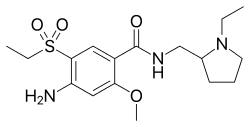
Amisulpride
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Solian, Barhemsys, others |
| Other names | APD421 |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category | |
| Routes of administration | By mouth, intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 48%[5][6] |
| Protein binding | 16%[6] |
| Metabolism | Liver (minimal; most excreted unchanged)[6] |
| Elimination half-life | 12 hours[5] |
| Excretion | Kidney[5] (23–46%),[7][8] Faecal[6] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Amisulpride is an antiemetic and antipsychotic medication used at lower doses intravenously to prevent and treat postoperative nausea and vomiting; and at higher doses by mouth to treat schizophrenia and acute psychotic episodes. It is sold under the brand names Barhemsys[9] (as an antiemetic) and Solian, Socian, Deniban and others (as an antipsychotic).[6] At very low doses it is also used to treat dysthymia.[10]
It is usually classed with the
Amisulpride is indicated for use in the United States in adults for the prevention of postoperative nausea and vomiting (PONV), either alone or in combination with an antiemetic of a different class; and to treat PONV in those who have received antiemetic prophylaxis with an agent of a different class or have not received prophylaxis.[9]
Amisulpride is believed to work by blocking, or antagonizing, the
It was introduced by
Medical uses
Schizophrenia
Although according to other studies it appears to have comparable efficacy to olanzapine in the treatment of schizophrenia,[15] amisulpride augmentation, similarly to sulpiride augmentation, has been considered a viable treatment option (although this is based on low-quality evidence) in clozapine-resistant cases of schizophrenia.[16][17] Another recent study concluded that amisulpride is an appropriate first-line treatment for the management of acute psychosis.[18]
Postoperative nausea and vomiting
Amisulpride is indicated for use in the United States in adults for the prevention of postoperative nausea and vomiting (PONV), either alone or in combination with an antiemetic of a different class; and to treat PONV in those who have received antiemetic prophylaxis with an agent of a different class or have not received prophylaxis.[9]
Contraindications
Amisulpride's use is contraindicated in the following disease states and populations[6][19][11]
- Pheochromocytoma
- Concomitant prolactin-dependent tumours e.g. prolactinoma, breast cancer
- Movement disorders (e.g. Parkinson's disease and dementia with Lewy bodies)
- Lactation
- Children before the onset of puberty
Neither is it recommended to use amisulpride in patients with hypersensitivities to amisulpride or the excipients found in its dosage form.[6]
Adverse effects
- Very Common (≥10% incidence)[4]
- ).
- Insomnia
- Somnolence
- Hypersalivation
- Nausea
- Headache
- Hyperactivity
- Vomiting
- Hyperprolactinaemia (which can lead to galactorrhoea, breast enlargement and tenderness, sexual dysfunction, etc.)
- Weight gain (produces less weight gain than chlorpromazine, clozapine, iloperidone, olanzapine, paliperidone, quetiapine, risperidone, sertindole, zotepine and more (although not statistically significantly) weight gain than haloperidol, lurasidone, ziprasidone and approximately as much weight gain as aripiprazole and asenapine)[12]
- Anticholinergic side effects (although it does not bind to the muscarinic acetylcholine receptors and hence these side effects are usually quite mild) such as
- - constipation
- - dry mouth
- - disorder of accommodation
- - Blurred vision
- Rare (<1% incidence)[4][6][20][19][11]
- Hyponatraemia
- Bradycardia
- Hypotension
- Palpitations
- Urticaria
- Seizures
- Mania
- Oculogyric crisis
- Tardive dyskinesia
- Blood leucopenia, neutropenia and agranulocytosis
- QT interval prolongation (in a recent meta-analysis of the safety and efficacy of 15 antipsychotic drugs amisulpride was found to have the 2nd highest effect size for causing QT interval prolongation[12])
Hyperprolactinaemia results from antagonism of the
Discontinuation
The British National Formulary recommends a gradual withdrawal when discontinuing antipsychotics to avoid acute withdrawal syndrome or rapid relapse.[24] Symptoms of withdrawal commonly include nausea, vomiting, and loss of appetite.[25] Other symptoms may include restlessness, increased sweating, and trouble sleeping.[25] Less commonly there may be a feeling of the world spinning, numbness, or muscle pains.[25] Symptoms generally resolve after a short period of time.[25]
There is tentative evidence that discontinuation of antipsychotics can result in psychosis.[26] It may also result in reoccurrence of the condition that is being treated.[27] Rarely tardive dyskinesia can occur when the medication is stopped.[25]
Overdose
Interactions
Amisulpride should not be used in conjunction with drugs that prolong the QT interval (such as
Pharmacology
Pharmacodynamics
| Site | Ki (nM) | Species | Ref |
|---|---|---|---|
| SERT | >10,000 | Human | [33] |
| NET | >10,000 | Human | [33] |
| DAT | >10,000 | Human | [33] |
| 5-HT1A | >10,000 | Human | [33] |
| 5-HT1B | 1,744 | Human | [33] |
| 5-HT1D | 1,341 | Human | [33] |
| 5-HT1E | >10,000 | Human | [33] |
| 5-HT2A | 8,304 | Human | [33] |
| 5-HT2B | 13 | Human | [33] |
| 5-HT2C | >10,000 | Human | [33] |
| 5-HT3 | >10,000 | Human | [33] |
| 5-HT5A | >10,000 | Human | [33] |
| 5-HT6 | 4,154 | Human | [33] |
| 5-HT7 | 11.5 | Human | [33] |
| α1A | >10,000 | Human | [33] |
| α1B | >10,000 | Human | [33] |
| α1D | >10,000 | Human | [33] |
| α2A | 1,114 | Human | [33] |
| α2C | 1,540 | Human | [33] |
| β1 | >10,000 | Human | [33] |
| β2 | >10,000 | Human | [33] |
| β3 | >10,000 | Human | [33] |
D1 |
>10,000 | Human | [33] |
D2 |
3.0 | Human | [33] |
D3 |
3.5 | Rat | [33] |
D4 |
2,369 | Human | [33] |
D5 |
>10,000 | Human | [33] |
H1 |
>10,000 | Human | [33] |
H2 |
>10,000 | Human | [33] |
H4 |
>10,000 | Human | [33] |
M1 |
>10,000 | Human | [33] |
M2 |
>10,000 | Human | [33] |
M3 |
>10,000 | Human | [33] |
M4 |
>10,000 | Human | [33] |
M5 |
>10,000 | Human | [33] |
| σ1 | >10,000 | Rat | [33] |
| σ2 | >10,000 | Rat | [33] |
MOR |
>10,000 | Human | [33] |
DOR |
>10,000 | Human | [33] |
KOR |
>10,000 | Human | [33] |
GHBHigh |
50 ( IC50 ) |
Rat | [34] |
| NMDA (PCP) |
>10,000 | Rat | [35] |
| Values are Ki (nM). The smaller the value, the more strongly the drug binds to the site. | |||
Amisulpride functions primarily as a
Amisulpride and its relatives
Amisulpride, sultopride and sulpiride respectively present decreasing in vitro affinities for the D2 receptor (IC50 = 27, 120 and 181 nM) and the D3 receptor (IC50 = 3.6, 4.8 and 17.5 nM).[36]
Though it was long widely assumed that dopaminergic modulation is solely responsible for the respective
Amisulpride also appears to bind with high affinity to the serotonin 5-HT2B receptor (Ki = 13 nM), where it acts as an antagonist.[33] The clinical implications of this, if any, are unclear.[33] In any case, there is no evidence that this action mediates any of the therapeutic effects of amisulpride.[33]
Amisulpride shows
Through a high direct unmetabolized excretion, it has, despite its high usual dose, also high affinity for dopamine-D2-D3-receptors. Also the available literature gives us hints about also relatively high receptor dissociation kinetics (through a delayed but high occupancy at dopamine receptors after 6 hours from a 100 mg exposure). Moreover, this dopamine exposure could be slightly more "balanced" providing some little advantages over haloperidol in using it for drug exposure. Due to its lack of compensatory serotonin effects and also not having an anticholinergic profile, it may not considered as an effective alternative if akathasia is a problem.[5][23][40]
Society and culture
Brand names
Brand names include: Amazeo, Amipride (
Availability
Amisulpride is not approved by the Food and Drug Administration for use in the United States in psychiatric indications, but it is approved and in use throughout Europe,[42] Asia, Mexico, New Zealand and Australia[6] to treat psychosis and schizophrenia.[43][44]
An IV formulation of Amisulpride was approved for the treatment of postoperative nausea and vomiting ("PONV") in the United States in February 2020.[45][9][46]
History of US clinical development
The U.S. Food and Drug Administration (FDA) approved a 10 mg/4mL amisulpride IV formulation for use in post-operative nausea based on evidence from four clinical trials of 2323 subjects undergoing surgery or experiencing nausea and vomiting after the surgery.[46] The trials were conducted at 80 sites in the United States, Canada and Europe.[46]
Two trials (Trials 1 and 2) enrolled subjects scheduled to have surgery.[46] Subjects were randomly assigned to receive either amisulpride or a placebo drug at the beginning of general anesthesia.[46] In Trial 1, subjects received amisulpride or placebo alone, and in Trial 2, they received amisulpride or placebo in combination with one medication approved for prevention of nausea and vomiting.[46] Neither the subjects nor the health care providers knew which treatment was being given until after the trial was complete.[46]
The trials counted the number of subjects who had no vomiting and did not use additional medications for nausea or vomiting in the first day (24 hours) after the surgery.[46] The results then compared amisulpride to placebo.[46]
The other two trials (Trials 3 and 4) enrolled subjects who were experiencing nausea and vomiting after surgery.[46] In Trial 3, subjects did not receive any medication to prevent nausea and vomiting before surgery and in Trial 4 they received the medication, but the treatment did not work.[46] In both trials, subjects were randomly assigned to receive either amisulpride or placebo.[46] Neither the subjects nor the health care providers knew which treatment was being given until after the trial was complete.[46]
The trials counted the number of subjects who had no vomiting and did not use additional medications for nausea or vomiting in the first day (24 hours) after the treatment.[46] The trial compared amisulpride to placebo.[46]
The FDA has not approved amisulpride for use in any psychiatric indication. LB Pharmaceuticals is developing N-methyl amisulpride for the use in the treatment of schizophrenia; a Phase 2 first-in-patient study is planned for 2023.[47]
See also
- SEP-4199, a non-racemic form of amisulpride
References
- ^ "Australian Product Information – Solian (Amisulpride) Tablets And Solution". TGA eBS. Retrieved 10 May 2020.
- ^ a b "Amisulpride (Barhemsys) Use During Pregnancy". Drugs.com. 2 September 2020. Retrieved 24 September 2020.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ a b c d "Amisulpride 100 mg Tablets - Summary of Product Characteristics (SmPC)". (emc). 5 July 2019. Archived from the original on 26 February 2020. Retrieved 26 February 2020.
- ^ S2CID 23877366.
- ^ a b c d e f g h i j k "Solian tablets and solution product information" (PDF). TGA eBusiness Services. Sanofi-Aventis Australia Pty Ltd. 27 September 2019. Retrieved 26 February 2020.
- S2CID 68853079.
- S2CID 71691764.
- ^ a b c d "Barhemsys (amisulpride) injection, for intravenous use" (PDF). U.S. Food and Drug Administration (FDA). February 2020. Retrieved 26 February 2020.
- ^ PMID 11920152.
- ^ ISBN 978-0-9805790-9-3.
- ^ S2CID 32085212.
- ^ a b Brayfield A, ed. (June 2017). "Amisulpride: Martindale: The Complete Drug Reference". MedicineComplete. Pharmaceutical Press. Retrieved 5 August 2017.
- .
- PMID 20091599.
- PMID 20048449.
- S2CID 29682562.
- PMID 18360610.
- ^ ISBN 978-0-85711-084-8.
- ^ a b Truven Health Analytics, Inc. DRUGDEX System (Internet) [cited 2013 Sep 19]. Greenwood Village, CO: Thomsen Healthcare; 2013.
- ISBN 978-0-07-162442-8.
- S2CID 9054960.
- ^ S2CID 11315672.
- ISBN 978-0-85369-845-6.
Withdrawal of antipsychotic drugs after long-term therapy should always be gradual and closely monitored to avoid the risk of acute withdrawal syndromes or rapid relapse.
- ^ ISBN 978-0-19-852748-0.
- S2CID 6267180.
- ISBN 978-8-84-702679-7.
- S2CID 205710487.
- S2CID 26412012.
- ^ ISBN 978-0-47-097948-8.
- S2CID 24628641.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- ^ PMID 19337725.
- ^ PMID 7914168.
- PMID 8996185.
- ISBN 978-1-4613-6857-1.
- ^ PMID 33961287.
- PMID 31497735.
- ^ "SEP 4199". AdisInsight. Springer Nature Switzerland AG.
- S2CID 146670706.
- ^ "Amisulpride international". Drugs.com. 3 February 2020. Retrieved 26 February 2020.
- ^ a b "Active substance: amisulpride" (PDF). 28 September 2017. EMA/658194/2017; Procedure no.: PSUSA/00000167/201701. Archived from the original (PDF) on 15 June 2018. Retrieved 26 February 2020.
- S2CID 21103201.
- ^ Kaplan A (2004). "Psychotropic Medications Around the World". Psychiatric Times. 21 (5).
- ^ "Barhemsys: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Retrieved 26 February 2020.
- ^ a b c d e f g h i j k l m n o "Drug Trials Snapshots: Barhemsys". U.S. Food and Drug Administration (FDA). 26 February 2020. Retrieved 27 March 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "Investor Presentation" (PDF). LB Pharmaceuticals. December 2022.
External links
- "Amisulpride". Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT01991860 for "US Phase III Study of APD421 in PONV" at ClinicalTrials.gov
- Clinical trial number NCT02337062 for "Phase IIIb Study of APD421 in Combination as PONV Prophylaxis" at ClinicalTrials.gov
