Antigenic variation
Antigenic variation or antigenic alteration refers to the mechanism by which an
In bacteria
Antigenic variation in bacteria is best demonstrated by species of the genus Neisseria (most notably, Neisseria meningitidis and Neisseria gonorrhoeae, the gonococcus); species of the genus Streptococcus and the Mycoplasma. The Neisseria species vary their pili (protein polymers made up of subunits called pilin which play a critical role in bacterial adhesion, and stimulate a vigorous host immune response) and the Streptococci vary their M-protein.
In the bacterium Borrelia burgdorferi, the cause of Lyme disease, the surface lipoprotein VlsE can undergo recombination which results in antigenic diversity. The bacterium carries a plasmid that contains fifteen silent vls cassettes and one functional copy of vlsE. Segments of the silent cassettes recombine with the vlsE gene, generating variants of the surface lipoprotein antigen.[7]
In protozoa
Antigenic variation is employed by a number of different
Trypanosoma brucei
Trypanosoma brucei, the organism that causes
replicates extracellularly in the bloodstream of infected mammals and is subjected to numerous host defense mechanisms including the complement system, and the innate and adaptive immune systems. To protect itself, the parasite decorates itself with a dense, homogeneous coat (~10^7 molecules) of the variant surface glycoprotein
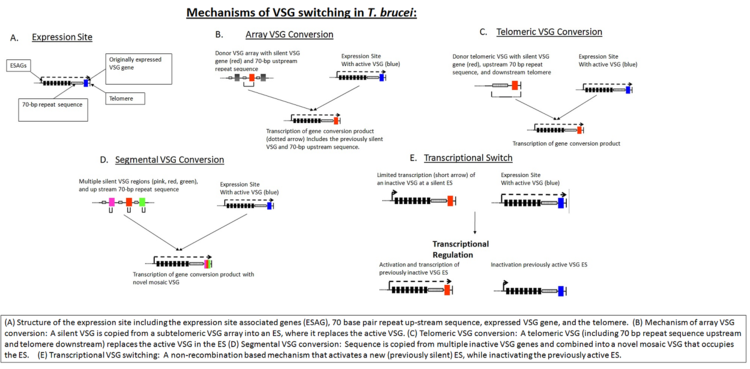
In the early stages of invasion, the VSG coat is sufficient to protect the parasite from immune detection. The host eventually identifies the VSG as a foreign antigen and mounts an attack against the microbe. However, the parasite's genome has over 1,000 genes that code for different variants of the VSG protein, located on the subtelomeric portion of large chromosomes, or on intermediate chromosomes. These VSG genes become activated by gene conversion in a hierarchical order: telomeric VSGs are activated first, followed by array VSGs, and finally pseudogene VSGs.[8] Only one VSG is expressed at any given time. Each new gene is switched in turn into a VSG expression site (ES).[9] This process is partially dependent on homologous recombination of DNA, which is mediated in part by the interaction of the T. brucei BRCA2 gene with RAD51 (however, this is not the only possible mechanism, as BRCA2 variants still display some VSG switching).[9]
In addition to homologous recombination, transcriptional regulation is also important in antigen switching, since T. brucei has multiple potential expression sites. A new VSG can either be selected by transcriptional activation of a previously silent ES, or by recombination of a VSG sequence into the active ES (see figure, "Mechanisms of VSG Switching in T. brucei").[8] Although the biological triggers that result in VSG switching are not fully known, mathematical modeling suggests that the ordered appearance of different VSG variants is controlled by at least two key parasite-derived factors: differential activation rates of parasite VSG and density-dependent parasite differentiation.[10][11]
Plasmodium falciparum
Plasmodium falciparum, the major etiologic agent of human malaria, has a very complex
In viruses
Different virus families have different levels of ability to alter their genomes and trick the immune system into not recognizing. Some viruses have relatively unchanging genomes like
- Antigenic drift: point mutations that occur through imperfect replication of the viral genome. All viruses exhibit genetic drift over time but the amount that they are able to drift without incurring a negative impact on their fitness varies between families.
- Antigenic shift: reassortment of the viral genome that occurs when a single host cell is co-infected with two unique virus particles. As the viruses replicate, they reassort and the genes of the two species get mixed up when packaged into a new budding virus. For influenza, this process could yield up to 256 new variations of the virus, and meaningful antigenic shift events tend to occur every couple of decades.
- Antigenic rift: Recombination of viral gene. This occurs when there are again two viral cells that infect the same host cell. In this instance the viruses recombine with pieces of each gene creating a new gene instead of simply switching out genes. Recombination has been extensively studied in avian influenza strains as to how the genetics of H5N1 have changed over time.[17]
- Antigenic sift: direct transmission with a zoonotic strain of a virus. This occurs when a human is infected during a spillover event.
- Antigenic lift: Viral transmission of host derived gene. Some viruses steal host genes and then incorporate them into their own viral genome, encoding genes that sometimes give them an increased virulence. An example of this is the pox virus vaccinia which encoded a viral growth factor that is very similar to the human growth factor and thought to be stolen from the human genome.[18]
- Antigenic gift: Occurs when humans deliberately modify a virus's genome either in a lab setting or in order to make a bioweapon.
Influenza virus
The antigenic properties of
Vaccination against influenza
After vaccination, IgG+ antibody-secreting plasma cells (ASCs) increase rapidly and reaches a maximum level at day 7 before returning to a minimum level at day 14. The influenza-specific memory B-cells reach their maxima at day 14–21. The secreted antibodies are specific to the vaccine virus. Further, most of the monoclonal antibodies isolated have binding affinities against HA and the remaining demonstrate affinity against NA, nucleoprotein (NP) and other antigens. These high affinity human monoclonal antibodies can be produced within a month after vaccination and because of their human origin, they will have very little, if any, antibody-related side-effects in humans. They can potentially be used to develop passive antibody therapy against influenza virus transmission.
Mapping antigenic evolution
The ability of an antiviral antibody to inhibit hemagglutination can be measured and used to generate a two-dimensional map using a process called antigenic cartography so that antigenic evolution can be visualized. These maps can show how changes in amino acids can alter the binding of an antibody to virus particle and help to analyze the pattern of genetic and antigenic evolution. Recent findings show that as a result of antibody-driven antigenic variation in one domain of the H1 hemagglutinin Sa site, a compensatory mutation in NA can result leading to NA antigenic variation. As a consequence, drug resistance develops to NA inhibitors. Such a phenomenon can mask the evolution of NA evolution in nature because the resistance to NA inhibitors could be due to antibody-driven, HA escape.[20]
HIV-1
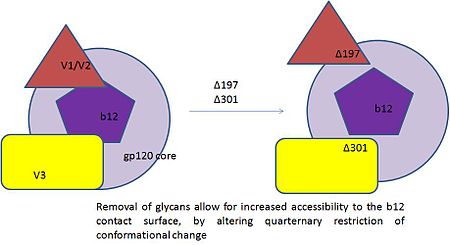
The major challenge in controlling
Flaviviruses
Flaviviridae is a family of viruses that encompasses well known viruses such as West Nile virus and Dengue virus. The genus Flavivirus has a prototypical envelope protein (E-protein) on its surface which serves as the target for virus neutralizing antibodies. E protein plays a role in binding to receptor and could play a role in evading the host immune system. It has three major antigenic domains namely A, B and C that correspond to the three structural domains II, III and I. Structural domain III is a putative receptor binding domain and antibodies against it neutralize the infectivity of flaviviruses. Mutations that lead to antigenic differences can be traced to the biochemical nature of the amino acid substitutions as well as the location of the mutation in the domain III. For example, substitutions at different amino acids results in varying levels of neutralization by antibodies. If mutation in a critical amino acid can dramatically alter neutralization by antibodies then WNV vaccines and diagnostic assays becomes difficult to rely on. Other flaviviruses that cause dengue, louping ill and yellow fever escape antibody neutralization via mutations in the domain III of the E protein.[23][24]
References
- S2CID 688880.
- PMID 11544233.
- PMID 8500868.
- PMID 9108482.
- S2CID 27337497.
- PMID 15258095.
- S2CID 25378695.
- ^ PMID 18666832.
- ^ PMID 18430140.
- PMID 17463092.
- S2CID 206881591.
- PMID 17644655.
- PMID 9736619.
- PMID 17257309.
- PMID 15797990.
- ^ S2CID 10305325.
- S2CID 4368679.
- PMID 8254780.
- PMID 19284639.
- PMID 21364978.
- PMID 18407775.
- PMID 19414790.
- S2CID 37056432.
- PMID 15823609.
