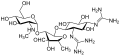
Aminoglycoside
Aminoglycoside is a

Nomenclature
Aminoglycosides that are derived from bacteria of the Streptomyces genus are named with the suffix -mycin, whereas those that are derived from Micromonospora[4] are named with the suffix -micin.[5] However, this nomenclature system is not specific for aminoglycosides, and so appearance of this set of suffixes does not imply common mechanism of action. (For instance, vancomycin, a glycopeptide antibiotic,[6] and erythromycin,[7] a macrolide antibiotic produced by Saccharopolyspora erythraea, along with its synthetic derivatives clarithromycin and azithromycin, all share the suffixes but have notably different mechanisms of action.)
In the following gallery, kanamycin A to netilmicin are examples of the 4,6-disubstituted deoxystreptamine sub-class of aminoglycosides, the neomycins are examples of the 4,5-disubstituted sub-class, and streptomycin is an example of a non-deoxystreptamine aminoglycoside.[2]
-
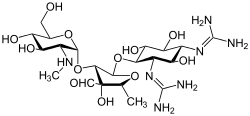
Kanamycin A
Mechanisms of action
Aminoglycosides display concentration-dependent bactericidal activity against "most gram-negative aerobic and facultative anaerobic bacilli" but not against gram-negative anaerobes and most gram-positive bacteria.[3] They require only short contact time, and are most effective against susceptible bacterial populations that are rapidly multiplying.[8] These activities are attributed to a primary mode of action as protein synthesis inhibitors, though additional mechanisms are implicated for some specific agents, and/or thorough mechanistic descriptions are as yet unavailable.[2][3][8]
The inhibition of protein synthesis is mediated through aminoglycosides' energy-dependent, sometimes irreversible binding, to the
It has been proposed that aminoglycoside antibiotics cause oxidation of guanine nucleotides in the bacterial nucleotide pool, and that this contributes to the cytotoxicity of these antibiotics.
Finally, a further "cell-membrane effect" also occurs with aminoglycosides; "functional integrity of the bacterial cell membrane" can be lost, later in time courses of aminoglycoside exposure and transport.[11]
Pharmacokinetics and pharmacodynamics
There is a significant variability in the relationship between the dose administered and the resultant plasma level in blood.[
Indications
Aminoglycosides are useful primarily in infections involving
As noted, aminoglycosides are mostly ineffective against anaerobic bacteria, fungi, and viruses.
Nonsense suppression
The interference with mRNA proofreading has been exploited to treat genetic diseases that result from premature
Routes of administration
Since they are not absorbed from the gut, they are administered
Clinical use
The recent emergence of infections due to Gram-negative bacterial strains with advanced patterns of antimicrobial resistance has prompted physicians to reevaluate the use of these antibacterial agents.[16] This revived interest in the use of aminoglycosides has brought back to light the debate on the two major issues related to these compounds, namely the spectrum of antimicrobial susceptibility and toxicity. Current evidence shows that aminoglycosides do retain activity against the majority of Gram-negative clinical bacterial isolates in many parts of the world. Still, the relatively frequent occurrence of nephrotoxicity and ototoxicity during aminoglycoside treatment makes physicians reluctant to use these compounds in everyday practice. Recent advances in the understanding of the effect of various dosage schedules of aminoglycosides on toxicity have provided a partial solution to this problem, although more research still needs to be done in order to overcome this problem entirely.[17]
Aminoglycosides are in
Adverse effects
Aminoglycosides can cause inner ear toxicity which can result in sensorineural hearing loss.[19] The incidence of inner ear toxicity varies from 7 to 90%, depending on the types of antibiotics used, susceptibility of the patient to such antibiotics, and the duration of antibiotic administration.[20]
Another serious and disabling side effect of aminoglycoside use is vestibular ototoxicity.[19] This leads to oscillopsia (gaze instability) and balance impairments that impact all aspects of an individual's antigravity function. This loss is permanent and can happen at any dose.[21][22][23][24]
Frequent use of aminoglycosides could result in kidney damage (Acute kidney injury), that could lead to chronic kidney disease.[25]
Contraindication for specific diseases
Aminoglycosides can exacerbate weakness in patients with myasthenia gravis, and use is therefore avoided in these patients.[26]
Aminoglycosides are contraindicated in patients with mitochondrial diseases as they may result in impaired mtDNA translation, which can lead to irreversible hearing loss, tinnitus, cardiac toxicity, and renal toxicity. However, hearing loss and tinnitus have also been observed in some patients without mitochondrial diseases.[27]
References
- ^ E.g., see www.merriam-webster.com/medical/aminoglycoside: "any of a group of antibiotics (as streptomycin and neomycin) that inhibit bacterial protein synthesis and are active especially against gram-negative bacteria".
- ^ PMID 10103173.
- ^ a b c ME Levison, MD, 2012, Aminoglycosides, The Merck Manual [1], accessed 22 February 2014.
- PMID 15997706.
- ISBN 978-0-470-74167-2.
- PMID 4451345.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - ^ The Mechanism of Action of Macrolides, Lincosamides and Streptogramin B Reveals the Nascent Peptide Exit Path in the Ribosome Martin Lovmar and Måns Ehrenberg
- ^ a b c d e DVM Boothe, DVM, PhD, 2012, Aminoglycosides (Aminocyclitols), The Merck Veterinary Manual "Aminoglycosides: Antibacterial Agents: Merck Veterinary Manual". Archived from the original on 2014-03-01. Retrieved 2014-02-22., accessed 22 February 2014.
- PMID 33619089.
- ^ a b Foti JJ, Devadoss B, Winkler JA, Collins JJ, Walker GC. Oxidation of the guanine nucleotide pool underlies cell death by bactericidal antibiotics. Science. 2012 Apr 20;336(6079):315-9. doi: 10.1126/science.1219192. PMID 22517853; PMCID: PMC3357493
- ^ As Boothe notes, "high concentrations of aminoglycosides may cause nonspecific membrane toxicity, even to the point of bacterial cell lysis", though the physiologic relevance of these concentrations to specific clinical situations is unclear. DVM Boothe, DVM, PhD, 2012, Aminoglycosides (Aminocyclitols), The Merck Veterinary Manual "Aminoglycosides: Antibacterial Agents: Merck Veterinary Manual". Archived from the original on 2014-03-01. Retrieved 2014-02-22., accessed 22 February 2014.
- ^ "Aminoglycosides - Infectious Diseases". MSD Manual Professional Edition. Retrieved 2021-12-14.
- S2CID 5809127.
- PMID 14534336.
- S2CID 30108514.
- S2CID 13158593.
- PMID 18976888.
- ^ Merck Manual: Bacteria and Antibacterial Drugs: Aminoglycosides Last full review/revision July 2009 by Matthew E. Levison, MD
- ^ PMID 17266591.
- .
- S2CID 40285162.
- PMID 22554194.
- S2CID 1412868.
- S2CID 5883996.
- PMID 20627927.
- ^ Gautam Mehta and Bilal Iqbal. Clinical Medicine for the MRCP PACES. Volume 1. Core Clinical Skills. Oxford University Press. 2010.
- S2CID 10280462.
External links
- MedlinePlus drug information - Aminoglycosides (Systemic)
- Science Daily Bacterial 'Battle for Survival' - Rhodostreptomycin