Antiandrogen
| Antiandrogen | ||
|---|---|---|
Chemical class Steroidal; Nonsteroidal; Peptide | | |
| External links | ||
| MeSH | D000726 | |
| Legal status | ||
| In Wikidata | ||
Antiandrogens, also known as androgen antagonists or testosterone blockers, are a class of
Antiandrogens are used to treat an assortment of
There are a few different major types of antiandrogens.
Medical uses
Antiandrogens are used in the treatment of an assortment of
Men and boys
Prostate cancer
Androgens like testosterone and particularly DHT are importantly involved in the development and progression of prostate cancer.
The most common methods of androgen deprivation therapy currently employed to treat prostate cancer are
In addition to active treatment of prostate cancer, antiandrogens are effective as
Enlarged prostate
The 5α-reductase inhibitors finasteride and dutasteride are used to treat benign prostatic hyperplasia, a condition in which the prostate becomes enlarged and this results in urinary obstruction and discomfort.[37] They are effective because androgens act as growth factors in the prostate gland.[37] The antiandrogens chlormadinone acetate and oxendolone and the functional antiandrogens allylestrenol and gestonorone caproate are also approved in some countries for the treatment of benign prostatic hyperplasia.[38][39]
Scalp hair loss
5α-Reductase inhibitors like finasteride, dutasteride, and
Acne
Systemic antiandrogens are generally not used to treat acne in males due to their high risk of feminization (e.g., gynecomastia) and sexual dysfunction.[50][51] However, they have been studied for acne in males and found to be effective.[52][44][45][53] Clascoterone, a topical antiandrogen, is effective for acne in males and has been approved by the FDA in August 2020.[54][55][56][57]
Paraphilia
Androgens increase
Early puberty
Antiandrogens are used to treat
Long-lasting erections
Antiandrogens are effective in the treatment of recurrent
Women and girls
Skin and hair conditions
Antiandrogens are used in the treatment of androgen-dependent
High androgen levels
Hyperandrogenism is a condition in women in which androgen levels are excessively and abnormally high.
Transgender hormone therapy
Antiandrogens are used to prevent or reverse
Available forms
There are several different types of antiandrogens, including the following:[6]
- Androgen receptor antagonists: Drugs that bind directly to and block the AR.oral contraceptives and/or in menopausal HRT including dienogest, drospirenone, medrogestone, nomegestrol acetate, promegestone, and trimegestone also have varying degrees of AR antagonistic activity.[88][89][90]
- Androgen synthesis inhibitors: Drugs that directly inhibit the Serenoa repens).[92] A number of other antiandrogens, including cyproterone acetate, spironolactone, medrogestone, flutamide, nilutamide, and bifluranol, are also known to weakly inhibit androgen synthesis.
- Antigonadotropins: Drugs that suppress the
- Miscellaneous: Drugs that oppose the effects of androgens by means other than the above. Examples include estrogens, especially precursor androstenedione (used only in veterinary medicine).
Certain antiandrogens combine multiple of the above mechanisms.[6][97] An example is the steroidal antiandrogen cyproterone acetate, which is a potent AR antagonist, a potent progestogen and hence antigonadotropin, a weak glucocorticoid and hence anticorticotropin, and a weak androgen synthesis inhibitor.[6][97][98][99]
| Generic name | Class | Type | Brand name(s) | Route(s) | Launch | Status | Hitsa |
|---|---|---|---|---|---|---|---|
| Abiraterone acetate | Steroidal | Androgen synthesis inhibitor | Zytiga | Oral | 2011 | Available | 523,000 |
| Allylestrenol | Steroidal | Progestin | Gestanin, Perselin | Oral | 1961 | Availableb | 61,800 |
| Aminoglutethimide | Nonsteroidal | Androgen synthesis inhibitor | Cytadren, Orimeten | Oral | 1960 | Availableb | 222,000 |
| Apalutamide | Nonsteroidal | AR antagonist | Erleada | Oral | 2018 | Available | 50,400 |
| Bicalutamide | Nonsteroidal | AR antagonist | Casodex | Oral | 1995 | Available | 754,000 |
| Chlormadinone acetate | Steroidal | Progestin; AR antagonist | Belara, Prostal | Oral | 1965 | Available | 220,000 |
| Cyproterone acetate | Steroidal | Progestin; AR antagonist | Androcur, Diane | Oral, IM | 1973 | Available | 461,000 |
| Delmadinone acetate | Steroidal | Progestin; AR antagonist | Tardak | Veterinary | 1972 | Veterinary | 42,600 |
| Enzalutamide | Nonsteroidal | AR antagonist | Xtandi | Oral | 2012 | Available | 328,000 |
| Flutamide | Nonsteroidal | AR antagonist | Eulexin | Oral | 1983 | Available | 712,000 |
| Gestonorone caproate | Steroidal | Progestin | Depostat, Primostat | IM | 1973 | Availableb | 119,000 |
| Hydroxyprogesterone caproate | Steroidal | Progestin | Delalutin, Proluton | IM | 1954 | Available | 108,000 |
| Ketoconazole | Nonsteroidal | Androgen synthesis inhibitor | Nizoral, others | Oral, topical | 1981 | Available | 3,650,000 |
| Medroxyprogesterone acetate | Steroidal | Progestin | Provera, Depo-Provera | Oral, IM, SC | 1958 | Available | 1,250,000 |
| Megestrol acetate | Steroidal | Progestin; AR antagonist | Megace | Oral | 1963 | Available | 253,000 |
| Nilutamide | Nonsteroidal | AR antagonist | Anandron, Nilandron | Oral | 1987 | Available | 132,000 |
| Osaterone acetate | Steroidal | Progestin; AR antagonist | Ypozane | Veterinary | 2007 | Veterinary | 87,600 |
| Oxendolone | Steroidal | Progestin; AR antagonist | Prostetin, Roxenone | IM | 1981 | Availableb | 36,100 |
| Spironolactone | Steroidal | AR antagonist | Aldactone | Oral, topical | 1959 | Available | 3,010,000 |
| Topilutamide | Nonsteroidal | AR antagonist | Eucapil | Topical | 2003 | Availableb | 36,300 |
| Footnotes: a = Hits = Google Search hits (as of February 2018). b = Availability limited / mostly discontinued. Class: Steroidal = Steroidal antiandrogen. Nonsteroidal = Nonsteroidal antiandrogen. Sources: See individual articles. | |||||||
Side effects
The side effects of antiandrogens vary depending on the type of antiandrogen – namely whether it is a selective AR antagonist or lowers androgen levels – as well as the presence of
In males, the major
A number of antiandrogens have been associated with hepatotoxicity.[115] These include, to varying extents, cyproterone acetate, flutamide, nilutamide, bicalutamide, aminoglutethimide, and ketoconazole.[115] In contrast, spironolactone, enzalutamide,[116] and other antiandrogens are not associated with significant rates of hepatotoxicity. However, although they do not pose a risk of hepatotoxicity, spironolactone has a risk of hyperkalemia and enzalutamide has a risk of seizures.[citation needed]
In women who are
Overdose
Antiandrogens are relatively safe in acute
Interactions
Inhibitors and inducers of cytochrome P450 enzymes may interact with various antiandrogens.[citation needed]
Mechanism of action
Androgen receptor antagonists
| Antiandrogen | Relative binding affinities | ||||
|---|---|---|---|---|---|
| AR | PR | ER | GR | MR | |
| Cyproterone acetate | 8–10 | 60 | <0.1 | 5 | 1 |
| Chlormadinone acetate | 5 | 175 | <0.1 | 38 | 1 |
| Megestrol acetate | 5 | 152 | <0.1 | 50 | 3 |
| Spironolactone | 7 | 0.4a | <0.1 | 2a | 182 |
| Trimethyltrienolone | 3.6 | <1 | <1 | <1 | <1 |
| Inocoterone | 0.8 | <0.1 | <0.1 | <0.1 | <0.1 |
| Inocoterone acetate | <0.1 | <0.1 | <0.1 | <0.1 | <0.1 |
| Flutamide | <0.1 | <0.1 | <0.1 | <0.1 | <0.1 |
| Hydroxyflutamide | 0.5–0.8 | <0.1 | <0.1 | <0.1 | <0.1 |
| Nilutamide | 0.5–0.8 | <0.1 | <0.1 | <0.1 | <0.1 |
| Bicalutamide | 1.8 | <0.1 | <0.1 | <0.1 | <0.1 |
| Notes: (1): Reference ligands (100%) were testosterone for the AR, progesterone for the PR, estradiol for the ER, dexamethasone for the GR, and aldosterone for the MR. (2): Tissues were rat prostate (AR), rabbit uterus (PR), mouse uterus (ER), rat thymus (GR), and rat kidney (MR). (3): Incubation times (0 °C) were 24 hours (AR, a), 2 hours (PR, ER), 4 hours (GR), and 1 hour (MR). (4): Assay methods were different for bicalutamide for receptors besides the AR. Sources: [119][120][121][122][123][124][125][126][127][128] | |||||
| Antiandrogen | Relative potency |
|---|---|
| Bicalutamide | 4.3 |
| Hydroxyflutamide | 3.5 |
| Flutamide | 3.3 |
| Cyproterone acetate | 1.0 |
| Zanoterone | 0.4 |
| Description: Relative potencies of male rats. Higher values mean greater potency. Sources: See template. | |
AR antagonists act by directly binding to and competitively displacing androgens like testosterone and DHT from the AR, thereby preventing them from activating the receptor and mediating their biological effects.[86][87] AR antagonists are classified into two types, based on chemical structure: steroidal and nonsteroidal.[7][8][86][87][94] Steroidal AR antagonists are structurally related to steroid hormones like testosterone and progesterone, whereas nonsteroidal AR antagonists are not steroids and are structurally distinct. Steroidal AR antagonists tend to have off-target hormonal actions due to their structural similarity to other steroid hormones.[94] In contrast, nonsteroidal AR antagonists are selective for the AR and have no off-target hormonal activity.[94] For this reason, they are sometimes described as "pure" antiandrogens.[94]
Although they are described as antiandrogens and indeed show only such effects generally, most or all steroidal AR antagonists are actually not
Nonsteroidal antiandrogens have relatively low
AR antagonists may not bind to or block membrane androgen receptors (mARs), which are distinct from the classical nuclear AR.[139][140][141] However, the mARs do not appear to be involved in masculinization. This is evidenced by the perfectly female phenotype of women with complete androgen insensitivity syndrome.[142][143] These women have a 46,XY karyotype (i.e., are genetically "male") and high levels of androgens but possess a defective AR and for this reason never masculinize.[142][143] They are described as highly feminine, both physically as well as mentally and behaviorally.[144][145][146]
N-Terminal domain antagonists
Androgen receptor degraders
Androgen synthesis inhibitors
Androgen synthesis inhibitors are
5α-Reductase inhibitors
5α-Reductase inhibitors such as finasteride and dutasteride are inhibitors of
Antigonadotropins
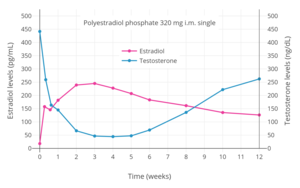

Examples of GnRH agonists include
Miscellaneous
Sex hormone-binding globulin modulators
In addition to their antigonadotropic effects, estrogens are also functional antiandrogens by decreasing free concentrations of androgens via increasing the
Corticosteroid-binding globulin modulators
Anticorticotropins
Anticorticotropins such as glucocorticoids and mineralocorticoids work by exerting negative feedback on the hypothalamic–pituitary–adrenal axis (HPA axis), thereby inhibiting the secretion of corticotropin-releasing hormone (CRH) and hence adrenocorticotropic hormone (ACTH; corticotropin) and consequently suppressing the production of androgen prohormones like dehydroepiandrosterone (DHEA), dehydroepiandrosterone sulfate (DHEA-S), and androstenedione in the adrenal gland.[184][185] They are rarely used clinically as functional antiandrogens, but are used as such in the case of congenital adrenal hyperplasia in girls and women, in which there are excessive production and levels of adrenal androgens due to glucocorticoid deficiency and hence HPA axis overactivity.[184][185]
Insulin sensitizers
In women with
Immunogens and vaccines
Chemistry
Antiandrogens can be divided into several different types based on
]History
Antigonadotropins like estrogens and progestogens were both first introduced in the 1930s.[191] The beneficial effects of androgen deprivation via surgical castration or high-dose estrogen therapy on prostate cancer were discovered in 1941.[31]: 56 [192] AR antagonists were first discovered in the early 1960s.[98] The steroidal antiandrogen cyproterone acetate was discovered in 1961 and introduced in 1973 and is often described as the first antiandrogen to have been marketed.[193][62] However, spironolactone was introduced in 1959,[194][195] although its antiandrogen effects were not recognized or taken advantage of until later and were originally an unintended off-target action of the drug.[196] In addition to spironolactone, chlormadinone acetate and megestrol acetate are steroidal antiandrogens that are weaker than cyproterone acetate but were also introduced earlier, in the 1960s.[197][198][199] Other early steroidal antiandrogens that were developed around this time but were never marketed include benorterone (SKF-7690; 17α-methyl-B-nortestosterone), BOMT (Ro 7–2340), cyproterone (SH-80881), and trimethyltrienolone (R-2956).[200][201]
The nonsteroidal antiandrogen flutamide was first reported in 1967.[24] It was introduced in 1983 and was the first nonsteroidal antiandrogen marketed.[202][203] Another early nonsteroidal antiandrogen,[204] DIMP (Ro 7–8117), which is structurally related to thalidomide[205] and is a relatively weak antiandrogen,[206][207] was first described in 1973 and was never marketed.[208] Flutamide was followed by nilutamide in 1989 and bicalutamide in 1995.[209] In addition to these three drugs, which have been regarded as first-generation nonsteroidal antiandrogens, the second-generation nonsteroidal antiandrogens enzalutamide and apalutamide were introduced in 2012 and 2018, respectively.[210][211][212] They differ from the earlier nonsteroidal antiandrogens namely in that they are much more efficacious in comparison.[211]
The androgen synthesis inhibitors aminoglutethimide and ketoconazole were first marketed in 1960 and 1977, respectively,[213][214] and the newer drug abiraterone acetate was introduced in 2011.[215] GnRH modulators were first introduced in the 1980s.[216] The 5α-reductase inhibitors finasteride and dutasteride were introduced in 1992 and 2002, respectively.[217][218] Elagolix, the first orally active GnRH modulator to be marketed, was introduced in 2018.[219]
Timeline
The following is a timeline of events in the history of antiandrogens:[220]
- 1941: Hudgins and Hodges show that androgen deprivation via high-dose estrogen therapy or surgical castration treats prostate cancer
- 1957: The steroidal antiandrogen spironolactone is first synthesized[221]
- 1960: Spironolactone is first introduced for medical use, as an antimineralocorticoid[221]
- 1961: The steroidal antiandrogen cyproterone acetate is first synthesized[222]
- 1962: Spironolactone is first reported to produce gynecomastia in men[221][223]
- 1966: Benorterone is the first known antiandrogen to be studied clinically, to treat acne and hirsutism in women[224][225]
- 1963: The antiandrogenic activity of cyproterone acetate is discovered[48][226]
- 1967: A known antiandrogen, benorterone, is first reported to induce gynecomastia in males[224]
- 1967: The first-generation nonsteroidal antiandrogen flutamide is first synthesized
- 1967: Cyproterone acetate was first studied clinically, to treat sexual deviance in men[227]
- 1969: Cyproterone acetate was first studied in the treatment of acne, hirsutism, seborrhea, and scalp hair loss in women[228]
- 1969: The antiandrogenic activity of spironolactone is discovered[229]
- 1972: The antiandrogenic activity of flutamide is first reported[230][231]
- 1973: Cyproterone acetate was first introduced for medical use, to treat sexual deviance[232]
- 1977: The first-generation antiandrogen nilutamide is first described[233]
- 1978: Spironolactone is first studied in the treatment of hirsutism in women[65][234]
- 1979: Combined androgen blockade is first studied[235][236]
- 1980: Medical castration via a GnRH analogue is first achieved[citation needed]
- 1982: The first-generation antiandrogen bicalutamide is first described[237]
- 1982: Combined androgen blockade for prostate cancer is developed
- 1983: Flutamide is first introduced, in Chile, for medical use, to treat prostate cancer[238][239]
- 1987: Nilutamide is first introduced, in France, for medical use, to treat prostate cancer[209]
- 1989: Combined androgen blockade via flutamide and a GnRH analogue is found to be superior to a GnRH analogue alone for prostate cancer
- 1989: Flutamide is first introduced for medical use in the United States, to treat prostate cancer[240]
- 1989: Flutamide is first studied in the treatment of hirsutism in women[9]
- 1992: The androgen synthesis inhibitor abiraterone acetate is first described[241]
- 1995: Bicalutamide is first introduced for medical use, to treat prostate cancer[209]
- 1996: Nilutamide is first introduced for medical use in the United States, to treat prostate cancer[242]
- 2006: The second-generation nonsteroidal antiandrogen enzalutamide is first described[243]
- 2007: The second-generation nonsteroidal antiandrogen apalutamide is first described[244]
- 2011: Abiraterone acetate is first introduced for medical use, to treat prostate cancer[245]
- 2012: Enzalutamide is first introduced for medical use, to treat prostate cancer[246]
- 2018: Apalutamide is first introduced for medical use, to treat prostate cancer[247]
- 2018: Elagolix is the first orally active GnRH antagonist to be introduced for medical use[219]
- 2019: Relugolix is the second orally active GnRH antagonist to be introduced for medical use[248]
Society and culture
Etymology
The term antiandrogen is generally used to refer specifically to AR antagonists, as described by Dorfman (1970):[249][250]
Antiandrogens are substances which prevent androgens from expressing their activity at target sites. The inhibitory effect of these substances, therefore, should be differentiated from compounds which decrease the synthesis and/or release of hypothalamic (releasing) factors, from anterior pituitary hormones (gonadotropins, particularly luteinizing hormone) and from material which acts directly on the gonads to inhibit biosynthesis and/or secretion of androgens.[249][250]
However, in spite of the above, the term may also be used to describe functional antiandrogens like androgen synthesis inhibitors and antigonadotropins, including even estrogens and progestogens.[2][6][251] For example, the progestogen and hence antigonadotropin medroxyprogesterone acetate is sometimes described as a steroidal antiandrogen, even though it is not an antagonist of the AR.[252][251]
Research
Topical administration
This section's factual accuracy may be compromised due to out-of-date information. The reason given is: Clascoterone has been approved for acne by the FDA in August 2020, and is considered to be highly effective (>= tretinoin 0.05%). (April 2024) |
There has been much interest and effort in the development of topical AR antagonists to treat androgen-dependent conditions like acne and pattern hair loss in males.
A variety of AR antagonists have been developed for topical use but have not completed development and hence have never been marketed. These include the steroidal AR antagonists clascoterone, cyproterone, rosterolone, and topterone and the nonsteroidal AR antagonists cioteronel, inocoterone acetate, RU-22930, RU-58642, and RU-58841. However, one topical AR antagonist, topilutamide (fluridil), has been introduced in a few European countries for the treatment of pattern hair loss in men.[40] In addition, a topical 5α-reductase inhibitor and weak estrogen, alfatradiol, has also been introduced in some European countries for the same indication, although its effectiveness is controversial.[40] Spironolactone has been marketed in Italy in the form of a topical cream under the brand name Spiroderm for the treatment of acne and hirsutism, but this formulation was discontinued and hence is no longer available.[254]
Male contraception
Antiandrogens, such as cyproterone acetate, have been studied for potential use as
Breast cancer
Antiandrogens such as bicalutamide, enzalutamide, and abiraterone acetate are under investigation for the potential treatment of breast cancer, including AR-expressing triple-negative breast cancer and other types of AR-expressing breast cancer.[267][268][269][270][271]
Miscellaneous
Antiandrogens may be effective in the treatment of obsessive–compulsive disorder.[272]
See also
References
- PMID 2530930.
- ^ ISBN 3527600906.
- ISBN 978-0-7817-4868-1.
- ^ PMID 31712062.
- S2CID 23888640.
- ^ PMID 11502457.
There are several classes of antiandrogens including (1) antigonadotropins (eg, LHRH agonists/antagonists, synthetic estrogens [diethylstilbestrol]); (2) nonsteroidal androgen-receptor antagonists (eg, flutamide, bicalutamide, nilutamide); (3) steroidal agents with mixed actions (eg, cyproterone acetate); (4) adrenal androgen inhibitors (eg, ketoconazole, hydrocortisone); (5) steroidal agents that inhibit androgen biosynthesis (eg, 5α-reductase inhibitors (type II) and dual-acting 5α-reductase inhibitors); [...]
- ^ ISBN 978-1-60761-471-5.
- ^ ISBN 978-1-60761-471-5.
- ^ S2CID 20274398.
- PMID 20092449.
- ISBN 978-1-139-48080-2.
- ^ PMID 16828411.
- ^ ISSN 0951-3590.
- ^ PMID 21714669.
- ^ (PDF) from the original on 2018-05-15. Retrieved 2018-05-14.
- ^ PMID 27019626.
- ^ PMID 21680543.
- S2CID 41988320.
- ^ S2CID 38486547.
- S2CID 13980764.
- ^ S2CID 28215804.
- PMID 11502439.
- PMID 12667881.
- ^ ISBN 978-0-203-30415-0.
- ISBN 978-1-60547-431-1. Archivedfrom the original on 10 January 2023. Retrieved 27 December 2016.
From a structural standpoint, antiandrogens are classified as steroidal, including cyproterone [acetate] (Androcur) and megestrol [acetate], or nonsteroidal, including flutamide (Eulexin, others), bicalutamide (Casodex), and nilutamide (Nilandron). The steroidal antiandrogens are rarely used.
- (PDF) from the original on 2017-05-10. Retrieved 2016-12-27.
- ^ Chang S (10 March 2010), Bicalutamide BPCA Drug Use Review in the Pediatric Population (PDF), U.S. Department of Health and Human Service, archived (PDF) from the original on 24 October 2016, retrieved 20 July 2016
- ISBN 978-1-935281-91-7.
- ISBN 978-3-8055-8524-8. Archivedfrom the original on 12 January 2023. Retrieved 27 December 2016.
- ISBN 978-1-935281-91-7.
- ^ ISBN 978-1-60327-829-4.
- ^ PMID 21604953.
- PMID 27735065.
- PMID 19509099.
- S2CID 1445627.
- PMID 24032068.
- ^ PMID 19030020.
- S2CID 8249363.
- ISBN 978-1-4831-8711-2.
- ^ ISBN 978-3-319-03233-7.
- ^ ISBN 9789997638991.
- ^ ISSN 0171-2004.
- ^ ISBN 978-0-8247-9363-0.
- ^ ISSN 0065-7743.
- ^ S2CID 12153042.
- PMID 19297634.
- ISBN 978-3-642-20112-7.
- ^ ISBN 978-3-642-45747-0.
- PMID 6376557.
- ISBN 978-3-642-59715-2.
- ISBN 978-1-60913-713-7.
- PMID 6235105.
- PMID 2945742.
- from the original on 2020-10-26. Retrieved 2020-03-30.
- PMID 27416311.
- ^ "Cassiopea Receives FDA Approval for Winlevi® (clascoterone cream 1%), First-in-Class Topical Acne Treatment Targeting the Androgen Receptor - Cassiopea". web.archive.org. 2020-08-28. Retrieved 2024-04-15.
- ^ "Drugs@FDA: FDA-Approved Drugs". www.accessdata.fda.gov. Retrieved 2024-04-15.
- ISBN 978-0-12-382185-0.
- ^ PMID 11221487.
- ^ PMID 19243704.
- ISBN 978-1-4899-0915-2.
- ^ ISBN 978-0-8058-0280-1.
- ISBN 978-1-4939-2416-5.
- ^ PMID 18345393.
- ^ PMID 6205409.
- PMID 2462132.
- ^ PMID 3109366.
- S2CID 25398066.
- PMID 1838080.
- ^ PMID 1903104.
- ^ PMID 16361981.
- PMID 22057380.
- PMID 20092449.
- S2CID 35399393.
- PMID 11796309.
- PMID 18087648.
- ISBN 978-1-85317-478-0.
- ISBN 978-1-60795-039-4.
- ISBN 978-1-4443-0017-8. Archivedfrom the original on 10 January 2023. Retrieved 27 December 2016.
- ^ S2CID 39120534.
- ^ ISBN 978-0-7817-1750-2.
- ^ ISBN 978-1-4511-1091-3.
- S2CID 52821192.
- PMID 33251587.
- PMID 33644622.
- ^ PMID 10637363.
- ^ ISBN 978-1-60327-831-7.
- S2CID 51622914.
- S2CID 28436828.
- S2CID 8400556.
- ^ ISBN 978-1-4557-2758-2.
- S2CID 44363501.
- ^ ISBN 978-1-4684-7313-1.
- ^ ISBN 978-1-60913-345-0.
- ^ S2CID 40392817.
- ISBN 978-1-316-21414-5.
- ^ ISBN 978-1-107-68362-4.
- ^ ISBN 978-3-319-13278-5.
- S2CID 25200595.
- ^ ISBN 978-1-4398-1048-4. Archivedfrom the original on 11 January 2023. Retrieved 27 December 2016.
- S2CID 8639102.
- ISBN 978-0-85729-727-3.
- ^ PMID 12667885.
- PMID 16321765.
- ISBN 978-1-4557-7169-1.
- ISBN 978-1-4613-8289-8.
- PMID 20082945.
- S2CID 205631980.
- PMID 19843067.
- PMID 33334002.
- PMID 29211888.
- PMID 36238954.
- PMID 3318361.
- PMID 30586774.
- ^ S2CID 24799765.
- S2CID 29563345.
- ^ ISBN 978-0-7817-3790-6.
- ISBN 978-0-205-40615-9. Archivedfrom the original on 2023-02-26. Retrieved 2016-12-27.
- PMID 3059062.
- PMID 1992602.
- PMID 3009970.
- S2CID 31404295.
- ISBN 978-0-88167-161-2.
- ISBN 978-1-349-81347-6.
- PMID 7421203.
- from the original on 2020-11-27. Retrieved 2021-10-31.
- PMID 171505.
- PMID 14600402.
- ^ S2CID 24746807.
- PMID 2462135.
- PMID 22665559.
- S2CID 38441469.
- ISBN 978-1-4831-9067-9.
- ^ PMID 9000189.
Because steroidal antiandrogens such as cyproterone acetate have intrinsic androgenic activity and lower antiandrogenic activity than the NSAAs such as flutamide and nilutamide,39–43 it is not surprising that the two classes of antiandrogens may have different efficacies.
- ^ PMID 10637363.
- ^ PMID 2788775.
- ^ PMID 14751673.
- ISBN 978-0-19-506739-2.
In patients receiving flutamide at the usual dosage of 250 mg every 8 hours, the minimal plasma concentration of hydroxyflutamide is about 5 uM, which is 5,000 times the plasma concentration of testosterone (1 nM) in patients treated with an LHRH agonist.127 As hydroxyflutamide is only one percent as potent as testosterone in competing for binding to the androgen receptor,126 a plasma level of 5 uM hydroxyflutamide is required to ensure effective competition.127 [...] Both cyproterone acetate and flutamide have been demonstrated to be effective therapy (roughly equivalent to an estrogen) when used alone in the treatment of carcinoma of the prostate.123
- PMID 19931639.
- PMID 25257522.
- S2CID 23918273.
- ^ ISBN 978-0-7817-4059-3.
- ^ ISBN 978-88-470-1405-3.
- ISBN 978-0-674-05879-8.
- ISBN 978-1-135-07932-1.
- ISBN 978-0-470-65760-7.
- ^ PMID 27302572.
- PMID 27356095.
- PMID 26389532.
- ^ PMID 27885283.
- ^ PMID 23219429.
- ^ "ASCJ 9". AdisInsight. Springer Nature Switzerland AG. Archived from the original on 2018-03-04. Retrieved 2017-12-24.
- PMID 2652864.
- ^ ISBN 978-0-7637-3075-8.
- PMID 27154414.
- PMID 12570838.
- ISBN 978-1-61779-222-9.
- ^ ISBN 978-0-12-095440-7.
- PMID 27672412.
- S2CID 33548251.
- ISSN 1561-0950.
- ISBN 978-1-903737-03-3. Archivedfrom the original on 2023-01-11. Retrieved 2016-12-27.
- PMID 368741.
- PMID 7000222.
- ISBN 978-1-4160-6911-9. Archivedfrom the original on 11 January 2023. Retrieved 27 December 2016.
- S2CID 5836155.
- PMID 18268497.
- PMID 6258683.
- PMID 694436.
- ISBN 978-0-306-46278-8.
- PMID 294107.
- ^ ISBN 978-1-107-01290-5.
- ^ ISBN 978-92-832-1291-1.
- PMID 22294742.
- ^ ISBN 978-81-250-1793-6.
- ISBN 978-1-4831-6366-6.
- ^ ISSN 0171-2004.
- ^ ISBN 978-1-59259-101-5.
- ^ S2CID 6815423.
- PMID 2958420.
- ^ S2CID 25309877.
- ^ PMID 7500443.
- S2CID 21510744.
- ^ ISBN 978-1-4377-3600-7.
- ^ ISBN 978-1-4377-2015-0.
- ^ S2CID 149443722.
- S2CID 4391302.
- PMID 33014044.
- ^ ISBN 978-94-009-5038-2.
- ^ ISBN 978-93-80235-35-6.
- ISBN 978-1-4511-4847-3.
- ISBN 978-1-4471-4404-5.
- ISBN 978-0-08-052628-7.
- ISBN 978-1-4939-0268-2. Archivedfrom the original on 11 January 2023. Retrieved 27 December 2016.
- ISBN 978-0-08-056877-5.
- ISBN 978-1-59745-179-6.
- ISBN 978-3-642-73790-9.
- ISBN 978-3-642-81650-5.
- ISBN 978-0-300-16791-7.
- ISBN 978-94-009-8195-9.
- ISBN 978-0-08-086630-7.
- ISBN 978-1-4471-2086-5.
- ISBN 978-3-527-62330-3.
- ISBN 978-0-8391-0776-7.
- S2CID 23228778.
- PMID 180344.
- ISBN 978-0-08-058365-5.
- PMID 4739363.
- ^ ISBN 978-0-470-28187-1. Archivedfrom the original on 12 January 2023. Retrieved 27 December 2016.
- S2CID 8725297.
- ^ PMID 19359544.
- ^ "FDA approves new treatment for a certain type of prostate cancer using novel clinical trial endpoint". Food and Drug Administration. 24 March 2020. Archived from the original on 23 April 2019. Retrieved 1 April 2018.
- ISBN 978-0-471-89979-2.
- ISBN 978-0-7817-8355-2. Archivedfrom the original on 2023-01-10. Retrieved 2016-12-27.
- ISBN 978-1-935281-91-7.
- ISBN 978-1-4051-7177-9.
- ISBN 978-1-904798-74-3.
- ISBN 978-1-60805-208-0.
- ^ a b "Elagolix - Abbvie/Neurocrine Biosciences". AdisInsight. Springer Nature Switzerland AG. Archived from the original on 2018-09-20. Retrieved 2018-07-30.
- S2CID 19162538.
- ^ PMID 28634268.
- PMID 9476091.
- ISSN 0140-6736.
- ^ S2CID 43649686.
- PMID 4225258.
- PMID 14071315.
- S2CID 14949511.
- S2CID 71214286.
- PMID 5344274.
- PMID 4264731.
- PMID 4116667.
- PMID 8005205.
- PMID 385986.
- PMID 717935.
- PMID 16631454.
- PMID 6819101.
- ISBN 978-3-13-179275-4.
- ISBN 978-0-8155-1856-3. Archivedfrom the original on 12 January 2023. Retrieved 2 January 2019.
- ^ The Irish Reports: Containing Reports of Cases Argued and Determined in the Court of Appeal, the High Court of Justice, the Court of Bankruptcy, in Ireland, and the Irish Land Commission. Incorporated Council of Law Reporting for Ireland. 1990. pp. 501–502. Archived from the original on 2023-01-12. Retrieved 2019-01-02.
- ISBN 978-3-642-30725-6.
- ^ "Abiraterone: A story of scientific innovation and commercial partnership - the Institute of Cancer Research, London". Archived from the original on 2019-01-01. Retrieved 2019-01-02.
- ^ "Nilutamide". AdisInsight. Springer Nature Switzerland AG. Archived from the original on 2021-05-05. Retrieved 2019-01-02.
- ^ Sawyers, C., Jung, M., Chen, C., Ouk, S., Welsbie, D., Tran, C., ... & Yoo, D. (2006). U.S. Patent Application No. 11/433,829. https://www.google.com/patents/US20070004753 Archived 2016-10-05 at the Wayback Machine
- ^ "Espacenet - Original document". Archived from the original on 2021-11-04. Retrieved 2019-01-02.
- ^ "Abiraterone acetate - Johnson & Johnson". AdisInsight. Springer Nature Switzerland AG. Archived from the original on 2019-01-03. Retrieved 2019-01-02.
- ^ "Enzalutamide - Astellas Pharma/Medivation". AdisInsight. Springer Nature Switzerland AG. Archived from the original on 2018-07-17. Retrieved 2019-01-02.
- ^ "Apalutamide - Janssen Research and Development". AdisInsight. Springer Nature Switzerland AG. Archived from the original on 2019-01-03. Retrieved 2019-01-02.
- ^ "Relugolix - Myovant/Takeda". AdisInsight. Springer Nature Switzerland AG. Archived from the original on 2021-08-19. Retrieved 2021-08-19.
- ^ S2CID 7224893.
- ^ S2CID 71393789.
- ^ ISBN 978-3-642-80861-6.
- ISBN 978-0-7817-4984-8.
- ^ ISBN 978-0-7817-5734-8.
- ISBN 978-0-387-09718-3. Archivedfrom the original on 11 January 2023. Retrieved 30 December 2016.
- S2CID 24859886.
- PMID 797248.
- PMID 206192.
- PMID 6354690.
- ^ PMID 2687939.
- ^ S2CID 36608203.
- ^ S2CID 22458746.
- ^ (PDF) from the original on 2020-12-05. Retrieved 2019-07-08.
- PMID 16313066.
- PMID 27016468.
- PMID 16497801.
- PMID 27907978.
- PMID 24740738.
- PMID 27816190.
- PMID 24888812.
- PMID 28216075.
- PMID 29940524.
- S2CID 208956450.
Further reading
- Neumann F, Steinbeck H (1974). "Antiandrogens". Androgens II and Antiandrogens / Androgene II und Antiandrogene. pp. 235–484. ISBN 978-3-642-80861-6.
- Gräf KJ, Brotherton J, Neumann F (1974). "Clinical Uses of Antiandrogens". Androgens II and Antiandrogens / Androgene II und Antiandrogene. pp. 485–542. ISBN 978-3-642-80861-6.
