of acetylene to ethylene in refineries (the steam cracking of alkanes yields a few percent acetylene, which is selectively hydrogenated in the presence of a palladium/silver catalyst). For more complex alkynes, the Lindlar catalyst is widely recommended to avoid formation of the alkane, for example in the conversion of phenylacetylene to styrene.[12] Similarly, halogenation of alkynes gives the alkene dihalides or alkyl tetrahalides:
The addition of one equivalent of H2 to internal alkynes gives cis-alkenes.
Alkynes characteristically are capable of adding two equivalents of halogens and hydrogen halides.
The addition of nonpolar E−H bonds across C≡C is general for silanes, boranes, and related hydrides. The
Addition of hydrogen halides has long been of interest. In the presence of
Hydration
The
Hydration of phenylacetylene gives acetophenone:[14]
(Ph3P)AuCH3 catalyzes hydration of 1,8-nonadiyne to 2,8-nonanedione:[15]
Tautomerism
Terminal alkyl alkynes exhibit tautomerism. Propyne exists in equilibrium with propadiene:
Cycloadditions and oxidation
Alkynes undergo diverse
Oxidative cleavage of alkynes proceeds via cycloaddition to metal oxides. Most famously, potassium permanganate converts alkynes to a pair of carboxylic acids.
Reactions specific for terminal alkynes
Terminal alkynes are readily converted to many derivatives, e.g. by coupling reactions and condensations. Via the condensation with formaldehyde and acetylene is produced butynediol:[7][16]
In the
This reactivity exploits the fact that terminal alkynes are weak acids, whose typical pKa values around 25 place them between that of ammonia (35) and ethanol (16):
where MX = NaNH2, LiBu, or RMgX.
The reactions of alkynes with certain metal cations, e.g. Ag+ and Cu+ also gives acetylides. Thus, few drops of
In the
Metal complexes
Alkynes form complexes with transition metals. Such complexes occur also in metal catalyzed reactions of alkynes such as
Alkynes in nature and medicine
According to
Alkynes occur in some pharmaceuticals, including the contraceptive noretynodrel. A carbon–carbon triple bond is also present in marketed drugs such as the antiretroviral Efavirenz and the antifungal Terbinafine. Molecules called ene-diynes feature a ring containing an alkene ("ene") between two alkyne groups ("diyne"). These compounds, e.g. calicheamicin, are some of the most aggressive antitumor drugs known, so much so that the ene-diyne subunit is sometimes referred to as a "warhead". Ene-diynes undergo rearrangement via the Bergman cyclization, generating highly reactive radical intermediates that attack DNA within the tumor.[21]
See also
References
- ^ Alkyne. Encyclopædia Britannica
- ISBN 9780470771563.
- ISBN 9780470084960.
- .
- ISBN 978-0-07-176797-2.
- ISBN 0-408-70144-7.
- ^ ISBN 978-3527306732.
- .
- .
- OCLC 3134811.
- ISBN 0003277402.
- ^ H. Lindlar; R. Dubuis (1973). "Palladium catalyst for partial reduction of acetylenes". Organic Syntheses; Collected Volumes, vol. 5, p. 880..
- ISBN 978-3527306732.
- .
- ^ Mizushima, E.; Cui, D.-M.; Nath, D. C. D.; Hayashi, T.; Tanaka, M. (2005). "Au(I)-Catalyzed hydratation of alkynes: 2,8-nonanedione". Organic Syntheses. 83: 55.
- ISBN 978-3527306732.
- .
- S2CID 95666091.
- PMID 16447152.
- ISBN 0-444-87115-2.
- PMID 1584797.














![{\displaystyle {\ce {2R-\!{\equiv }\!-H->[{\ce {Cu(OAc)2}}][{\ce {pyridine}}]R-\!{\equiv }\!-\!{\equiv }\!-R}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/8fe5690544a4da60b118164e70291e6eae02f82e)