Aluminium hydroxide
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Aluminium hydroxide | |
| Systematic IUPAC name
Trihydroxidoaluminium | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.040.433 |
| KEGG | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| Properties[1][2] | |
| Al(OH)3 | |
| Molar mass | 78.003 g·mol−1 |
| Appearance | White amorphous powder
|
| Density | 2.42 g/cm3, solid |
| Melting point | 300 °C (572 °F; 573 K) |
| 0.0001 g/(100 mL) | |
Solubility product (Ksp)
|
3×10−34 |
| Solubility | soluble in acids and alkalis |
| Acidity (pKa) | >7 |
| Isoelectric point | 7.7 |
| Thermochemistry[3] | |
Std enthalpy of (ΔfH⦵298)formation |
−1277 kJ·mol−1 |
| Pharmacology[4] | |
| A02AB01 (WHO) | |
| Hazards | |
| GHS labelling: | |
| no GHS pictograms | |
| no hazard statements | |
| P261, P264, P271, P280, P304+P340, P305+P351+P338, P312, P337+P313 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
>5000 mg/kg (rat, oral) |
| Safety data sheet (SDS) | External MSDS |
| Related compounds | |
Other anions
|
None |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Aluminium hydroxide,
Structure
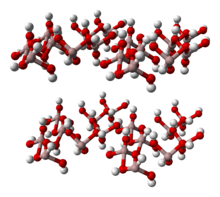
Al(OH)3 is built up of double layers of hydroxyl groups with aluminium ions occupying two-thirds of the octahedral holes between the two layers.
- gibbsite is also known as γ-Al(OH)3 [8] or α-Al(OH)3 [citation needed]
- ]
- nordstrandite is also known as Al(OH)3[8]
- doyleite
Hydrargillite, once thought to be aluminium hydroxide, is an aluminium phosphate. Nonetheless, both gibbsite and hydrargillite refer to the same polymorphism of aluminium hydroxide, with gibbsite used most commonly in the United States and hydrargillite used more often in Europe. Hydrargillite is named after the Greek words for water (hydra) and clay (argylles).[citation needed]
Properties
Aluminium hydroxide is
- 3 HCl + Al(OH)3 → AlCl3 + 3 H2O
In bases, it acts as a Lewis acid by binding hydroxide ions:[9]
- Al(OH)3 + OH− → [Al(OH)4]−
Production

Virtually all the aluminium hydroxide used commercially is manufactured by the
The residue or
Uses
Filler and fire retardant
Aluminium hydroxide finds use as a fire retardant filler for polymer applications. It is selected for these applications because it is colorless (like most polymers), inexpensive, and has good fire retardant properties.[12] Magnesium hydroxide and mixtures of huntite and hydromagnesite are used similarly.[13][14][15][16][17] It decomposes at about 180 °C (356 °F), absorbing a considerable amount of heat in the process and giving off water vapour.
In addition to behaving as a fire retardant, it is very effective as a smoke suppressant in a wide range of polymers, most especially in polyesters, acrylics, ethylene vinyl acetate, epoxies, polyvinyl chloride (PVC) and rubber.[18]
Aluminium hydroxide is used as filler in some artificial stone compound material, often in acrylic resin.[citation needed]
Precursor to Al compounds
Aluminium hydroxide is a
Freshly precipitated aluminium hydroxide forms
Pharmaceutical
Under the generic name "algeldrate", aluminium hydroxide is used as an
This compound is also used to control
Precipitated aluminium hydroxide is included as an adjuvant in some vaccines (e.g. anthrax vaccine). One of the well-known brands of aluminium hydroxide adjuvant is Alhydrogel, made by Brenntag Biosector.[25][full citation needed][dead link] Since it absorbs protein well, it also functions to stabilize vaccines by preventing the proteins in the vaccine from precipitating or sticking to the walls of the container during storage. Aluminium hydroxide is sometimes called "alum", a term generally reserved for one of several sulfates.[citation needed]
Vaccine formulations containing aluminium hydroxide stimulate the
Safety
In the 1960s and 1970s it was speculated that aluminium was related to various neurological disorders, including Alzheimer's disease.[29][30] Since then, multiple epidemiological studies have found no connection between exposure to environmental or swallowed aluminium and neurological disorders, though injected aluminium was not looked at in these studies.[31][32][33]
Neural disorders were found in experiments on mice motivated by
References
- ^ For solubility product: "Solubility product constants". Archived from the original on 15 June 2012. Retrieved 17 May 2012.
- ISSN 0008-4042.
- ISBN 978-0-618-94690-7.
- PMID 12825840. Retrieved 1 July 2017.
- ^ Wells, A. F. (1975), Structural Inorganic Chemistry (4th ed.), Oxford: Clarendon Press
- ^ ISBN 9780751401035.
- ISBN 978-0-470-58768-3.
- ^ OCLC 894928306.
- ^ a b Boundless (26 July 2016). "Basic and Amphoteric Hydroxides". Boundless Chemistry. Archived from the original on 22 August 2017. Retrieved 2 July 2017.
- .
- ^ "Hungary Battles to Stem Torrent of Toxic Sludge". BBC News Website. 5 October 2010.
- ^ ISBN 978-3527306732.
- .
- .
- .
- .
- S2CID 96208830.
- ^ Huber Engineered Materials. "Huber Non-Halogen Fire Retardant Additives" (PDF). Retrieved 3 July 2017.
- ^ Galbraith, A; Bullock, S; Manias, E; Hunt, B; Richards, A (1999). Fundamentals of pharmacology: a text for nurses and health professionals. Harlow: Pearson. p. 482.
- ISBN 9781416028888.
- ISBN 978-0-8493-5444-1.
- ISBN 9780323444026.
- ISBN 9780470959640.
- ^ Lifelearn Inc. (1 November 2010). "Aluminum Hydroxide". Know Your Pet. Retrieved 30 June 2017.
- ^ "About Brenntag Biosector - Brenntag". brenntag.com. Retrieved 19 April 2018.
- PMID 18362170.
- S2CID 154670.
- ISBN 978-1-59259-399-6.
- ^ "Alzheimer's Myth's". Alzheimer's Association. Retrieved 29 July 2012.
- ^ Khan, A (1 September 2008). "Aluminium and Alzheimer's disease". Alzheimer's Society. Archived from the original on 11 March 2012. Retrieved 8 March 2012.
- PMID 12222737.
- S2CID 32190038.
- PMID 9861186.
- PMID 19740540.
External links
- International Chemical Safety Card 0373
- "Some properties of aluminum hydroxide precipitated in the presence of clays", Soil Research Institute, R C Turner, Department of Agriculture, Ottawa[permanent dead link]
- Effect of ageing on properties of polynuclear hydroxyaluminium cations
- A second species of polynuclear hydroxyaluminium cation, its formation and some of its properties

