Alpha particle
ħ[4] |
Alpha particles, also called alpha rays or alpha radiation, consist of two protons and two neutrons bound together into a particle identical to a helium-4 nucleus.[5] They are generally produced in the process of alpha decay but may also be produced in other ways. Alpha particles are named after the first letter in the Greek alphabet, α. The symbol for the alpha particle is α or α2+. Because they are identical to helium nuclei, they are also sometimes written as He2+
or 4
2He2+
indicating a helium ion with a +2 charge (missing its two electrons). Once the ion gains electrons from its environment, the alpha particle becomes a normal (electrically neutral) helium atom 4
2He.
Alpha particles have a net spin of zero. When produced in standard alpha
However, so-called
Name
The term "alpha particle" was coined by Ernest Rutherford in reporting his studies of the properties of Uranium radiation.[6] The radiation appeared to have two different characters, the first he called " radiation" and the more penetrating one he called " radiation". After five years of additional experimental work, Rutherford and Hans Geiger determined that the alpha particle, after it has lost its positive charge, is a Helium atom.[7][8][9]: 61
Alpha radiation consists of particles equivalent to doubly-ionized helium nuclei (He2+
) which can gain electrons from passing through matter. This mechanism is the origin of terrestrial Helium gas.[10]
Sources
Alpha decay


The best-known source of alpha particles is alpha decay of heavier (> 106 u atomic weight) atoms. When an atom emits an alpha particle in alpha decay, the atom's mass number decreases by four due to the loss of the four nucleons in the alpha particle. The atomic number of the atom goes down by two, as a result of the loss of two protons – the atom becomes a new element. Examples of this sort of nuclear transmutation by alpha decay are the decay of uranium to thorium, and that of radium to radon.
Alpha particles are commonly emitted by all of the larger
Mechanism of production in alpha decay
In contrast to
However, the
Ternary fission
Especially energetic alpha particles deriving from a nuclear process are produced in the relatively rare (one in a few hundred)
Accelerators
Energetic helium nuclei (helium ions) may be produced by cyclotrons, synchrotrons, and other particle accelerators. Convention is that they are not normally referred to as "alpha particles."[citation needed]
Solar core reactions
Helium nuclei may participate in nuclear reactions in stars, and occasionally and historically these have been referred to as alpha reactions (see triple-alpha process and alpha process).
Cosmic rays
In addition, extremely high energy helium nuclei sometimes referred to as alpha particles make up about 10 to 12% of cosmic rays. The mechanisms of cosmic ray production continue to be debated.
Energy and absorption

The energy of the alpha particle emitted in alpha decay is mildly dependent on the half-life for the emission process, with many orders of magnitude differences in half-life being associated with energy changes of less than 50%, shown by the Geiger–Nuttall law.
The energy of alpha particles emitted varies, with higher energy alpha particles being emitted from larger nuclei, but most alpha particles have energies of between 3 and 7
With a typical kinetic energy of 5 MeV; the speed of emitted alpha particles is 15,000 km/s, which is 5% of the speed of light. This energy is a substantial amount of energy for a single particle, but their high mass means alpha particles have a lower speed than any other common type of radiation, e.g. β particles, neutrons.[12]
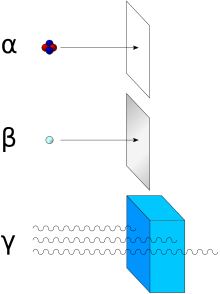
Because of their charge and large mass, alpha particles are easily absorbed by materials, and they can travel only a few centimetres in air. They can be absorbed by tissue paper or by the outer layers of human skin. They typically penetrate skin about 40 micrometres, equivalent to a few cells deep.
Biological effects
Due to the short range of absorption and inability to penetrate the outer layers of skin, alpha particles are not, in general, dangerous to life unless the source is ingested or inhaled.
When alpha particle emitting
History of discovery and use


In 1899, physicists
Rutherford's work also included measurements of the ratio of an alpha particle's mass to its charge, which led him to the hypothesis that alpha particles were doubly charged helium ions (later shown to be bare helium nuclei).[18] In 1907, Ernest Rutherford and Thomas Royds finally proved that alpha particles were indeed helium ions.[19] To do this they collected and purified the gas emitted by radium, a known alpha particle emitter, in a glass tube. An electric spark discharge inside the tube produced light. Subsequent study of the spectra of this light showed that the gas was helium and thus the alpha particles were indeed the helium ions.[9]: 61
Because alpha particles occur naturally, but can have
It was found that some of the alpha particles were deflected at much larger angles than expected (at a suggestion by Rutherford to check it) and some even bounced almost directly back. Although most of the alpha particles went straight through as expected, Rutherford commented that the few particles that were deflected was akin to shooting a fifteen-inch shell at tissue paper only to have it bounce off, again assuming the "plum pudding" theory was correct. It was determined that the atom's positive charge was concentrated in a small area in its center, making the positive charge dense enough to deflect any positively charged alpha particles that came close to what was later termed the nucleus.
Prior to this discovery, it was not known that alpha particles were themselves atomic nuclei, nor was the existence of protons or neutrons known. After this discovery, J.J. Thomson's "plum pudding" model was abandoned, and Rutherford's experiment led to the Bohr model and later the modern wave-mechanical model of the atom.
In 1917, Rutherford went on to use alpha particles to accidentally produce what he later understood as a directed nuclear transmutation of one element to another. Transmutation of elements from one to another had been understood since 1901 as a result of natural radioactive decay, but when Rutherford projected alpha particles from alpha decay into air, he discovered this produced a new type of radiation which proved to be hydrogen nuclei (Rutherford named these protons). Further experimentation showed the protons to be coming from the nitrogen component of air, and the reaction was deduced to be a transmutation of nitrogen into oxygen in the reaction
- 14N + α → 17O + p
This was the first discovered nuclear reaction.
To the adjacent pictures: According to the energy-loss curve by Bragg, it is recognizable that the alpha particle indeed loses more energy on the end of the trace.[20]
Anti-alpha particle
In 2011, members of the international
Applications
Devices
- Some smoke detectors contain a small amount of the alpha emitter americium-241.[23] The alpha particles ionize air within a small gap. A small current is passed through that ionized air. Smoke particles from fire that enter the air gap reduce the current flow, sounding the alarm. The isotope is extremely dangerous if inhaled or ingested, but the danger is minimal if the source is kept sealed. Many municipalities have established programs to collect and dispose of old smoke detectors, to keep them out of the general waste stream. However the US EPA says they "may be thrown away with household garbage".[23]
- Alpha decay can provide a safe power source for space probes. Alpha decay is much more easily shielded against than other forms of radioactive decay. Plutonium-238, a source of alpha particles, requires only 2.5 mm of leadshielding to protect against unwanted radiation.
- Static eliminators typically use polonium-210, an alpha emitter, to ionize air, allowing the "static cling" to more rapidly dissipate.[25][26]
Cancer treatment
Alpha-emitting
Radium-223 is an alpha emitter that is naturally attracted to the bone because it is a calcium mimetic. Radium-223 (as radium-223 dichloride) can be infused into a cancer patient's veins, after which it migrates to parts of the bone where there is rapid turnover of cells due to the presence of metastasized tumors. Once within the bone, Ra-223 emits alpha radiation that can destroy tumor cells within a 100-micron distance. This approach has been in use since 2013 to treat prostate cancer which has metastasized to the bone.[27] Radionuclides infused into the circulation are able to reach sites that are accessible to blood vessels. This means, however, that the interior of a large tumor that is not vascularized (i.e. is not well penetrated by blood vessels) may not be effectively eradicated by the radioactivity.
Radium-224 is a radioactive atom that is utilized as a source of alpha radiation in a cancer treatment device called DaRT (Diffusing Alpha-emitters Radiation Therapy). Each radium-224 atom undergoes a decay process producing 6 daughter atoms. During this process, 4 alpha particles are emitted. The range of an alpha particle—up to 100 microns—is insufficient to cover the width of many tumors. However, radium-224's daughter atoms can diffuse up to 2–3 mm in the tissue, thus creating a "kill region" with enough radiation to potentially destroy an entire tumor, if the seeds are placed appropriately.[28] Radium-224's half-life is short enough at 3.6 days to produce a rapid clinical effect while avoiding the risk of radiation damage due to overexposure. At the same time, the half-life is long enough to allow for handling and shipping the seeds to a cancer treatment center at any location across the globe.
Targeted alpha therapy for solid tumors involves attaching an alpha-particle-emitting radionuclide to a tumor-targeting molecule such as an antibody, that can be delivered by intravenous administration to a cancer patient.[29]
Alpha radiation and DRAM errors
In computer technology,
See also
- Alpha nuclide
- Alpha process (Also known as alpha-capture, or the alpha-ladder)
- Beta particle
- Cosmic rays
- Helion, the nucleus of helium-3 rather than helium-4
- List of alpha emitting materials
- Nuclear physics
- Particle physics
- Radioactive isotope
- Rays:
- Rutherford scattering
References
- ^ "2018 CODATA Value: alpha particle mass". The NIST Reference on Constants, Units, and Uncertainty. NIST. 20 May 2019. Retrieved 10 September 2022.
- ^ "2018 CODATA Value: alpha particle mass in u". The NIST Reference on Constants, Units, and Uncertainty. NIST. 20 May 2019. Retrieved 11 September 2022.
- ^ "2018 CODATA Value: alpha particle mass energy equivalent in MeV". The NIST Reference on Constants, Units, and Uncertainty. NIST. 20 May 2019. Retrieved 11 September 2022.
- ^ a b
Krane, Kenneth S. (1988). Introductory Nuclear Physics. ISBN 978-0-471-80553-3.
- OCLC 940282526.
- ^ a b Rutherford distinguished and named α and β rays on page 116 of: E. Rutherford (1899) "Uranium radiation and the electrical conduction produced by it," Philosophical Magazine, Series 5, vol. 47, no. 284, pages 109–163. Rutherford named γ rays on page 177 of: E. Rutherford (1903) "The magnetic and electric deviation of the easily absorbed rays from radium," Philosophical Magazine, Series 6, vol. 5, no. 26, pages 177–187.
- ^ Rutherford, Ernest, and Hans Geiger. "The Charge and Nature of the α-Particle." The Collected Papers of Lord Rutherford of Nelson. Routledge, 2014. 109-120.
- ISSN 0950-1207.
- ^ ISBN 978-0-19-851997-3.
- ISSN 0077-8923.
- OCLC 43118182.
- electromagnetic (light) they move at the speed of light (c). Beta particles often move at a large fraction of c, and exceed 60% c whenever their energy is > 64 keV, which it commonly is. Neutron velocity from nuclear reactions ranges from about 6% c for fission to as much as 17% c for fusion.
- ^
Christensen, D. M.; Iddins, C. J.; Sugarman, S. L. (2014). "Ionizing radiation injuries and illnesses". Emergency Medicine Clinics of North America. 32 (1): 245–65. PMID 24275177.
- ^
Grellier, James; et al. (2017). "Risk of lung cancer mortality in nuclear workers from internal exposure to alpha particle-emitting radionuclides". Epidemiology. 28 (5): 675–684. PMID 28520643.
- ^
Radford, Edward P.; Hunt, Vilma R. (1964). "Polonium-210: A Volatile Radioelement in Cigarettes". S2CID 23455633.
- ^ Cowell, Alan (24 November 2006). "Radiation Poisoning Killed Ex-Russian Spy". The New York Times. Retrieved 15 September 2011.
- ^
Little, John B.; Kennedy, Ann R.; McGandy, Robert B. (1985). "Effect of Dose Rate on the Induction of Experimental Lung Cancer in Hamsters by α Radiation". Radiation Research. 103 (2): 293–9. PMID 4023181.
- ISBN 0671621300.
- ^ E. Rutherford and T. Royds (1908) "Spectrum of the radium emanation," Philosophical Magazine, Series 6, vol. 16, pages 313–317.
- ^ Magazine "nuclear energy" (III/18 (203) special edition, Volume 10, Issue 2 /1967.
- ^
Agakishiev, H.; et al. (S2CID 4359058.
- ^
"Antihelium-4: Physicists nab new record for heaviest antimatter". PhysOrg. 24 April 2011. Retrieved 15 November 2011.
- ^ a b "Americium in Ionization Smoke Detectors". U.S. Environmental Protection Agency. Archived from the original on 27 September 2023. Retrieved 30 December 2023.
- ^ Schulman, Fred. "Isotopes and Isotope Thermoelectric Generators." Space Power Systems Advanced Technology Conference. No. N67-10265. 1966.
- ^ "Static Eliminators (1960s and 1980s)". Retrieved 30 December 2023.
- ^ Silson, John E. "Hazards in the use of radioactive static eliminators and their control." American Journal of Public Health and the Nations Health 40.8 (1950): 943-952.
- PMID 23863050.
- S2CID 1585204.
- PMID 31779154.
- S2CID 43748644.
Further reading
- Tipler, Paul; Llewellyn, Ralph (2002). Modern Physics (4th ed.). ISBN 978-0-7167-4345-3.
External links
![]() Media related to Alpha particles at Wikimedia Commons
Media related to Alpha particles at Wikimedia Commons


