Aragonite
| Aragonite | ||
|---|---|---|
2V angle Measured 18–19° | | |
| Dispersion | Weak | |
| Extinction | Parallel | |
| Ultraviolet fluorescence | Faint white-blue to blue-violet | |
| Solubility | Soluble in acids, and saltwater (but takes longer) | |
| Common impurities | Commonly strontium, zirconium, lead | |
| Other characteristics | Thermodynamically unstable, Morphs slowly back into calcite | |
| References | [3][4] | |
Aragonite is a
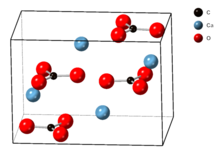
The
Occurrence
The
An aragonite cave, the Ochtinská Aragonite Cave, is situated in Slovakia.[9]
In the US, aragonite in the form of
Massive deposits of

Aragonite is the high pressure
Aragonite forms naturally in almost all
Aragonite also forms naturally in the endocarp of Celtis occidentalis.[19]
The skeleton of some calcareous sponges is made of aragonite.[citation needed]
Aragonite also forms in the ocean inorganic precipitates called marine cements (in the sediment) or as free crystals (in the water column).[20][21] Inorganic precipitation of aragonite in caves can occur in the form of speleothems.[22] Aragonite is common in serpentinites where magnesium-rich pore solutions apparently inhibit calcite growth and promote aragonite precipitation.[23]
Aragonite is
Aragonite can be synthesized by adding a calcium chloride solution to a sodium carbonate solution at temperatures above 60 °C (140 °F) or in water-ethanol mixtures at ambient temperatures.[25]
Physical properties
Aragonite is a
The mineral
Uses
In
Aragonite has been successfully tested for the removal of pollutants like zinc, cobalt and lead from contaminated wastewaters.[34]
Claims that magnetic water treatment can reduce scaling, by converting calcite to aragonite, have been met with skepticism,[35] but continue to be investigated.[36][37]
Gallery
-
Aragonite crystals from Cuenca, Castile-La Mancha, Spain
-
Aragonite crystal cluster from Spain
-
Remnant biogenic aragonite (thin, rainbow-colored shell) on the ammonite Baculites (Pierre Shale, Late Cretaceous, South Dakota)
-
Scanning electron microscope image of aragonite layers in the nacre of a blue mussel (Mytilus edulis)
-
Fluorescence of aragonite
See also
- Aragonite sea
- Ikaite, CaCO3·6H2O
- List of minerals
- Monohydrocalcite, CaCO3·H2O
- Nacre, otherwise known as "Mother-of-Pearl"
References
- S2CID 235729616.
- PMID 34876711.
- ^ "Aragonite Properties, Occurrence » Geology Science". 26 October 2021.
- ^ Aragonite, Mindat.org
- ISSN 0950-1207.
- ^ ISBN 0442276249.
- ISBN 978-1-43170-084-4.
- ISBN 978-84-95063-98-4.
- .
- ISBN 978-1-4612-3748-8.
- ISBN 978-0-88150-738-6.
- S2CID 129571671.
- ISBN 9780195106916.
- ISBN 0131547283.
- ^ S2CID 4285912.
- .
- S2CID 130591343.
- . Retrieved 1 August 2021.
- S2CID 49232599.[permanent dead link]
- PMID 36153366.
- ISBN 9781444314175.
- ^ Nesse 2000, p. 337.
- .
- .
- .
- ^ Carlson, W.D. (1980). "The calcite–aragonite equilibrium: effects of Sr substitution and anion orientational disorder". American Mineralogist. 65 (11–12): 1252–1262. Retrieved 31 July 2021.
- S2CID 250059382.
- ^ Fyfe, W.S. (1964). "Calcite aragonite problem" (PDF). AAPG Bulletin. 48 (4): 526. Retrieved 31 July 2021.
- .
- ISBN 0136427103.
- PMID 25031482.
- .
- ^ Orr, J. C., et al. (2005) Anthropogenic ocean acidification over the 21st century and its impact on calcifying organisms. Nature 437: 681-686
- ^
Krauter, PW; Harrar, JE; Orloff, SP; Bahowick, SM (1996). "Test of a Magnetic Device for Amelioration of Scale Formation at Treatment Facility D" (PDF). Internal Report. OSTI 567404. Retrieved 2009-12-11.
- S2CID 96367372.
- .





