Allotype (immunology)
The word allotype comes from two Greek roots, allo meaning 'other or differing from the norm' and typos meaning 'mark'.
Definition and organisation of allotypes in humans
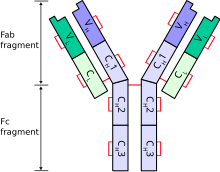
Human allotypes nomenclature was first described in alphabetical system and further systematized in numerical system, but both could be found in the literature.[7][8] For example, allotype expressed on constant region of heavy chain on IgG are designated by Gm which stands for ‘genetic marker ‘ together with IgG subclass (IgG1 àG1m, IgG2 àG2m) and the allotype number or letter [ G1m1/ G1m (a) ]. Polymorphisms within IgA are denoted in the same way as A2m (eg. A2m1/2) and kappa light chains constant region polymorphisms as Km (eg. Km1). Despite the fact, that there are multiple known lambda chain isotypes, there have not been reported any lambda chain serological polymorphisms.[9]
All these before mentioned allotypes are expressed on constant regions of the immunoglobulin. Genes responsible for encoding structure of constant regions of heavy chains are closely linked and therefore inherited together as one haplotype with low number of crossovers. Although some crossovers did occur during human evolution resulting in the creation of current populations characteristic haplotypes and importance of allotype system in population studies.[10],[11]
Implications for monoclonal antibody therapy
Antibody allotypes came back to spotlight due to development and use of therapies based on monoclonal antibodies. These recombinant human glycoproteins and proteins are now well established in clinical practise, but sometimes leads to adverse effects such as generation of antitherapeutic antibodies that negates therapy or even cause severe reactions to the therapy. This reaction may be attributed to differences between therapeutics itself or may arise between same therapeutics produced by different companies or even between different lots produced by the same company. To prevent production of such antitherapeutic antibodies, ideally, all clinical used proteins and glycoproteins should poses same allotype as natural patient’s product, this way the presence of ‘altered self‘ which poses a potential target for immune system, is limited. Whilst many parameters connected to developing and manufacturing process that might predispose monoclonal antibodies to cause immune response are well known and appropriate steps are taken to monitor and control these unwanted effects, complications linked with administration of monoclonal antibodies to genetically diverse human population are less well described. Humans exhibit abundance of genotypes and phenotypes, however all currently licensed IgG therapeutic immunoglobulins are developed as single allotypic/ polymorphic form. Patients that are homozygous for alternative phenotype are therefore at higher risk of developing potential immune response to the therapy.[4]
See also
References
- ^ "Pathology, Microbiology and Immunology". University of South Carolina School of Medicine. Retrieved 2020-09-06.
- ISBN 978-0-12-635501-7.
- PMID 2698222.
- ^ PMID 20073133.
- S2CID 42336232.
- PMID 25368619.
- .
- S2CID 32562449.
- PMID 2908491.
- ^ "IMGT Repertoire (IG and TR)". International ImMunoGeneTics Information System (IMGT). Archived from the original on 2021-11-01. Retrieved 2020-09-06.
- PMID 22665258.
External links
- Immunoglobulin+allotypes at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
