Chemotherapy
| Chemotherapy | |
|---|---|
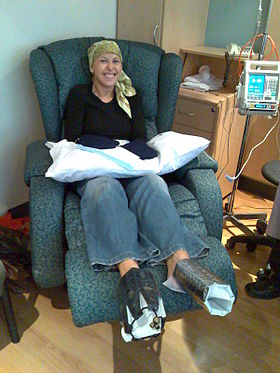 A woman being treated with docetaxel chemotherapy for breast cancer. Cold mittens and cold booties are placed on her hands and feet to reduce harm to her nails. | |
| Other names | chemo, CTX, CTx |
Chemotherapy (often abbreviated to chemo and sometimes CTX or CTx) is a type of
The term chemotherapy has come to connote non-specific usage of intracellular poisons to inhibit mitosis (cell division) or induce DNA damage, which is why inhibition of DNA repair can augment chemotherapy.[3] The connotation of the word chemotherapy excludes more selective agents that block extracellular signals (signal transduction). The development of therapies with specific molecular or genetic targets, which inhibit growth-promoting signals from classic endocrine hormones (primarily estrogens for breast cancer and androgens for prostate cancer) are now called hormonal therapies. By contrast, other inhibitions of growth-signals like those associated with receptor tyrosine kinases are referred to as targeted therapy.
Importantly, the use of drugs (whether chemotherapy, hormonal therapy or targeted therapy) constitutes systemic therapy for cancer in that they are introduced into the blood stream and are therefore in principle able to address cancer at any anatomic location in the body. Systemic therapy is often used in conjunction with other modalities that constitute local therapy (i.e., treatments whose efficacy is confined to the anatomic area where they are applied) for cancer such as radiation therapy, surgery or hyperthermia therapy.
Traditional chemotherapeutic agents are
Treatment strategies
| Cancer type | Drugs | Acronym |
|---|---|---|
| Breast cancer | 5-fluorouracil, vinorelbine |
CMF |
| Doxorubicin, cyclophosphamide | AC | |
Hodgkin's lymphoma
|
Docetaxel, doxorubicin, cyclophosphamide | TAC |
| Doxorubicin, bleomycin, vinblastine, dacarbazine | ABVD | |
| Mustine, vincristine, procarbazine, prednisolone | MOPP | |
Non-Hodgkin's lymphoma
|
Cyclophosphamide, doxorubicin, vincristine, prednisolone | CHOP, R-CVP |
| Germ cell tumor | Bleomycin, etoposide, cisplatin | BEP |
| Stomach cancer[5] | Epirubicin, cisplatin, 5-fluorouracil | ECF |
| Epirubicin, cisplatin, capecitabine | ECX | |
| Bladder cancer | Methotrexate, vincristine, doxorubicin, cisplatin | MVAC |
| Lung cancer | Cyclophosphamide, doxorubicin, vincristine, vinorelbine | CAV |
| Colorectal cancer | 5-fluorouracil, folinic acid, oxaliplatin |
FOLFOX |
| Pancreatic cancer | 5-fluorouracil |
FOLFOX |
Bone cancer
|
Doxorubicin, cisplatin, methotrexate, ifosfamide, etoposide | MAP/MAPIE |
There are a number of strategies in the administration of chemotherapeutic drugs used today. Chemotherapy may be given with a curative intent or it may aim to prolong life or to palliate symptoms.
- Induction chemotherapy is the first line treatment of cancer with a chemotherapeutic drug. This type of chemotherapy is used for curative intent.[1][6]: 55–59
- Combined modality chemotherapy is the use of drugs with other cancer treatments, such as surgery, radiation therapy, or hyperthermia therapy.
- Consolidation chemotherapy is given after remission in order to prolong the overall disease-free time and improve overall survival. The drug that is administered is the same as the drug that achieved remission.[6]: 55–59
- Intensification chemotherapy is identical to consolidation chemotherapy but a different drug than the induction chemotherapy is used.[6]: 55–59
- Combination chemotherapy involves treating a person with a number of different drugs simultaneously. The drugs differ in their mechanism and side-effects. The biggest advantage is minimising the chances of resistance developing to any one agent. Also, the drugs can often be used at lower doses, reducing toxicity.[6]: 55–59 [7]: 17–18 [5]
- Neoadjuvant chemotherapy is given prior to a local treatment such as surgery, and is designed to shrink the primary tumor.[6]: 55–59 It is also given for cancers with a high risk of micrometastatic disease.[8]: 42
- micrometastases can be treated with adjuvant chemotherapy and can reduce relapse rates caused by these disseminated cells.[9]
- Maintenance chemotherapy is a repeated low-dose treatment to prolong remission.[5][6]: 55–59
- Salvage chemotherapy or palliative chemotherapy is given without curative intent, but simply to decrease tumor load and increase life expectancy. For these regimens, in general, a better toxicity profile is expected.[6]: 55–59
All chemotherapy regimens require that the recipient be capable of undergoing the treatment. Performance status is often used as a measure to determine whether a person can receive chemotherapy, or whether dose reduction is required. Because only a fraction of the cells in a tumor die with each treatment (fractional kill), repeated doses must be administered to continue to reduce the size of the tumor.[10] Current chemotherapy regimens apply drug treatment in cycles, with the frequency and duration of treatments limited by toxicity.[11]
Effectiveness
The effectiveness of chemotherapy depends on the type of cancer and the stage. The overall effectiveness ranges from being curative for some cancers, such as some
Dosage

Dosage of chemotherapy can be difficult: If the dose is too low, it will be ineffective against the tumor, whereas, at excessive doses, the toxicity (side-effects) will be intolerable to the person receiving it.[4] The standard method of determining chemotherapy dosage is based on calculated body surface area (BSA). The BSA is usually calculated with a mathematical formula or a nomogram, using the recipient's weight and height, rather than by direct measurement of body area. This formula was originally derived in a 1916 study and attempted to translate medicinal doses established with laboratory animals to equivalent doses for humans.[16] The study only included nine human subjects.[17] When chemotherapy was introduced in the 1950s, the BSA formula was adopted as the official standard for chemotherapy dosing for lack of a better option.[18][19]
The validity of this method in calculating uniform doses has been questioned because the formula only takes into account the individual's weight and height. Drug absorption and clearance are influenced by multiple factors, including age, sex, metabolism, disease state, organ function, drug-to-drug interactions, genetics, and obesity, which have major impacts on the actual concentration of the drug in the person's bloodstream.[18][20][21] As a result, there is high variability in the systemic chemotherapy drug concentration in people dosed by BSA, and this variability has been demonstrated to be more than ten-fold for many drugs.[17][22] In other words, if two people receive the same dose of a given drug based on BSA, the concentration of that drug in the bloodstream of one person may be 10 times higher or lower compared to that of the other person.[22] This variability is typical with many chemotherapy drugs dosed by BSA, and, as shown below, was demonstrated in a study of 14 common chemotherapy drugs.[17]
The result of this pharmacokinetic variability among people is that many people do not receive the right dose to achieve optimal treatment effectiveness with minimized toxic side effects. Some people are overdosed while others are underdosed.[18][20][21][23][24][25][26] For example, in a randomized clinical trial, investigators found 85% of metastatic colorectal cancer patients treated with 5-fluorouracil (5-FU) did not receive the optimal therapeutic dose when dosed by the BSA standard—68% were underdosed and 17% were overdosed.[23]
There has been controversy over the use of BSA to calculate chemotherapy doses for people who are
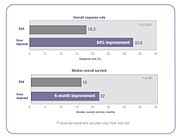
Several clinical studies have demonstrated that when chemotherapy dosing is individualized to achieve optimal systemic drug exposure, treatment outcomes are improved and toxic side effects are reduced.[23][25] In the 5-FU clinical study cited above, people whose dose was adjusted to achieve a pre-determined target exposure realized an 84% improvement in treatment response rate and a six-month improvement in overall survival (OS) compared with those dosed by BSA.[23]
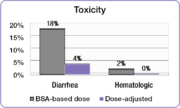
In the same study, investigators compared the incidence of common 5-FU-associated grade 3/4 toxicities between the dose-adjusted people and people dosed per BSA.[23] The incidence of debilitating grades of diarrhea was reduced from 18% in the BSA-dosed group to 4% in the dose-adjusted group and serious hematologic side effects were eliminated.[23] Because of the reduced toxicity, dose-adjusted patients were able to be treated for longer periods of time.[23] BSA-dosed people were treated for a total of 680 months while people in the dose-adjusted group were treated for a total of 791 months.[23] Completing the course of treatment is an important factor in achieving better treatment outcomes.
Similar results were found in a study involving people with colorectal cancer who have been treated with the popular FOLFOX regimen.[25] The incidence of serious diarrhea was reduced from 12% in the BSA-dosed group of patients to 1.7% in the dose-adjusted group, and the incidence of severe mucositis was reduced from 15% to 0.8%.[25]
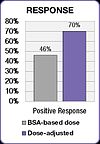
The FOLFOX study also demonstrated an improvement in treatment outcomes.[25] Positive response increased from 46% in the BSA-dosed group to 70% in the dose-adjusted group. Median progression free survival (PFS) and overall survival (OS) both improved by six months in the dose adjusted group.[25]
One approach that can help clinicians individualize chemotherapy dosing is to measure the drug levels in blood plasma over time and adjust dose according to a formula or algorithm to achieve optimal exposure. With an established target exposure for optimized treatment effectiveness with minimized toxicities, dosing can be personalized to achieve target exposure and optimal results for each person. Such an algorithm was used in the clinical trials cited above and resulted in significantly improved treatment outcomes.[28]
Oncologists are already individualizing dosing of some cancer drugs based on exposure. Carboplatin[29]: 4 and busulfan[30][31] dosing rely upon results from blood tests to calculate the optimal dose for each person. Simple blood tests are also available for dose optimization of methotrexate,[32] 5-FU, paclitaxel, and docetaxel.[33][34]
The serum albumin level immediately prior to chemotherapy administration is an independent prognostic predictor of survival in various cancer types.[35]
Types

Alkylating agents
Alkylating agents are the oldest group of chemotherapeutics in use today. Originally derived from mustard gas used in World War I, there are now many types of alkylating agents in use.[4] They are so named because of their ability to alkylate many molecules, including proteins, RNA and DNA. This ability to bind covalently to DNA via their alkyl group is the primary cause for their anti-cancer effects.[37] DNA is made of two strands and the molecules may either bind twice to one strand of DNA (intrastrand crosslink) or may bind once to both strands (interstrand crosslink). If the cell tries to replicate crosslinked DNA during cell division, or tries to repair it, the DNA strands can break. This leads to a form of programmed cell death called apoptosis.[36][38] Alkylating agents will work at any point in the cell cycle and thus are known as cell cycle-independent drugs. For this reason, the effect on the cell is dose dependent; the fraction of cells that die is directly proportional to the dose of drug.[39]
The subtypes of alkylating agents are the
Antimetabolites

The anti-folates include
Anti-microtubule agents

Vinca alkaloids are derived from the
Taxanes are natural and semi-synthetic drugs. The first drug of their class, paclitaxel, was originally extracted from Taxus brevifolia, the Pacific yew. Now this drug and another in this class, docetaxel, are produced semi-synthetically from a chemical found in the bark of another yew tree, Taxus baccata.[56]
Podophyllotoxin is an antineoplastic lignan obtained primarily from the American mayapple (Podophyllum peltatum) and Himalayan mayapple (Sinopodophyllum hexandrum). It has anti-microtubule activity, and its mechanism is similar to that of vinca alkaloids in that they bind to tubulin, inhibiting microtubule formation. Podophyllotoxin is used to produce two other drugs with different mechanisms of action: etoposide and teniposide.[57][58]
Topoisomerase inhibitors

Topoisomerase inhibitors are drugs that affect the activity of two enzymes:
Two topoisomerase I inhibitors,
Cytotoxic antibiotics
The cytotoxic
Among the anthracyclines,
Actinomycin is a complex molecule that intercalates DNA and prevents
Bleomycin, a
: 87Mitomycin is a cytotoxic antibiotic with the ability to alkylate DNA.[68]
Delivery

Most chemotherapy is delivered intravenously, although a number of agents can be administered orally (e.g., melphalan, busulfan, capecitabine). According to a recent (2016) systematic review, oral therapies present additional challenges for patients and care teams to maintain and support adherence to treatment plans.[69]
There are many intravenous methods of drug delivery, known as vascular access devices. These include the
Depending on the person, the cancer, the stage of cancer, the type of chemotherapy, and the dosage, intravenous chemotherapy may be given on either an
Topical chemotherapies, such as
If the cancer has
Adverse effects
Chemotherapeutic techniques have a range of side effects that depend on the type of medications used. The most common medications affect mainly the
Immunosuppression and myelosuppression
Virtually all chemotherapeutic regimens can cause depression of the immune system, often by paralysing the bone marrow and leading to a decrease of white blood cells, red blood cells, and platelets.
In very severe
Although people receiving chemotherapy are encouraged to wash their hands, avoid sick people, and take other infection-reducing steps, about 85% of infections are due to naturally occurring microorganisms in the person's own
In
Trilaciclib is an inhibitor of cyclin-dependent kinase 4/6 approved for the prevention of myelosuppression caused by chemotherapy. The drug is given before chemotherapy to protect bone marrow function.[81]
Neutropenic enterocolitis
Due to immune system suppression,
Gastrointestinal distress
Anemia
Nausea and vomiting
Hair loss

Hair loss (alopecia) can be caused by chemotherapy that kills rapidly dividing cells; other medications may cause hair to thin. These are most often temporary effects: hair usually starts to regrow a few weeks after the last treatment, but sometimes with a change in color, texture, thickness or style. Sometimes hair has a tendency to curl after regrowth, resulting in "chemo curls." Severe hair loss occurs most often with drugs such as doxorubicin, daunorubicin, paclitaxel, docetaxel, cyclophosphamide, ifosfamide and etoposide. Permanent thinning or hair loss can result from some standard chemotherapy regimens.[101]
Chemotherapy induced hair loss occurs by a non-androgenic mechanism, and can manifest as alopecia totalis, telogen effluvium, or less often alopecia areata.[102] It is usually associated with systemic treatment due to the high mitotic rate of hair follicles, and more reversible than androgenic hair loss,[103][104] although permanent cases can occur.[105] Chemotherapy induces hair loss in women more often than men.[106]
Scalp cooling offers a means of preventing both permanent and temporary hair loss; however, concerns about this method have been raised.[107][108]
Secondary neoplasm
Development of secondary neoplasia after successful chemotherapy or radiotherapy treatment can occur. The most common
Infertility
Some types of chemotherapy are gonadotoxic and may cause infertility.[111] Chemotherapies with high risk include procarbazine and other alkylating drugs such as cyclophosphamide, ifosfamide, busulfan, melphalan, chlorambucil, and chlormethine.[111] Drugs with medium risk include doxorubicin and platinum analogs such as cisplatin and carboplatin.[111] On the other hand, therapies with low risk of gonadotoxicity include plant derivatives such as vincristine and vinblastine, antibiotics such as bleomycin and dactinomycin, and antimetabolites such as methotrexate, mercaptopurine, and 5-fluorouracil.[111]
People may choose between several methods of fertility preservation prior to chemotherapy, including cryopreservation of semen, ovarian tissue, oocytes, or embryos.[113] As more than half of cancer patients are elderly, this adverse effect is only relevant for a minority of patients. A study in France between 1999 and 2011 came to the result that embryo freezing before administration of gonadotoxic agents to females caused a delay of treatment in 34% of cases, and a live birth in 27% of surviving cases who wanted to become pregnant, with the follow-up time varying between 1 and 13 years.[114]
Potential protective or attenuating agents include
In chemotherapy as a
Teratogenicity
Chemotherapy is
In males previously having undergone chemotherapy or radiotherapy, there appears to be no increase in genetic defects or congenital malformations in their children conceived after therapy.
Peripheral neuropathy
Between 30 and 40 percent of people undergoing chemotherapy experience chemotherapy-induced peripheral neuropathy (CIPN), a progressive, enduring, and often irreversible condition, causing pain, tingling, numbness and sensitivity to cold, beginning in the hands and feet and sometimes progressing to the arms and legs.[118] Chemotherapy drugs associated with CIPN include thalidomide, epothilones, vinca alkaloids, taxanes, proteasome inhibitors, and the platinum-based drugs.[118][119] Whether CIPN arises, and to what degree, is determined by the choice of drug, duration of use, the total amount consumed and whether the person already has peripheral neuropathy. Though the symptoms are mainly sensory, in some cases motor nerves and the autonomic nervous system are affected.[120] CIPN often follows the first chemotherapy dose and increases in severity as treatment continues, but this progression usually levels off at completion of treatment. The platinum-based drugs are the exception; with these drugs, sensation may continue to deteriorate for several months after the end of treatment.[121] Some CIPN appears to be irreversible.[121] Pain can often be managed with drug or other treatment but the numbness is usually resistant to treatment.[122]
Cognitive impairment
Some people receiving chemotherapy report fatigue or non-specific neurocognitive problems, such as an inability to concentrate; this is sometimes called post-chemotherapy cognitive impairment, referred to as "chemo brain" in popular and social media.[123]
Tumor lysis syndrome
In particularly large tumors and cancers with high
Organ damage
Nephrotoxicity (kidney damage) can be caused by tumor lysis syndrome and also due direct effects of drug clearance by the kidneys. Different drugs will affect different parts of the kidney and the toxicity may be asymptomatic (only seen on blood or urine tests) or may cause acute kidney injury.[128][129]
Other side-effects
Less common side-effects include red skin (
Specific chemotherapeutic agents are associated with organ-specific toxicities, including
Hand-foot syndrome is another side effect to cytotoxic chemotherapy.[136]
Nutritional problems are also frequently seen in cancer patients at diagnosis and through chemotherapy treatment. Research suggests that in children and young people undergoing cancer treatment, parenteral nutrition may help with this leading to weight gain and increased calorie and protein intake, when compared to enteral nutrition.[137]
Limitations
Chemotherapy does not always work, and even when it is useful, it may not completely destroy the cancer. People frequently fail to understand its limitations. In one study of people who had been newly diagnosed with incurable,
The
Blood vessels in tumors are very different from those seen in normal tissues. As a tumor grows, tumor cells furthest away from the blood vessels become low in oxygen (hypoxic). To counteract this they then signal for new blood vessels to grow. The newly formed tumor vasculature is poorly formed and does not deliver an adequate blood supply to all areas of the tumor. This leads to issues with drug delivery because many drugs will be delivered to the tumor by the circulatory system.[142]
Resistance
Resistance is a major cause of treatment failure in chemotherapeutic drugs. There are a few possible causes of resistance in cancer, one of which is the presence of small pumps on the surface of cancer cells that actively move chemotherapy from inside the cell to the outside. Cancer cells produce high amounts of these pumps, known as p-glycoprotein, in order to protect themselves from chemotherapeutics. Research on p-glycoprotein and other such chemotherapy efflux pumps is currently ongoing. Medications to inhibit the function of p-glycoprotein are undergoing investigation, but due to toxicities and interactions with anti-cancer drugs their development has been difficult.[143][144] Another mechanism of resistance is gene amplification, a process in which multiple copies of a gene are produced by cancer cells. This overcomes the effect of drugs that reduce the expression of genes involved in replication. With more copies of the gene, the drug can not prevent all expression of the gene and therefore the cell can restore its proliferative ability. Cancer cells can also cause defects in the cellular pathways of apoptosis (programmed cell death). As most chemotherapy drugs kill cancer cells in this manner, defective apoptosis allows survival of these cells, making them resistant. Many chemotherapy drugs also cause DNA damage, which can be repaired by enzymes in the cell that carry out DNA repair. Upregulation of these genes can overcome the DNA damage and prevent the induction of apoptosis. Mutations in genes that produce drug target proteins, such as tubulin, can occur which prevent the drugs from binding to the protein, leading to resistance to these types of drugs.[145] Drugs used in chemotherapy can induce cell stress, which can kill a cancer cell; however, under certain conditions, cells stress can induce changes in gene expression that enables resistance to several types of drugs.[146] In lung cancer, the transcription factor NFκB is thought to play a role in resistance to chemotherapy, via inflammatory pathways.[147][148][149]
Cytotoxics and targeted therapies
Mechanism of action

In the broad sense, most chemotherapeutic drugs work by impairing
As chemotherapy affects cell division, tumors with high
Cells from the immune system also make crucial contributions to the antitumor effects of chemotherapy.[162] For example, the chemotherapeutic drugs oxaliplatin and cyclophosphamide can cause tumor cells to die in a way that is detectable by the immune system (called immunogenic cell death), which mobilizes immune cells with antitumor functions.[163] Chemotherapeutic drugs that cause cancer immunogenic tumor cell death can make unresponsive tumors sensitive to immune checkpoint therapy.[164]
Other uses
Some chemotherapy drugs are used in diseases other than cancer, such as in autoimmune disorders,
Chemotherapy drugs are also used in
Occupational exposure and safe handling
This section is written like a manual or guide. (June 2023) |
In the 1970s, antineoplastic (chemotherapy) drugs were identified as hazardous, and the American Society of Health-System Pharmacists (ASHP) has since then introduced the concept of hazardous drugs after publishing a recommendation in 1983 regarding handling hazardous drugs. The adaptation of federal regulations came when the U.S. Occupational Safety and Health Administration (OSHA) first released its guidelines in 1986 and then updated them in 1996, 1999, and, most recently, 2006.[174]
The National Institute for Occupational Safety and Health (NIOSH) has been conducting an assessment in the workplace since then regarding these drugs. Occupational exposure to antineoplastic drugs has been linked to multiple health effects, including infertility and possible carcinogenic effects. A few cases have been reported by the NIOSH alert report, such as one in which a female pharmacist was diagnosed with papillary transitional cell carcinoma. Twelve years before the pharmacist was diagnosed with the condition, she had worked for 20 months in a hospital where she was responsible for preparing multiple antineoplastic drugs.[175] The pharmacist did not have any other risk factor for cancer, and therefore, her cancer was attributed to the exposure to the antineoplastic drugs, although a cause-and-effect relationship has not been established in the literature. Another case happened when a malfunction in biosafety cabinetry is believed to have exposed nursing personnel to antineoplastic drugs. Investigations revealed evidence of genotoxic biomarkers two and nine months after that exposure.
Routes of exposure
Antineoplastic drugs are usually given through
Dermal exposure is thought to be the main route of exposure due to the fact that significant amounts of the antineoplastic agents have been found in the gloves worn by healthcare workers who prepare, handle, and administer the agents. Another noteworthy route of exposure is inhalation of the drugs' vapors. Multiple studies have investigated inhalation as a route of exposure, and although air sampling has not shown any dangerous levels, it is still a potential route of exposure. Ingestion by hand to mouth is a route of exposure that is less likely compared to others because of the enforced hygienic standard in the health institutions. However, it is still a potential route, especially in the workplace, outside of a health institute. One can also be exposed to these hazardous drugs through injection by needle sticks. Research conducted in this area has established that occupational exposure occurs by examining evidence in multiple urine samples from health care workers.[177]
Hazards
Hazardous drugs expose health care workers to serious health risks. Many studies show that antineoplastic drugs could have many side effects on the reproductive system, such as fetal loss, congenital malformation, and infertility. Health care workers who are exposed to antineoplastic drugs on many occasions have adverse reproductive outcomes such as spontaneous abortions, stillbirths, and congenital malformations. Moreover, studies have shown that exposure to these drugs leads to menstrual cycle irregularities. Antineoplastic drugs may also increase the risk of learning disabilities among children of health care workers who are exposed to these hazardous substances.[178]
Moreover, these drugs have carcinogenic effects. In the past five decades, multiple studies have shown the carcinogenic effects of exposure to antineoplastic drugs. Similarly, there have been research studies that linked alkylating agents with humans developing leukemias. Studies have reported elevated risk of breast cancer, nonmelanoma skin cancer, and cancer of the rectum among nurses who are exposed to these drugs. Other investigations revealed that there is a potential genotoxic effect from anti-neoplastic drugs to workers in health care settings.[175]
Safe handling in health care settings
As of 2018, there were no occupational exposure limits set for antineoplastic drugs, i.e., OSHA or the American Conference of Governmental Industrial Hygienists (ACGIH) have not set workplace safety guidelines.[179]
Preparation
NIOSH recommends using a ventilated cabinet that is designed to decrease worker exposure. Additionally, it recommends training of all staff, the use of cabinets, implementing an initial evaluation of the technique of the safety program, and wearing protective gloves and gowns when opening drug packaging, handling vials, or labeling. When wearing personal protective equipment, one should inspect gloves for physical defects before use and always wear double gloves and protective gowns. Health care workers are also required to wash their hands with water and soap before and after working with antineoplastic drugs, change gloves every 30 minutes or whenever punctured, and discard them immediately in a chemotherapy waste container.[180]
The gowns used should be disposable gowns made of polyethylene-coated polypropylene. When wearing gowns, individuals should make sure that the gowns are closed and have long sleeves. When preparation is done, the final product should be completely sealed in a plastic bag.[181]
The health care worker should also wipe all waste containers inside the ventilated cabinet before removing them from the cabinet. Finally, workers should remove all protective wear and put them in a bag for their disposal inside the ventilated cabinet.[176]
Administration
Drugs should only be administered using protective medical devices such as needle lists and closed systems and techniques such as priming of IV tubing by pharmacy personnel inside a ventilated cabinet. Workers should always wear personal protective equipment such as double gloves, goggles, and protective gowns when opening the outer bag and assembling the delivery system to deliver the drug to the patient, and when disposing of all material used in the administration of the drugs.[179]
Hospital workers should never remove tubing from an IV bag that contains an antineoplastic drug, and when disconnecting the tubing in the system, they should make sure the tubing has been thoroughly flushed. After removing the IV bag, the workers should place it together with other disposable items directly in the yellow chemotherapy waste container with the lid closed. Protective equipment should be removed and put into a disposable chemotherapy waste container. After this has been done, one should double bag the chemotherapy waste before or after removing one's inner gloves. Moreover, one must always wash one's hands with soap and water before leaving the drug administration site.[182]
Employee training
All employees whose jobs in health care facilities expose them to hazardous drugs must receive training. Training should include shipping and receiving personnel, housekeepers, pharmacists, assistants, and all individuals involved in the transportation and storage of antineoplastic drugs. These individuals should receive information and training to inform them of the hazards of the drugs present in their areas of work. They should be informed and trained on operations and procedures in their work areas where they can encounter hazards, different methods used to detect the presence of hazardous drugs and how the hazards are released, and the physical and health hazards of the drugs, including their reproductive and carcinogenic hazard potential. Additionally, they should be informed and trained on the measures they should take to avoid and protect themselves from these hazards. This information ought to be provided when health care workers come into contact with the drugs, that is, perform the initial assignment in a work area with hazardous drugs. Moreover, training should also be provided when new hazards emerge as well as when new drugs, procedures, or equipment are introduced.[179]
Housekeeping and waste disposal
When performing cleaning and decontaminating the work area where antineoplastic drugs are used, one should make sure that there is sufficient ventilation to prevent the buildup of airborne drug concentrations. When cleaning the work surface, hospital workers should use deactivation and cleaning agents before and after each activity as well as at the end of their shifts. Cleaning should always be done using double protective gloves and disposable gowns. After employees finish up cleaning, they should dispose of the items used in the activity in a yellow chemotherapy waste container while still wearing protective gloves. After removing the gloves, they should thoroughly wash their hands with soap and water. Anything that comes into contact or has a trace of the antineoplastic drugs, such as needles, empty vials, syringes, gowns, and gloves, should be put in the chemotherapy waste container.[183]
Spill control
A written policy needs to be in place in case of a spill of antineoplastic products. The policy should address the possibility of various sizes of spills as well as the procedure and personal protective equipment required for each size. A trained worker should handle a large spill and always dispose of all cleanup materials in the chemical waste container according to EPA regulations, not in a yellow chemotherapy waste container.[184]
Occupational monitoring
A
Urinary mutagenicity is a marker of exposure to antineoplastic drugs that was first used by Falck and colleagues in 1979 and uses bacterial mutagenicity assays. Apart from being nonspecific, the test can be influenced by extraneous factors such as dietary intake and smoking and is, therefore, used sparingly. However, the test played a significant role in changing the use of horizontal flow cabinets to vertical flow biological safety cabinets during the preparation of antineoplastic drugs because the former exposed health care workers to high levels of drugs. This changed the handling of drugs and effectively reduced workers' exposure to antineoplastic drugs.[175]
Biomarkers of exposure to antineoplastic drugs commonly include urinary
Available agents
There is an extensive list of antineoplastic agents. Several classification schemes have been used to subdivide the medicines used for cancer into several different types.[185][186]
History

The first use of
The term chemotherapy
The word chemotherapy without a modifier usually refers to cancer treatment, but its historical meaning was broader. The term was coined in the early 1900s by
Research
Targeted delivery vehicles
Specially targeted delivery vehicles aim to increase effective levels of chemotherapy for tumor cells while reducing effective levels for other cells. This should result in an increased tumor kill or reduced toxicity or both.[194]
Antibody-drug conjugates
The first approved drug of this type was
Nanoparticles
Electrochemotherapy
Electrochemotherapy is the combined treatment in which injection of a chemotherapeutic drug is followed by application of high-voltage electric pulses locally to the tumor. The treatment enables the chemotherapeutic drugs, which otherwise cannot or hardly go through the membrane of cells (such as bleomycin and cisplatin), to enter the cancer cells. Hence, greater effectiveness of antitumor treatment is achieved.[200]
Clinical electrochemotherapy has been successfully used for treatment of cutaneous and subcutaneous tumors irrespective of their histological origin.[201][202] The method has been reported as safe, simple and highly effective in all reports on clinical use of electrochemotherapy. According to the ESOPE project (European Standard Operating Procedures of Electrochemotherapy), the Standard Operating Procedures (SOP) for electrochemotherapy were prepared, based on the experience of the leading European cancer centres on electrochemotherapy.[203][204] Recently, new electrochemotherapy modalities have been developed for treatment of internal tumors using surgical procedures, endoscopic routes or percutaneous approaches to gain access to the treatment area.[205][206]
Hyperthermia therapy
Hyperthermia therapy is heat treatment for cancer that can be a powerful tool when used in combination with chemotherapy (thermochemotherapy) or radiation for the control of a variety of cancers. The heat can be applied locally to the tumor site, which will dilate blood vessels to the tumor, allowing more chemotherapeutic medication to enter the tumor. Additionally, the tumor cell membrane will become more porous, further allowing more of the chemotherapeutic medicine to enter the tumor cell.
Hyperthermia has also been shown to help prevent or reverse "chemo-resistance." Chemotherapy resistance sometimes develops over time as the tumors adapt and can overcome the toxicity of the chemo medication. "Overcoming chemoresistance has been extensively studied within the past, especially using CDDP-resistant cells. In regard to the potential benefit that drug-resistant cells can be recruited for effective therapy by combining chemotherapy with hyperthermia, it was important to show that chemoresistance against several anticancer drugs (e.g. mitomycin C, anthracyclines, BCNU, melphalan) including CDDP could be reversed at least partially by the addition of heat.[207]
Other animals
Chemotherapy is used in veterinary medicine similar to how it is used in human medicine.[208]
See also
- Anti-Cancer Drugs (journal)
- Antimicrobial chemotherapy
- Cancer and nausea
- Cancer-related fatigue
- Chemo brain
- Chemotherapy regimens
- Cytostasis
- Experimental cancer treatments
- Safe Handling of Hazardous Drugs
- Drug delivery
- Hyperthermia therapy
- Immunotherapy
- National Comprehensive Cancer Network
- Radiation induced cognitive decline
- Radiotherapy
- Virotherapy
References
- ^ PMID 26180516.
- S2CID 7429296.
- PMID 29514064.
- ^ .
- ^ PMID 28850174.
- ^ ISBN 978-0-470-09254-5.
- ^ ISBN 978-0-443-07101-0.
- ISBN 978-0-7817-7328-7.
- PMID 16061845.
- ISBN 978-0-7817-3629-9.
- ISBN 978-0-7817-5628-0.
- PMID 22730604.
- S2CID 35447562.
- PMID 15146036.
- PMID 20171403.
- PMID 2520314.
- ^ PMID 12175683.
- ^ PMID 17305252.
- PMID 13573353.
- ^ PMID 11953888.
- ^ PMID 22965963.
- ^ PMID 12488482.
- ^ S2CID 9557055.
- PMID 21855044.
- ^ PMID 22683364.
- PMID 22382460.
- ^ PMID 18922643.
- S2CID 36211632.
- ISBN 978-0-521-61816-8.
- S2CID 28646787.
- S2CID 21072472.
- ^ "ARK Methotrexate Assay". Ark Diagnostics. Archived from the original on 28 April 2014. Retrieved 28 April 2014.
- ^ "Customizing Chemotherapy for Better Cancer Care". My Care Diagnostics. Archived from the original on 28 April 2014. Retrieved 25 November 2018.
- ^ "A Brief History of BSA Dosing". My Care Diagnostics.
- S2CID 19558612.
- ^ ISBN 978-0470025062.
- ^ .
- ^ PMID 19002790.
- ^ PMID 14508075.
- PMID 3245973.
- ^ Takimoto CH, Calvo E (2008). "Principles of Oncologic Pharmacotherapy". In Pazdur R, Wagman LD, Camphausen KA, Hoskins WJ (eds.). Cancer Management: A Multidisciplinary Approach (11th ed.). Archived from the original on 15 May 2009. Retrieved 18 June 2009.
- ^ PMID 19476376.
- S2CID 31467685.
- PMID 12515569.
- PMID 1687171.
- S2CID 22483746.
- ^ PMID 20577942.
- ISBN 9783540563914.
- PMID 15032608.
- ISBN 9783540728795.
- ISBN 9781498704304.
- ^ PMID 22609781.
- ISBN 9783527326693.
- PMID 21695100.
- PMID 19072542.
- PMID 20622989.
- S2CID 247493530.
- .
- ^ Lodish H, Berk A, Zipursky SL, et al. (2000). Molecular Cell Biology. 4th edition. The Role of Topoisomerases in DNA Replication. New York: W. H. Freeman.
- S2CID 9257158.
- PMID 19377506.
- ISBN 9783540389163.
- ISBN 9783540389163.
- PMID 3048848.
- S2CID 13138853.
- PMID 2410919.
- PMID 1384141.
- PMID 2131038.
- PMID 26921292.
- PMID 21460264.
- PMID 20348274.
- PMID 18722924.
- PMID 10421507.
- S2CID 597295.
- PMID 27128488.
- PMID 15136552.
- ISBN 978-0-9645467-5-2.
- S2CID 2969472.
- ^ PMID 22258955.
- ^ "Coriolus Versicolor". Cancer.org. 10 June 2008. Archived from the original on 25 June 2010. Retrieved 7 August 2012.
- ^ Commissioner, Office of the (12 February 2021). "FDA Approves Drug to Reduce Bone Marrow Suppression Caused by Chemotherapy". FDA. Retrieved 9 March 2021.
- S2CID 25479771.
- ^ Sinicrope, Frank A. (2003). "Typhlitis". Holland-Frei Cancer Medicine. 6th Edition.
- ^ S2CID 46090832.
- S2CID 109778.
- PMID 30168576.
- PMID 28104979.
- PMID 10511589.
- PMID 16925107.
- PMID 18597709.
- PMID 19245931.
- S2CID 16527763.
- PMID 22592695.
- PMID 26576687.
- S2CID 70400559.
- PMID 16500197.
- PMID 23152233.
- PMID 26389491, retrieved 8 November 2022
- ISBN 978-0-387-24291-0.
Nausea and vomiting are two of the most feared cancer treatment-related side effects for cancer patients and their families.
- PMID 30233361.
- ^ "Chemotherapy-Related Hair Loss (Alopecia) in Children - Health Encyclopedia - University of Rochester Medical Center". www.urmc.rochester.edu. Retrieved 8 November 2022.
- PMID 17642856.
- PMID 23187775.
- PMID 9589208.
- S2CID 43583651.
- PMID 22901547.
- PMID 19341937.
- PMID 22178150.
- OCLC 606559421.
- PMID 17374815.
- ^ S2CID 20672988.
- ^ PMID 22647504.
- PMID 18790328.
- PMID 23832792.
- PMID 24833728.
- S2CID 25139582.
In turn citing: Sanders JE, Hawley J, Levy W, Gooley T, Buckner CD, Deeg HJ, Doney K, Storb R, Sullivan K, Witherspoon R, Appelbaum FR (April 1996). "Pregnancies following high-dose cyclophosphamide with or without high-dose busulfan or total-body irradiation and bone marrow transplantation". Blood. 87 (7): 3045–52.PMID 8639928. - ^ PMID 11476352.
- ^ a b del Pino BM (23 February 2010). "Chemotherapy-induced Peripheral Neuropathy". NCI Cancer Bulletin. 7 (4): 6.
- ^ Grisold W, Oberndorfer S, Windebank AJ (2012). "Chemotherapy and polyneuropathies" (PDF). European Association of Neurooncology Magazine. 12 (1). Archived (PDF) from the original on 12 August 2012.
- PMID 22271810.
- ^ S2CID 20411101.
- PMID 17623791.
- PMID 15169812.
- PMID 30085527, retrieved 8 November 2022
- S2CID 35723897.
- PMID 24099024.
- S2CID 39518402.
- PMID 16473645.
- PMID 15574506.
- PMID 22547603.
- PMID 19851045.
- PMID 31961948.
- PMID 27486906.
- PMID 31063591.
- PMC 7123056.
- PMID 32431787.
- PMID 26301790.
- PMID 23094723.
- PMID 17363519.
- S2CID 25331035.
- PMID 17538177.
- S2CID 42818461.
- PMID 12591977.
- ^ Crowley E, McDevitt CA, Callaghan R (2009). Multidrug Resistance in Cancer. Generating Inhibitors of P-Glycoprotein: Where to, Now?. Springer Protocols. pp. 405–432.
- PMID 16103712.
- S2CID 28992964.
- PMID 25025901.
- S2CID 199025494.
- PMID 23720710.
- PMID 22437869.
- PMID 18297955.
- S2CID 21014917.
- S2CID 30482752.
- S2CID 17934366.
- S2CID 1478778.
- PMID 18196605.
- PMID 9353182.
- ISBN 978-0-7817-4689-2.
- PMID 19651324.
- S2CID 22909070.
- PMID 23299535.
- S2CID 205490545.
- S2CID 4045072.
- PMID 26872698.
- ^ PMID 15100330.
- ^ PMID 11454634.
- S2CID 13015911.
- ^ PMID 23450553.
- ^ PMID 15106195.
- ^ S2CID 46120577.
- S2CID 11902371.
- ^ "NCCN Guidelines: Systemic Light Chain Amyloidosis" (PDF). National Comprehensive Cancer Network. Archived from the original (PDF) on 25 June 2021. Retrieved 25 February 2015.
- PMID 19896087.
- S2CID 7081365.
- ^ a b c d e Thomas h. Connor, PhD (March 2014). "Chemotherapy: Biomarkers of Exposure, Effect, Reproductive Hazards, and Cancer". The Oncology Pharmacist. Archived from the original on 25 June 2021. Retrieved 22 November 2018.
- ^ .
- ISBN 978-90-393-4331-9.
- PMID 25153300.
- ^ a b c "preventing occupational exposure to antineoplastic and other hazardous drugs in health care settings" (PDF). Archived (PDF) from the original on 13 September 2004.
- ^ "Hazardous Drugs Program Guides". lni.wa.gov. Archived from the original on 27 October 2019. Retrieved 22 November 2018.
- PMID 26989351.
- ISBN 9781315894430.[page needed]
- ^ "Safe management of wastes from health-care activities" (PDF). WHO.
- S2CID 3879822.
- ISBN 978-1-4419-1225-1.
- ISBN 978-1-60795-014-1.
- .
- ^ PMID 21247779.
- S2CID 205467419.
- ISBN 978-1-4020-3618-7.
- PMID 18308256.
- ^ PMID 18974103.
- PMID 19868743.
- ^ PMID 21501554.
- ^ PMID 22003066.
- PMID 28410237.
- S2CID 27827980.
- PMID 35457065.
- PMID 19554862.
- PMID 17435555.
- PMID 17435555.
- S2CID 46534637.
- .
- .
- PMID 15964138.
- PMID 20178589.
- S2CID 37581171.
- S2CID 39060053.