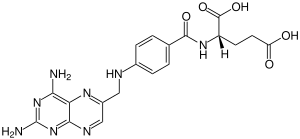
Aminopterin
| |
| |
| Names | |
|---|---|
| IUPAC name
N-(4-{[(2,4-Diaminopteridin-6-yl)methyl]amino}benzoyl)-L-glutamic acid
| |
| Systematic IUPAC name
(2S)-2-(4-{[(2,4-Diaminopteridin-6-yl)methyl]amino}benzamido)pentanedioic acid | |
| Other names
4-Aminofolic acid
4-Aminopteroylglutamic acid Aminopterin sodium Aminopteroylglutamic acid | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.000.191 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C19H20N8O5 | |
| Molar mass | 440.41 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Aminopterin (or 4-aminopteroic acid), the 4–amino derivative of
It is classified as an
Uses
Discovered by
During the period Aminopterin was marketed, the agent was used off-label to safely treat over 4,000 patients with psoriasis in the United States, producing dramatic clearing of lesions.[4]
The use of aminopterin in cancer treatment was supplanted in the 1950s by methotrexate due to the latter's better therapeutic index in a rodent tumor model.[5] Now in a more pure preparation and supported by laboratory evidence of superior tumor cell uptake in vitro, aminopterin is being investigated in clinical trials in leukemia as a potentially superior antifolate to methotrexate.[6]
The compound was explored as an
The reference to the use of Aminopterin as a rodenticide (i.e. rat poison) dates back to a 1951 patent issued to the American Cyanamid Company that is commonly cited by a variety of reference textbooks including the Merck Manual, although the use of aminopterin as a
Aminopterin is widely used in selection media (such as
Implication in 2007 Menu Foods recall
On March 23, 2007, ABC News reported[12] that aminopterin was the chemical linked to the 2007
On March 27, the
Exposure and treatment
Symptoms of exposure in humans include:[18] [19]
- nausea
- vomiting
- anorexia
- weight loss
- chills
- fever
- stomatitis – inflammation of the oral mucosa
- pharyngitis – inflammation of the pharynx
- erythematous rashes – red rashes on the skin
- pigmentationassociated with cleared psoriatic lesions
- gastrointestinal hemorrhage
- renal failure– in high doses necessarily involving concomitant leucovorin rescue
- pregnantwomen
Supralethal doses of aminopterin may be rescued with the antidote
In humans, leucovorin rescue at overdosages lower than 10 mg aminopterin in an average 70 kg adult should comprise an initial leucovorin dose of at least 20 mg (10.0 mg/m2), given intravenously (preferably), or orally.[23] Subsequent doses of 20 mg (which may be taken orally) should be given at 6 hour intervals until hematological abnormalities are improved.
Massive aminopterin overdosage in humans (i.e. > 40 mg AMT in an average 70 kg adult), should be approached with an initial leucovorin dose of 100 mg (50 mg/m2), given intravenously and continued at 6 hour intervals until the hematological abnormalities are improved (likely 8–12 courses or more).[22] Additionally, to prevent reversible aminopterin-mediated nephrotoxicity manifesting as increases in serum creatinine and which further delays drug elimination, urinary alkalinization with NaHCO
3 and volume expansion should be considered in cases of massive aminopterin overdosage, particularly those involving greater than 100 mg AMT in an average 70 kg adult human.
Consistent with the known enterohepatic cycling of the related antifolate methotrexate, oral
may promote excretion if an overdose of aminopterin is suspected. However, rescue with leucovorin should form the backbone of treatment.The vitamin folic acid is an oxidized precursor to reduced folates that is upstream of the blockade at dihydrofolate reductase, and compared to leucovorin is recognized as a very weak antidote to the toxic effects of antifolates that is inappropriate for use in cases of acute intoxication. Minnich et al. dosed mongrel dogs subcutaneously with aminopterin and folic acid simultaneously to test whether folic acid can rescue animals from the lethality and toxicity of aminopterin[24] Dogs were given 0.020, 0.046, 0.044 escalated to 0.088, and 0.097 mg/kg aminopterin each day for 7 to 12 days. Folic acid was given in a weight ratio to aminopterin of 200:1 to 800:1. All animals survived. In contrast, animals given aminopterin in an amount of 0.041 mg/kg/day x 6 days without folic acid died. Thus, when the ratio of folic acid to aminopterin was 200:1 and greater, all of the subjects survived on regimens that would have otherwise been uniformly fatal to all subjects.
Similar effects have been noted in rodent species as well, where the range for rescue by folic acid was fairly narrow and highly dependent on the timing (optimal of 1 hour prior to aminopterin) of administration in relation to aminopterin.[25][26] The temporal relationship between folic acid administration and rescue has been interpreted as the necessary period of time required for the vitamin to be converted in vivo to reduced forms.
References
- ^ "40 C.F.R.: Appendix A to Part 355—The List of Extremely Hazardous Substances and Their Threshold Planning Quantities" (PDF) (July 1, 2008 ed.). Government Printing Office. Archived from the original (PDF) on February 25, 2012. Retrieved October 29, 2011.
{{cite journal}}: Cite journal requires|journal=(help) - ^ "Who was Sidney Farber, MD?". Dana–Farber Cancer Institute. Archived from the original on 7 February 2007. Retrieved 2007-02-05.
- PMID 18860765.
- PMID 14206858.[permanent dead link]
- PMID 14381889.
- ^ PMID 16299240.
- PMID 13890101.
- ^ "Multiple Congenital Anomaly/Mental Retardation (MCA/MR) Syndromes: Fetal aminopterin syndrome". United States National Library of Medicine. Retrieved 2007-03-26.
- ^ "No Aminopterin in Tissues of Animals Killed by Recalled Pet Food". PRNewsWire. March 30, 2007. Archived from the original on 9 May 2007. Retrieved 2007-04-14.
- ^ US 2575168, Franklin, Alfred L., "Rodenticide comprising 4-amino-pteroylglutamic acid", published November 13, 1951, assigned to American Cyanamid Company
- ^ "EPA Chemical Profile: Aminopterin". United States Environmental Protection Agency. October 31, 1985. Archived from the original on July 7, 2007. Retrieved 2007-03-23.
- ^ Kerley, David (March 23, 2007). "Rat Poison to Blame for Pet Food Contamination". ABC News. Retrieved 2007-03-23.
- ^ "Menu Foods Issues Recall Of Specific Can And Small Foil Pouch Wet Pet Foods". New York Department of Agriculture. March 16, 2007. Archived from the original on 2016-12-25. Retrieved 2007-03-25.
- ^
Johnson, Mark (March 23, 2007). "Rat poison found in tainted pet food". BusinessWeek. Archived from the originalon May 22, 2011. Retrieved 2007-03-23.
- ^ "New York Laboratories Identify Toxin In Recalled Pet Food". New York Department of Agriculture. March 23, 2007. Archived from the original on 2017-02-11. Retrieved 2007-03-23.
- ^ "ASPCA Advises Caution As Pet Food Recall Crisis Grows; Other Contaminants May Be Involved in the Menu Foods Recall". March 27, 2007. Archived from the original on 2007-05-13. Retrieved 2007-03-27.
- ^ "FDA finds new chemical in recalled pet food, sick animals". CNN. March 30, 2007. Archived from the original on 2007-04-08. Retrieved 2007-03-30.
- ^ "Chemical data sheet for Aminopterin". CAMEO Chemicals. U.S. National Oceanic and Atmospheric Administration. Retrieved 2007-03-25.
- PMID 14315108.
- S2CID 44623774.
- PMID 13973810.[permanent dead link]
- ^ PMID 383286.
- ^ PMID 9552052. Archived from the originalon 2013-04-15.
- PMID 14789323.
- S2CID 41223103.
- PMID 14772718.
