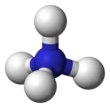
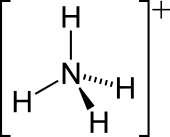
Ammonium
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Ammonium ion
| |||
| Systematic IUPAC name
Azanium[1] | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
| MeSH | D000644 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| [NH4]+ | |||
| Molar mass | 18.039 g·mol−1 | ||
| Acidity (pKa) | 9.25 | ||
Conjugate base
|
Ammonia | ||
| Structure | |||
| Tetrahedral | |||
| Related compounds | |||
Other cations
|
| ||
Related compounds
|
Ammonium radical •NH4 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
The ammonium
Acid–base properties

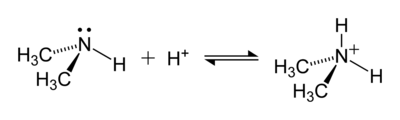
The ammonium ion is generated when ammonia, a weak base, reacts with
- H+ + NH3 → [NH4]+
The ammonium ion is mildly acidic, reacting with Brønsted bases to return to the uncharged ammonia molecule:
- [NH4]+ + B− → HB + NH3
Thus, the treatment of concentrated solutions of ammonium salts with a strong base gives ammonia. When ammonia is dissolved in water, a tiny amount of it converts to ammonium ions:
- H2O + NH3 ⇌ OH− + [NH4]+
The degree to which ammonia forms the ammonium ion depends on the
Formation of ammonium compounds can also occur in the vapor phase; for example, when ammonia vapor comes in contact with hydrogen chloride vapor, a white cloud of ammonium chloride forms, which eventually settles out as a solid in a thin white layer on surfaces.
Salts and characteristic reactions

Ammonium cation is found in a variety of
In an unusual process, ammonium ions form an amalgam. Such species are prepared by the addition of sodium amalgam to a solution of ammonium chloride.[2] This amalgam eventually decomposes to release ammonia and hydrogen.[3]
To find whether the ammonium ion is present in the salt, first, the salt is heated in presence of alkali hydroxide releasing a gas with a characteristic smell, which is ammonia.
- [NH4]+ + OH− NH3 + H2O
To further confirm ammonia, it passed through a glass rod dipped in an HCl solution (hydrochloric acid), creating white dense fumes of ammonium chloride.
- NH3(g) + HCl(aq) → [NH4]Cl(s)
Ammonia, when passed through CuSO4 (copper(II) sulfate) solution, changes its color from blue to deep blue, forming Schweizer's reagent.
- CuSO4(aq) + 4 NH3(aq) + 4 H2O → [Cu(NH3)4(H2O)2](OH)2(aq) + H2SO4(aq)
Ammonia or ammonium ion when added to
Ammonium ion when added to chloroplatinic acid gives a yellow precipitate of ammonium hexachloroplatinate(IV).
- H2[PtCl6](aq) + [NH4]+(aq) → [NH4]2[PtCl6](s) + 2 H+
Ammonium ion when added to
- Na3[Co(NO2)6](aq) + 3 [NH4]+(aq) → [NH4]3[Co(NO2)6](s) + 3 Na+(aq)
Ammonium ion gives a white precipitate of ammonium bitartrate when added to potassium bitartrate.
- KC4H5O6(aq) + [NH4]+(aq) → [NH4]C4H5O6(s) + K+(aq)
Structure and bonding
This section needs additional citations for verification. (March 2022) |
The lone electron pair on the nitrogen atom (N) in ammonia, represented as a line above the N, forms a coordinate bond with a proton (H+). After that, all four N−H bonds are equivalent, being polar covalent bonds. The ion has a tetrahedral structure and is isoelectronic with methane and the borohydride anion. In terms of size, the ammonium cation (rionic = 175 pm)[citation needed] resembles the caesium cation (rionic = 183 pm).[citation needed]
Organic ions
The hydrogen atoms in the ammonium ion can be substituted with an
An example of a reaction forming an ammonium ion is that between
Quaternary ammonium cations have four organic groups attached to the nitrogen atom, they lack a hydrogen atom bonded to the nitrogen atom. These cations, such as the
An unusual class of organic ammonium salts is derivatives of amine radical cations, [•NR3]+ such as tris(4-bromophenyl)ammoniumyl hexachloroantimonate.
Biology
Ammonium ions are a waste product of the
Ammonium is an important source of nitrogen for many plant species, especially those growing on hypoxic soils.[4] However, it is also toxic to most crop species and is rarely applied as a sole nitrogen source.[6]
Metal
The ammonium cation has very similar properties to the heavier
Under normal conditions, ammonium does not exist as a pure metal but does as an amalgam (alloy with mercury).[10]
See also
- Onium compounds
- Fluoronium, ([H2F]+ and substituted derivatives)
- organyl)
- Hydronium ([H3O]+, the simplest oxonium ion)
- Quaternary ammonium cation ([NR4]+, where R is organyl)
- Tetrafluoroammonium ([NF4]+)
- Hydrazinium ([H2N−NH3]+ and substituted derivatives)
- Hydrazinediium ([H3N−NH3]2+ and substituted derivatives)
- Iminium ([H2C=NH2]+ and substituted derivatives)
- Diazonium ([H−N≡N]+ and substituted derivatives)
- Diazynediium ([H−N≡N−H]2+ and substituted derivatives)
- Aminodiazonium ([H2N=N=N]+ ⇌ [H2N−N≡N]+ and substituted derivatives)
- Hydroxylammonium ([HO−NH3]+ and substituted derivatives)
- Ammonium transporter
- f-ratio
- Nitrification
- The Magnificent Possession (Isaac Asimov short story)
- Ammonium hydroxide
References
- ISBN 0-85404-438-8. pp. 71,105,314. Electronic version.
- ^ "Pseudo-binary compounds". Archived from the original on 2020-07-27. Retrieved 2007-10-12.
- ^ "Ammonium Salts". VIAS Encyclopedia.
- ^ ISBN 978-0-12-814608-8.)
{{cite book}}: CS1 maint: date and year (link - ISBN 978-0-8053-6624-2.
- .
- ISBN 0-12-352651-5
- ^ S2CID 4199721.
- ^ .
- .