Antioxidant
Antioxidants are compounds that inhibit oxidation (usually occurring as autoxidation), a chemical reaction that can produce free radicals. Autoxidation leads to degradation of organic compounds, including living matter. Antioxidants are frequently added to industrial products, such as polymers, fuels, and lubricants, to extend their usable lifetimes.[1] Foods are also treated with antioxidants to forestall spoilage, in particular the rancidification of oils and fats. In cells, antioxidants such as glutathione, mycothiol, or bacillithiol, and enzyme systems like superoxide dismutase, can prevent damage from oxidative stress.[2]
Known dietary antioxidants are vitamins A, C, and E, but the term antioxidant has also been applied to numerous other dietary compounds that only have antioxidant properties in vitro, with little evidence for antioxidant properties in vivo.[3] Dietary supplements marketed as antioxidants have not been shown to maintain health or prevent disease in humans.[3][4]
History
As part of their adaptation from marine life, terrestrial plants began producing non-marine antioxidants such as
Early research on the role of antioxidants in biology focused on their use in preventing the oxidation of
Uses in technology
Food preservatives
Antioxidants are used as food additives to help guard against food deterioration. Exposure to oxygen and sunlight are the two main factors in the oxidation of food, so food is preserved by keeping in the dark and sealing it in containers or even coating it in wax, as with cucumbers. However, as oxygen is also important for plant respiration, storing plant materials in anaerobic conditions produces unpleasant flavors and unappealing colors.[12] Consequently, packaging of fresh fruits and vegetables contains an ≈8% oxygen atmosphere. Antioxidants are an especially important class of preservatives as, unlike bacterial or fungal spoilage, oxidation reactions still occur relatively rapidly in frozen or refrigerated food.[13] These preservatives include natural antioxidants such as ascorbic acid (AA, E300) and tocopherols (E306), as well as synthetic antioxidants such as propyl gallate (PG, E310), tertiary butylhydroquinone (TBHQ), butylated hydroxyanisole (BHA, E320) and butylated hydroxytoluene (BHT, E321).[14][15]
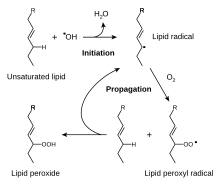
Unsaturated fats can be highly susceptible to oxidation, causing
Cosmetics preservatives
Antioxidant stabilizers are also added to fat-based cosmetics such as lipstick and moisturizers to prevent rancidity.[19] Antioxidants in cosmetic products prevent oxidation of active ingredients and lipid content. For example, phenolic antioxidants such as stilbenes, flavonoids, and hydroxycinnamic acid strongly absorb UV radiation due to the presence of chromophores. They reduce oxidative stress from sun exposure by absorbing UV light.[20]
Industrial uses

Antioxidants may be added to industrial products, such as
| Fuel additive | Components[22] | Applications[22] |
|---|---|---|
| AO-22 | N,N'-di-2-butyl-1,4-phenylenediamine
|
Turbine oils, transformer oils, hydraulic fluids, waxes, and greases |
| AO-24 | N,N'-di-2-butyl-1,4-phenylenediamine | Low-temperature oils |
| AO-29 | 2,6-di-tert-butyl-4-methylphenol (BHT)
|
Turbine oils, transformer oils, hydraulic fluids, waxes, greases, and gasolines |
| AO-30 | 2,4-dimethyl-6-tert-butylphenol
|
Jet fuels and gasolines, including aviation gasolines |
| AO-31 | 2,4-dimethyl-6-tert-butylphenol | Jet fuels and gasolines, including aviation gasolines |
| AO-32 | 2,4-dimethyl-6-tert-butylphenol and 2,6-di-tert-butyl-4-methylphenol | Jet fuels and gasolines, including aviation gasolines |
| AO-37 | 2,6-di-tert-butylphenol
|
Jet fuels and gasolines, widely approved for aviation fuels |
Antioxidant
Environmental and health hazards
Synthetic phenolic antioxidants (SPAs) and aminic antioxidants have potential human and environmental health hazards. SPAs are common in indoor dust, small air particles, sediment, sewage, river water and wastewater.[26] They are synthesized from phenolic compounds and include 2,6-di-tert-butyl-4-methylphenol (BHT), 2,6-di-tert-butyl-p-benzoquinone (BHT-Q), 2,4-di-tert-butyl-phenol (DBP) and 3-tert-butyl-4-hydroxyanisole (BHA). BHT can cause hepatotoxicity and damage to the endocrine system and may increase tumor development rates due to 1,1-dimethylhydrazine.[27] BHT-Q can cause DNA damage and mismatches[28] through the cleavage process, generating superoxide radicals.[26] DBP is toxic to marine life if exposed long-term. Phenolic antioxidants have low biodegradability, but they do not have severe toxicity toward aquatic organisms at low concentrations. Another type of antioxidant, diphenylamine (DPA), is commonly used in the production of commercial, industrial lubricants and rubber products and it also acts as a supplement for automotive engine oils.[29]
Oxidative challenge in biology
The vast majority of complex
Reactive oxygen species produced in cells include
The use of oxygen as part of the process for generating metabolic energy produces reactive oxygen species.
Examples of bioactive antioxidant compounds
The interactions between these different antioxidants may be synergistic and interdependent.[51][52] The action of one antioxidant may therefore depend on the proper function of other members of the antioxidant system.[32] The amount of protection provided by any one antioxidant will also depend on its concentration, its reactivity towards the particular reactive oxygen species being considered, and the status of the antioxidants with which it interacts.[32]
Some compounds contribute to antioxidant defense by chelating transition metals and preventing them from catalyzing the production of free radicals in the cell. The ability to sequester iron for iron-binding proteins, such as transferrin and ferritin, is one such function.[44] Selenium and zinc are commonly referred to as antioxidant minerals, but these chemical elements have no antioxidant action themselves, but rather are required for the activity of antioxidant enzymes, such as glutathione reductase and superoxide dismutase. (See also selenium in biology and zinc in biology.)
| Antioxidant | Solubility | Concentration in human serum (μM) | Concentration in liver tissue (μmol/kg) |
|---|---|---|---|
Ascorbic acid (vitamin C )
|
Water | 50–60[53] | 260 (human)[54] |
| Glutathione | Water | 4[55] | 6,400 (human)[54] |
| Lipoic acid | Water | 0.1–0.7[56] | 4–5 (rat)[57] |
| Uric acid | Water | 200–400[58] | 1,600 (human)[54] |
| Carotenes | Lipid | β-carotene: 0.5–1[59] | 5 (human, total carotenoids)[61] |
| α-Tocopherol (vitamin E) | Lipid | 10–40[60] | 50 (human)[54] |
Ubiquinol (coenzyme Q)
|
Lipid | 5[62] | 200 (human)[63] |
Uric acid
Uric acid has the highest concentration of any blood antioxidant[58] and provides over half of the total antioxidant capacity of human serum.[64] Uric acid's antioxidant activities are also complex, given that it does not react with some oxidants, such as superoxide, but does act against peroxynitrite,[65] peroxides, and hypochlorous acid.[66] Concerns over elevated UA's contribution to gout must be considered one of many risk factors.[67] By itself, UA-related risk of gout at high levels (415–530 μmol/L) is only 0.5% per year with an increase to 4.5% per year at UA supersaturation levels (535+ μmol/L).[68] Many of these aforementioned studies determined UA's antioxidant actions within normal physiological levels,[69][65] and some found antioxidant activity at levels as high as 285 μmol/L.[70]
Vitamin C
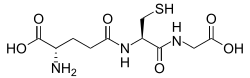
Glutathione
Vitamin E
It has been claimed that the α-tocopherol form is the most important lipid-soluble antioxidant, and that it protects membranes from oxidation by reacting with lipid radicals produced in the lipid peroxidation chain reaction.[81][84] This removes the free radical intermediates and prevents the propagation reaction from continuing. This reaction produces oxidised α-tocopheroxyl radicals that can be recycled back to the active reduced form through reduction by other antioxidants, such as ascorbate, retinol or ubiquinol.[85] This is in line with findings showing that α-tocopherol, but not water-soluble antioxidants, efficiently protects glutathione peroxidase 4 (GPX4)-deficient cells from cell death.[86] GPx4 is the only known enzyme that efficiently reduces lipid-hydroperoxides within biological membranes.
However, the roles and importance of the various forms of vitamin E are presently unclear,[87][88] and it has even been suggested that the most important function of α-tocopherol is as a signaling molecule, with this molecule having no significant role in antioxidant metabolism.[89][90] The functions of the other forms of vitamin E are even less well understood, although γ-tocopherol is a nucleophile that may react with electrophilic mutagens,[83] and tocotrienols may be important in protecting neurons from damage.[91]
Pro-oxidant activities
Antioxidants that are reducing agents can also act as pro-oxidants. For example, vitamin C has antioxidant activity when it reduces oxidizing substances such as hydrogen peroxide;[92] however, it will also reduce metal ions such as iron and copper[93] that generate free radicals through the Fenton reaction.[35][94] While ascorbic acid is effective antioxidant, it can also oxidatively change the flavor and color of food. With the presence of transition metals, there are low concentrations of ascorbic acid that can act as a radical scavenger in the Fenton reaction.[93]
2 Fe3+ + Ascorbate → 2 Fe2+ + Dehydroascorbate
2 Fe2+ + 2 H2O2 → 2 Fe3+ + 2 OH· + 2 OH−
The relative importance of the antioxidant and pro-oxidant activities of antioxidants is an area of current research, but vitamin C, which exerts its effects as a vitamin by oxidizing polypeptides, appears to have a mostly antioxidant action in the human body.[94]
Enzyme systems
As with the chemical antioxidants, cells are protected against oxidative stress by an interacting network of antioxidant enzymes.[30][31] Here, the superoxide released by processes such as oxidative phosphorylation is first converted to hydrogen peroxide and then further reduced to give water. This detoxification pathway is the result of multiple enzymes, with superoxide dismutases catalysing the first step and then catalases and various peroxidases removing hydrogen peroxide. As with antioxidant metabolites, the contributions of these enzymes to antioxidant defenses can be hard to separate from one another, but the generation of transgenic mice lacking just one antioxidant enzyme can be informative.[95]
Superoxide dismutase, catalase, and peroxiredoxins
Superoxide dismutases (SODs) are a class of closely related enzymes that catalyze the breakdown of the superoxide anion into oxygen and hydrogen peroxide.[96][97] SOD enzymes are present in almost all aerobic cells and in extracellular fluids.[98] Superoxide dismutase enzymes contain metal ion cofactors that, depending on the isozyme, can be copper, zinc, manganese or iron. In humans, the copper/zinc SOD is present in the cytosol, while manganese SOD is present in the mitochondrion.[97] There also exists a third form of SOD in extracellular fluids, which contains copper and zinc in its active sites.[99] The mitochondrial isozyme seems to be the most biologically important of these three, since mice lacking this enzyme die soon after birth.[100] In contrast, the mice lacking copper/zinc SOD (Sod1) are viable but have numerous pathologies and a reduced lifespan (see article on superoxide), while mice without the extracellular SOD have minimal defects (sensitive to hyperoxia).[95][101] In plants, SOD isozymes are present in the cytosol and mitochondria, with an iron SOD found in chloroplasts that is absent from vertebrates and yeast.[102]
Thioredoxin and glutathione systems
The
The glutathione system includes glutathione, glutathione reductase, glutathione peroxidases, and glutathione S-transferases.[76] This system is found in animals, plants and microorganisms.[76][121] Glutathione peroxidase is an enzyme containing four selenium-cofactors that catalyzes the breakdown of hydrogen peroxide and organic hydroperoxides. There are at least four different glutathione peroxidase isozymes in animals.[122] Glutathione peroxidase 1 is the most abundant and is a very efficient scavenger of hydrogen peroxide, while glutathione peroxidase 4 is most active with lipid hydroperoxides. Surprisingly, glutathione peroxidase 1 is dispensable, as mice lacking this enzyme have normal lifespans,[123] but they are hypersensitive to induced oxidative stress.[124] In addition, the glutathione S-transferases show high activity with lipid peroxides.[125] These enzymes are at particularly high levels in the liver and also serve in detoxification metabolism.[126]
Health research
Relation to diet
The dietary antioxidant vitamins A, C, and E are essential and required in specific daily amounts to prevent diseases.[3][127][128] Polyphenols, which have antioxidant properties in vitro due to their free hydroxy groups,[129] are extensively metabolized by catechol-O-methyltransferase which methylates free hydroxyl groups, and thereby prevents them from acting as antioxidants in vivo.[130][131]
Interactions
Common pharmaceuticals (and supplements) with antioxidant properties may interfere with the efficacy of certain
Adverse effects
Relatively strong reducing acids can have
| Foods | Reducing acid present |
|---|---|
| Cocoa bean and chocolate, spinach, turnip and rhubarb[137] | Oxalic acid |
| Whole grains, maize, legumes[138] | Phytic acid |
| Tea, beans, cabbage[137][139] | Tannins |
High doses of some antioxidants may have harmful long-term effects. The
Exercise and muscle soreness
A 2017 review showed that taking antioxidant dietary supplements before or after exercise is unlikely to produce a noticeable reduction in muscle soreness after a person exercises.[149]
Levels in food

Antioxidant vitamins are found in vegetables, fruits, eggs, legumes and nuts. Vitamins A, C, and E can be destroyed by long-term storage or prolonged cooking.[150] The effects of cooking and food processing are complex, as these processes can also increase the bioavailability of antioxidants, such as some carotenoids in vegetables.[151] Processed food contains fewer antioxidant vitamins than fresh and uncooked foods, as preparation exposes food to heat and oxygen.[152]
| Antioxidant vitamins | Foods containing high levels of antioxidant vitamins[139][153][154] |
|---|---|
| Vitamin C (ascorbic acid) | Fresh or frozen fruits and vegetables |
| Vitamin E (tocopherols, tocotrienols) | Vegetable oils, nuts, and seeds |
provitamin A )
|
Fruit, vegetables and eggs |
Other antioxidants are not obtained from the diet, but instead are made in the body. For example, ubiquinol (coenzyme Q) is poorly absorbed from the gut and is made through the mevalonate pathway.[63] Another example is glutathione, which is made from amino acids. As any glutathione in the gut is broken down to free cysteine, glycine and glutamic acid before being absorbed, even large oral intake has little effect on the concentration of glutathione in the body.[155][156] Although large amounts of sulfur-containing amino acids such as acetylcysteine can increase glutathione,[157] no evidence exists that eating high levels of these glutathione precursors is beneficial for healthy adults.[158]
Measurement and invalidation of ORAC
Measurement of polyphenol and carotenoid content in food is not a straightforward process, as antioxidants collectively are a diverse group of compounds with different reactivities to various reactive oxygen species. In food science analyses in vitro, the oxygen radical absorbance capacity (ORAC) was once an industry standard for estimating antioxidant strength of whole foods, juices and food additives, mainly from the presence of polyphenols.[159][160] Earlier measurements and ratings by the United States Department of Agriculture were withdrawn in 2012 as biologically irrelevant to human health, referring to an absence of physiological evidence for polyphenols having antioxidant properties in vivo.[161] Consequently, the ORAC method, derived only from in vitro experiments, is no longer considered relevant to human diets or biology, as of 2010.[161]
Alternative in vitro measurements of antioxidant content in foods – also based on the presence of polyphenols – include the
References
- ISBN 3527306730.
- ^ S2CID 235200305.
- ^ a b c "Antioxidants: In Depth". National Center for Complementary and Integrative Health, US National Institutes of Health. 1 November 2013. Retrieved 17 March 2023.
- PMID 12361782.
- PMID 14527634.
- PMID 20259061.
- PMID 10335367.
- PMID 8821966.
- PMID 9846200.
- ^ Moureu C, Dufraisse C (1922). "Sur l'autoxydation: Les antioxygènes". Comptes Rendus des Séances et Mémoires de la Société de Biologie (in French). 86: 321–322.
- PMID 15735064.
- PMID 2647417.
- PMID 1184900.
- PMID 7600543.
- ^ "E number index". UK food guide. Archived from the original on 4 March 2007. Retrieved 5 March 2007.
- PMID 3288002.
- PMID 15212450.
- ^ ISBN 978-0-9531949-8-8, retrieved 15 April 2023
- S2CID 39562131.
Propyl Gallate is a generally recognized as safe (GRAS) antioxidant to protect fats, oils, and fat-containing food from rancidity that results from the formation of peroxides.
- S2CID 201156301.
- .
- ^ a b "Fuel antioxidants". Innospec Chemicals. Archived from the original on 15 October 2006. Retrieved 27 February 2007.
- ^ "Why use Antioxidants?". SpecialChem Adhesives. Archived from the original on 11 February 2007. Retrieved 27 February 2007.
- S2CID 221637214.
- PMID 36361475.
- ^ S2CID 56486425.
- S2CID 221637214.
- PMID 34146526.
- S2CID 213719260.
- ^ PMID 8660387.
- ^ S2CID 20240552.
- ^ PMID 15134565.
- S2CID 83598498.
- PMID 16978905.
- ^ PMID 7744317.
- S2CID 20217256.
- S2CID 207547763.
- PMID 1355616.
- PMID 11050436.
- S2CID 45366190.
- S2CID 2502238.
- PMID 18793173.
- PMID 15361522.
- ^ PMID 14527285.
- S2CID 27486669.
- PMID 15310815.
- PMID 18458346.
- PMID 15995679.
- S2CID 25306222.
- PMID 8993856.
- PMID 10541450.
- PMID 7688300.
- PMID 7787643.
- ^ PMID 11368163.
- PMID 10577998.
- PMID 1490813.
- PMID 9570844.
- ^ PMID 16375736.
- PMID 12081831.
- ^ PMID 8131277.
- PMID 1550343.
- S2CID 19895215.
- ^ PMID 14757233.
- PMID 8325534.
- ^ PMID 18600514.
- S2CID 6145651.
- PMID 17910294.
- PMID 3826098.
- PMID 17494796.
- PMID 17663642.
- ^ a b "Vitamin C". Micronutrient Information Center, Linus Pauling Institute, Oregon State University, Corvallis, OR. 1 July 2018. Retrieved 19 June 2019.
- S2CID 21196776.
- PMID 11997377.
- S2CID 85060539.
- PMID 8144521.
- ^ PMID 6137189.
- PMID 20308541.
- PMID 19578333.
- PMID 11544359.
- PMID 1444271.
- ^ S2CID 7272312.
- PMID 11160563.
- ^ S2CID 7031925.
- PMID 17561088.
- PMID 10793887.
- PMID 18762024.
- PMID 17561087.
- PMID 18160049.
- PMID 17561089.
- PMID 15134510. Archived from the originalon 6 October 2011.
- PMID 16458936.
- S2CID 39962659.
- ^ PMID 33795736.
- ^ S2CID 15426564.
- ^ PMID 9788901.
- PMID 12126755.
- ^ PMID 3315461.
- PMID 16099495.
- PMID 16087389.
- S2CID 20843002.
- S2CID 13070253.
- PMID 9214590.
- S2CID 4411482.
- PMID 10446501.
- PMID 1334030.
- PMID 12121759.
- PMID 9389716.
- S2CID 264033871.
- PMID 15917183.
- PMID 12517450.
- S2CID 29055779.
- PMID 18084892.
- S2CID 3570549.
- PMID 12586629.
- PMID 16606633.
- PMID 11728801.
- PMID 16782394.
- PMID 11012661.
- PMID 10657232.
- PMID 8736785.
- PMID 10569628.
- PMID 9195979.
- PMID 9712879.
- PMID 15025930.
- PMID 15822171.
- ^ PMID 15153272.
- ISBN 978-0-9722522-2-5.
- PMID 8743980.
- PMID 22794138.
- ^ "Flavonoids". Linus Pauling Institute, Oregon State University, Corvallis. 2016. Retrieved 24 July 2016.
- S2CID 205951215.
- PMID 34194012.
- PMID 12949395.
- PMID 12936958.
- PMID 16672077.
- ^ S2CID 1118651.
- PMID 12498628.
- ^ PMID 14519822.
- PMID 8901853.
- PMID 10359235.
- ^ PMID 17327526.
- ^ PMID 22419320.
- ^ Study Citing Antioxidant Vitamin Risks Based On Flawed Methodology, Experts Argue News release from Oregon State University published on ScienceDaily. Retrieved 19 April 2007
- PMID 15537682.
- ^ S2CID 20452618.
- PMID 32130738.
- PMID 16376462.
- PMID 29238948.
- PMID 15806788.
- PMID 19035552. Archived from the originalon 27 September 2018. Retrieved 18 April 2017.
- PMID 12002789.
- ^ "Antioxidants and Cancer Prevention: Fact Sheet". National Cancer Institute. Archived from the original on 4 March 2007. Retrieved 27 February 2007.
- PMID 17378953.
- S2CID 27606314.
- PMID 8183721.
- S2CID 74736842.
- PMID 16702341.
- PMID 8458588.
- PMID 11599998.
- ^ a b "Withdrawn: Oxygen Radical Absorbance Capacity (ORAC) of Selected Foods, Release 2 (2010)". United States Department of Agriculture, Agricultural Research Service. 16 May 2012. Retrieved 13 June 2012.
- PMID 15884874. Archived from the original(PDF) on 29 December 2016. Retrieved 24 October 2017.
Further reading
- Halliwell B, Gutteridge JM (2015). Free Radicals in Biology and Medicine (5th ed.). Oxford University Press. ISBN 978-0-19-856869-8.
- Lane N (2003). Oxygen: The Molecule That Made the World. Oxford University Press. ISBN 978-0-19-860783-0.
- Pokorny J, Yanishlieva N, Gordon MH (2001). Antioxidants in Food: Practical Applications. CRC Press. ISBN 978-0-8493-1222-9.
External links
 Media related to Antioxidants at Wikimedia Commons
Media related to Antioxidants at Wikimedia Commons

![{\displaystyle {\ce {{\underset {Oxygen}{O2}}->{\underset {Superoxide}{*O2^{-}}}->[{\ce {Superoxide \atop dismutase}}]{\underset {Hydrogen \atop peroxide}{H2O2}}->[{\ce {Peroxidases \atop catalase}}]{\underset {Water}{H2O}}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/8feecd26377be56b431d182ea9a398ab6b5d3b7f)