Alpha helix

An alpha helix (or α-helix) is a sequence of amino acids in a protein that are twisted into a coil (a helix).
The alpha helix is the most common structural arrangement in the secondary structure of proteins. It is also the most extreme type of local structure, and it is the local structure that is most easily predicted from a sequence of amino acids.
The alpha helix has a
Other names
The alpha helix is also commonly called a:
- Pauling–Corey–Branson α-helix (from the names of three scientists who described its structure)
- 3.613-helix because there are 3.6 amino acids in one ring, with 13 atoms being involved in the ring formed by the hydrogen bond (starting with amidic hydrogen and ending with carbonyl oxygen)


Discovery
In the early 1930s,
Astbury initially proposed a linked-chain structure for the fibers. He later joined other researchers (notably the American chemist Maurice Huggins) in proposing that:
- the unstretched protein molecules formed a helix (which he called the α-form)
- the stretching caused the helix to uncoil, forming an extended state (which he called the β-form).
Although incorrect in their details, Astbury's models of these forms were correct in essence and correspond to modern elements of
that somewhat resemble the modern α-helix.Two key developments in the modeling of the modern α-helix were: the correct bond geometry, thanks to the crystal structure determinations of amino acids and peptides and Pauling's prediction of planar peptide bonds; and his relinquishing of the assumption of an integral number of residues per turn of the helix. The pivotal moment came in the early spring of 1948, when Pauling caught a cold and went to bed. Being bored, he drew a polypeptide chain of roughly correct dimensions on a strip of paper and folded it into a helix, being careful to maintain the planar peptide bonds. After a few attempts, he produced a model with physically plausible hydrogen bonds. Pauling then worked with Corey and Branson to confirm his model before publication.[6] In 1954, Pauling was awarded his first Nobel Prize "for his research into the nature of the chemical bond and its application to the elucidation of the structure of complex substances"[7] (such as proteins), prominently including the structure of the α-helix.
Structure
Geometry and hydrogen bonding
The amino acids in an α-helix are arranged in a right-handed
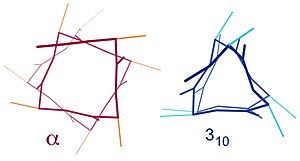
Similar structures include the 310 helix (i + 3 → i hydrogen bonding) and the π-helix (i + 5 → i hydrogen bonding). The α-helix can be described as a 3.613 helix, since the i + 4 spacing adds three more atoms to the H-bonded loop compared to the tighter 310 helix, and on average, 3.6 amino acids are involved in one ring of α-helix. The subscripts refer to the number of atoms (including the hydrogen) in the closed loop formed by the hydrogen bond.[12]

Residues in α-helices typically adopt backbone (φ, ψ)
- 3 cos Ω = 1 − 4 cos2 φ + ψ/2
The α-helix is tightly packed; there is almost no free space within the helix. The amino-acid side-chains are on the outside of the helix, and point roughly "downward" (i.e., toward the N-terminus), like the branches of an evergreen tree (Christmas tree effect). This directionality is sometimes used in preliminary, low-resolution electron-density maps to determine the direction of the protein backbone.[16]
Stability
Helices observed in proteins can range from four to over forty residues long, but a typical helix contains about ten amino acids (about three turns). In general, short

Visualization
The 3 most popular ways of visualizing the alpha-helical secondary structure of oligopeptide sequences are (1) a helical wheel,[21] (2) a wenxiang diagram,[22] and (3) a helical net.[23] Each of these can be visualized with various software packages and web servers. To generate a small number of diagrams, Heliquest[24] can be used for helical wheels, and NetWheels[25] can be used for helical wheels and helical nets. To programmatically generate a large number of diagrams, helixvis[26][27] can be used to draw helical wheels and wenxiang diagrams in the R and Python programming languages.
Experimental determination
Since the α-helix is defined by its hydrogen bonds and backbone conformation, the most detailed experimental evidence for α-helical structure comes from atomic-resolution
There are several lower-resolution methods for assigning general helical structure. The
Long homopolymers of amino acids often form helices if soluble. Such long, isolated helices can also be detected by other methods, such as
Amino-acid propensities
Different amino-acid sequences have different propensities for forming α-helical structure.
Table of standard amino acid alpha-helical propensities
Estimated differences in
Differences in free energy change per residue[29] Amino acid 3-
letter1-
letterHelical penalty kcal/mol kJ/molAlanine Ala A 0.00 0.00 Arginine Arg R 0.21 0.88 Asparagine Asn N 0.65 2.72 Aspartic acid Asp D 0.69 2.89 Cysteine Cys C 0.68 2.85 Glutamic acid Glu E 0.40 1.67 Glutamine Gln Q 0.39 1.63 Glycine Gly G 1.00 4.18 Histidine His H 0.61 2.55 Isoleucine Ile I 0.41 1.72 Leucine Leu L 0.21 0.88 Lysine Lys K 0.26 1.09 Methionine Met M 0.24 1.00 Phenylalanine Phe F 0.54 2.26 Proline Pro P 3.16 13.22 Serine Ser S 0.50 2.09 Threonine Thr T 0.66 2.76 Tryptophan Trp W 0.49 2.05 Tyrosine Tyr Y 0.53 2.22 Valine Val V 0.61 2.55
Dipole moment
A helix has an overall
Coiled coils
Coiled-coil α helices are highly stable forms in which two or more helices wrap around each other in a "supercoil" structure.
Facial arrangements
The amino acids that make up a particular helix can be plotted on a
Changes in binding orientation also occur for facially-organized oligopeptides. This pattern is especially common in antimicrobial peptides, and many models have been devised to describe how this relates to their function. Common to many of them is that the hydrophobic face of the antimicrobial peptide forms pores in the plasma membrane after associating with the fatty chains at the membrane core.[33][34]
Larger-scale assemblies

Myoglobin and hemoglobin, the first two proteins whose structures were solved by X-ray crystallography, have very similar folds made up of about 70% α-helix, with the rest being non-repetitive regions, or "loops" that connect the helices. In classifying proteins by their dominant fold, the Structural Classification of Proteins database maintains a large category specifically for all-α proteins.
Hemoglobin then has an even larger-scale
Functional roles

DNA binding
α-Helices have particular significance in
Membrane spanning
α-Helices are also the most common protein structure element that crosses biological membranes (
Mechanical properties
α-Helices under axial tensile deformation, a characteristic loading condition that appears in many alpha-helix-rich filaments and tissues, results in a characteristic three-phase behavior of stiff-soft-stiff tangent modulus.[38] Phase I corresponds to the small-deformation regime during which the helix is stretched homogeneously, followed by phase II, in which alpha-helical turns break mediated by the rupture of groups of H-bonds. Phase III is typically associated with large-deformation covalent bond stretching.
Dynamical features
Alpha-helices in proteins may have low-frequency accordion-like motion as observed by the Raman spectroscopy[39] and analyzed via the quasi-continuum model.[40][41] Helices not stabilized by tertiary interactions show dynamic behavior, which can be mainly attributed to helix fraying from the ends.[42]
Helix–coil transition
Homopolymers of amino acids (such as polylysine) can adopt α-helical structure at low temperature that is "melted out" at high temperatures. This helix–coil transition was once thought to be analogous to protein denaturation. The statistical mechanics of this transition can be modeled using an elegant transfer matrix method, characterized by two parameters: the propensity to initiate a helix and the propensity to extend a helix.
In art

At least five artists have made explicit reference to the α-helix in their work: Julie Newdoll in painting and Julian Voss-Andreae, Bathsheba Grossman, Byron Rubin, and Mike Tyka in sculpture.
San Francisco area artist Julie Newdoll,[43] who holds a degree in microbiology with a minor in art, has specialized in paintings inspired by microscopic images and molecules since 1990. Her painting "Rise of the Alpha Helix" (2003) features human figures arranged in an α helical arrangement. According to the artist, "the flowers reflect the various types of sidechains that each amino acid holds out to the world".[43] This same metaphor is also echoed from the scientist's side: "β sheets do not show a stiff repetitious regularity but flow in graceful, twisting curves, and even the α-helix is regular more in the manner of a flower stem, whose branching nodes show the influence of environment, developmental history, and the evolution of each part to match its own idiosyncratic function."[12]
Julian Voss-Andreae is a German-born sculptor with degrees in experimental physics and sculpture. Since 2001 Voss-Andreae creates "protein sculptures"[44] based on protein structure with the α-helix being one of his preferred objects. Voss-Andreae has made α-helix sculptures from diverse materials including bamboo and whole trees. A monument Voss-Andreae created in 2004 to celebrate the memory of Linus Pauling, the discoverer of the α-helix, is fashioned from a large steel beam rearranged in the structure of the α-helix. The 10-foot-tall (3 m), bright-red sculpture stands in front of Pauling's childhood home in Portland, Oregon.
Mike Tyka is a computational biochemist at the University of Washington working with David Baker. Tyka has been making sculptures of protein molecules since 2010 from copper and steel, including ubiquitin and a potassium channel tetramer.[47]
See also
- 310 helix
- Beta sheet
- Davydov soliton
- Folding (chemistry)
- Knobs into holes packing
- Pi helix
- Proteopedia Helices_in_Proteins
References
- S2CID 4167651.
- .
- JSTOR 985121.
- .
- S2CID 93804323.
- PMID 14816373.
- ^ "The Nobel Prize in Chemistry 1954".
- PMID 29712120.
- .
- ^ "Polypeptide Conformations 1 and 2". www.sbcs.qmul.ac.uk. Retrieved 5 November 2018.
- S2CID 29185760.
- ^ PMID 7020376.
- S2CID 8358424.
- ^ Dickerson RE, Geis I (1969), Structure and Action of Proteins, Harper, New York
- ISBN 9781439882047.
- PMID 20179338.
- .
- PMID 19334772.
- PMID 27935949.
- PMID 28706065.
- PMID 6048867.
- S2CID 26944184.
- PMID 5699810.
- PMID 18662927.
- S2CID 92137153.
- S2CID 56486576.
- ^ Subramanian V, Wadhwa RR, Stevens-Truss R (2020). "Helixvis: Visualize alpha-helical peptides in Python". ChemRxiv.
- PMID 9649402.
- PMID 9649402.
- S2CID 4147335.
- PMID 8251939.
- PMID 9385654.
- PMID 29401708.
- PMID 15880793.
- ^ Branden & Tooze, chapter 10
- ^ Branden & Tooze, chapter 12.
- PMID 25732028.
- PMID 17925444.
- PMID 7115900.
- PMID 6362659.
- PMID 6428481.
- PMID 19131517.
- ^ a b "Julie Newdoll Scientifically Inspired Art, Music, Board Games". www.brushwithscience.com. Retrieved 2016-04-06.
- S2CID 57558522.
- ^ Grossman, Bathsheba. "About the Artist". Bathsheba Sculpture. Retrieved 2016-04-06.
- ^ "About". molecularsculpture.com. Retrieved 2016-04-06.
- ^ Tyka, Mike. "About". www.miketyka.com. Retrieved 2016-04-06.
Further reading
- Tooze J, Brändén CI (1999). Introduction to protein structure. New York: Garland Pub. ISBN 0-8153-2304-2..
- Eisenberg D (September 2003). "The discovery of the alpha-helix and beta-sheet, the principal structural features of proteins". Proceedings of the National Academy of Sciences of the United States of America. 100 (20): 11207–10. PMID 12966187.
- Astbury WT, Woods HJ (1931). "The Molecular Weights of Proteins". Nature. 127 (3209): 663–665. S2CID 4133226.
- Astbury WT, Street A (1931). "X-ray studies of the structures of hair, wool and related fibres. I. General". Philosophical Transactions of the Royal Society of London, Series A. 230: 75–101. .
- Astbury WT (1933). "Some Problems in the X-ray Analysis of the Structure of Animal Hairs and Other Protein Fibers". Trans. Faraday Soc. 29 (140): 193–211. .
- Astbury WT, Woods HJ (1934). "X-ray studies of the structures of hair, wool and related fibres. II. The molecular structure and elastic properties of hair keratin". Philosophical Transactions of the Royal Society of London, Series A. 232 (707–720): 333–394. .
- Astbury WT, Sisson WA (1935). "X-ray studies of the structures of hair, wool and related fibres. III. The configuration of the keratin molecule and its orientation in the biological cell". Proceedings of the Royal Society of London, Series A. 150 (871): 533–551. .
- Sugeta H, Miyazawa T (1967). "General Method for Calculating Helical Parameters of Polymer Chains from Bond Lengths, Bond Angles, and Internal-Rotation Angles". Biopolymers. 5 (7): 673–679. S2CID 97785907.
- Wada A (1976). "The alpha-helix as an electric macro-dipole". Advances in Biophysics: 1–63. PMID 797240.
- Chothia C, Levitt M, Richardson D (October 1977). "Structure of proteins: packing of alpha-helices and pleated sheets". Proceedings of the National Academy of Sciences of the United States of America. 74 (10): 4130–4. PMID 270659.
- Chothia C, Levitt M, Richardson D (January 1981). "Helix to helix packing in proteins". Journal of Molecular Biology. 145 (1): 215–50. PMID 7265198.
- Hol WG (1985). "The role of the alpha-helix dipole in protein function and structure". Progress in Biophysics and Molecular Biology. 45 (3): 149–95. PMID 3892583.
- Barlow DJ, Thornton JM (June 1988). "Helix geometry in proteins". Journal of Molecular Biology. 201 (3): 601–19. PMID 3418712.
- Murzin AG, Finkelstein AV (December 1988). "General architecture of the alpha-helical globule". Journal of Molecular Biology. 204 (3): 749–69. PMID 3225849.

