Appel reaction
| Appel reaction | |
|---|---|
| Named after | Rolf Appel |
| Reaction type | Substitution reaction |
| Identifiers | |
| Organic Chemistry Portal | appel-reaction |
| RSC ontology ID | RXNO:0000406 |
The Appel reaction is an
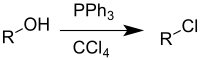
Drawbacks to the reaction are the use of toxic halogenating agents and the coproduction of organophosphorus product that must be separated from the organic product.[4] The phosphorus reagent can be used in catalytic quantities.[5][6] The corresponding alkyl bromide can also be synthesised by addition of lithium bromide as a source of bromide ions. A greener, more sustainable Appel reaction, free from chlorinated solvents and requiring only a catalytic amount of phosphine reagent, has also been reported.[7]

Mechanism
The Appel reaction begins with the formation of the
The driving force behind this and similar reactions is the formation of the strong PO double bond.

Illustrative use of the Appel reaction is the chlorination of geraniol to geranyl chloride.[11]
Modifications
The Appel reaction is also effective on
See also
References
- .
- ^ "Chemie". Archived from the original on 2019-08-11. Retrieved 2011-08-24.
- ISSN 0009-3068.
- ISBN 978-0-12-154350-1.
- PMID 21744876.
- PMID 21882274.
- S2CID 213147247.
- ISBN 9780470638859.
- ^ "Archived copy" (PDF). Archived from the original (PDF) on 2012-07-22. Retrieved 2012-07-11.
{{cite web}}: CS1 maint: archived copy as title (link) - ISBN 978-0-471-72091-1
- .
- .

