Aripiprazole
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌɛərɪˈpɪprəzoʊl/ AIR-ih-PIP-rə-zohl Abilify /əˈbɪlɪfaɪ/ ə-BIL-if-eye |
| Trade names | Abilify, Aristada, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a603012 |
| License data |
|
| Pregnancy category |
|
intramuscular | |
| Drug class | Atypical antipsychotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 87%[4][5][6] |
| Protein binding | >99%[4][5][6] |
| Metabolism | Liver (mostly via CYP3A4 and 2D6[4][5][6]) |
| Elimination half-life | 75 hours (active metabolite is 94 hours)[4][5][6] |
| Excretion | Kidney (27%; <1% unchanged) feces (60%; 18% unchanged)[4][5][6] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Aripiprazole, sold under the brand names Abilify and Aristada, among others, is an
In adults, side effects with greater than 10% incidence include weight gain, headache,
Aripiprazole was approved for medical use in the United States in 2002.
Medical uses
Aripiprazole is primarily used for the treatment of schizophrenia or bipolar disorder.[6][7][16]
Schizophrenia
The 2016
A 2014 Cochrane review comparing aripiprazole and other atypical antipsychotics found that it is difficult to determine differences as data quality is poor.[19] A 2011 Cochrane review comparing aripiprazole with placebo concluded that high dropout rates in clinical trials, and a lack of outcome data regarding general functioning, behavior, mortality, economic outcomes, or cognitive functioning make it difficult to definitively conclude that aripiprazole is useful for the prevention of relapse.[8] A Cochrane review found only low quality evidence of effectiveness in treating schizophrenia.[8] Accordingly, part of its methodology on quality of evidence is based on quantity of qualified studies.[20]
A 2013 review placed aripiprazole in the middle range of 15 antipsychotics for effectiveness, approximately as effective as
In 2013 the World Federation of Societies for Biological Psychiatry recommended aripiprazole for the treatment of acute exacerbations of schizophrenia as a Grade 1 recommendation and evidence level A.[23]
The British Association for Psychopharmacology similarly recommends that all persons presenting with psychosis receive treatment with an antipsychotic, and that such treatment should continue for at least 1–2 years, as "There is no doubt that antipsychotic discontinuation is strongly associated with relapse during this period". The guideline further notes that "Established schizophrenia requires continued maintenance with doses of antipsychotic medication within the recommended range (Evidence level A)".[24]
The British Association for Psychopharmacology[24] and the World Federation of Societies for Biological Psychiatry suggest that there is little difference in effectiveness between antipsychotics in prevention of relapse, and recommend that the specific choice of antipsychotic be chosen based on each person's preference and side effect profile. The latter group recommends switching to aripiprazole when excessive weight gain is encountered during treatment with other antipsychotics.[23]
Bipolar disorder
Aripiprazole is effective for the treatment of acute manic episodes of bipolar disorder in adults, children, and adolescents.[25][26] Used as maintenance therapy, it is useful for the prevention of manic episodes, but is not useful for bipolar depression.[27][28] Thus, it is often used in combination with an additional mood stabilizer; however, co-administration with a mood stabilizer increases the risk of extrapyramidal side effects.[29] In September 2014, aripiprazole had a United Kingdom marketing authorization for up to twelve weeks of treatment for moderate to severe manic episodes in bipolar I disorder in young people aged thirteen and older. Aripiprazole in low doses of 2.5mg can cause mania in those with Bipolar disorder. [30][31][32]
Major depression
Aripiprazole is an effective add-on treatment for major depressive disorder; however, there is a greater rate of side effects such as weight gain and movement disorders.[33][34][35] The overall benefit is small to moderate and its use appears to neither improve quality of life nor functioning.[33] Aripiprazole may interact with some antidepressants, especially selective serotonin reuptake inhibitors (SSRIs) that are metabolized by CYP2D6. There are known interactions with fluoxetine and paroxetine[36] and it appears lesser interactions with sertraline, escitalopram, citalopram and fluvoxamine. CYP2D6 inhibitors increase aripiprazole concentrations to 2–3 times their normal level.[9] When strong CYP2D6 SSRI inhibitors are co-administered (e.g., fluoxetine, paroxetine), FDA recommends dose monitoring, although it is not clear if SSRI dose should be lowered if paroxetine or fluoxetine and aripiprazole are co-administered.[16][37][38][39]
Autism
Short-term data (8 weeks) shows reduced irritability, hyperactivity, inappropriate speech, and stereotypy, but no change in lethargic behaviours.[40] Adverse effects include weight gain, sleepiness, drooling and tremors.[40] It is suggested that children and adolescents need to be monitored regularly while taking this medication, to evaluate if this treatment option is still effective after long-term use and note if side effects are worsening. Further studies are needed to understand if this drug is helpful for children after long term use.[40]
Tic disorders
Aripiprazole is approved for the treatment of
Obsessive–compulsive disorder
A 2014 systematic review and meta-analysis concluded that add-on therapy with low dose aripiprazole is an effective treatment for obsessive–compulsive disorder (OCD) that does not improve with selective serotonin reuptake inhibitors (SSRIs) alone.[48] The conclusion was based on the results of two relatively small, short-term trials, each of which demonstrated improvements in symptoms.[48][49][50][51] However, aripiprazole is cautiously recommended by a 2017 review on antipsychotics for OCD.[52] Aripiprazole is not currently approved for the treatment of OCD and is instead used off-label for this indication.[41] Depending on the dose, aripiprazole can increase impulse control issues in a small percentage of people. FDA Drug Safety Communication warned about this side effect.[53]
Adverse effects
In the elderly with dementia, there is an increased risk of death.[54] In children, adolescents and young adults, there is an increased risk of suicide.[54][55]
In adults, side effects with greater than 10% incidence include weight gain, mania, headache, akathisia, insomnia, delirium, and gastro-intestinal effects like nausea and constipation, and lightheadedness.[4][5][6][9][10] Side effects in children are similar, and include sleepiness, increased appetite, and stuffy nose.[9] A strong desire to gamble, binge eat, shop, and engage in sexual activity may also occur rarely.[56][57] These urges can be uncontrollable.[56]
Uncontrolled movement such as restlessness, tremors, and muscle stiffness may occur.[9]
Discontinuation
The
There is tentative evidence that discontinuation of antipsychotics can result in psychosis as a part of a withdrawal syndrome.[60] It may also result in reoccurrence of the condition that is being treated.[61] Rarely tardive dyskinesia can occur when the medication is stopped.[59]
Overdose
Children or adults who ingested acute overdoses have usually manifested central nervous system depression ranging from mild sedation to coma; serum concentrations of aripiprazole and dehydroaripiprazole in these people were elevated by up to 3–4 fold over normal therapeutic levels; as of 2008 no deaths had been recorded.[62][63]
Interactions
Aripiprazole is a substrate of CYP2D6 and CYP3A4. Coadministration with medications that inhibit (e.g. paroxetine, fluoxetine) or induce (e.g. carbamazepine) these metabolic enzymes are known to increase and decrease, respectively, plasma levels of aripiprazole.[64][16]
Precautions should be taken in people with an established diagnosis of diabetes mellitus who are started on atypical antipsychotics along with other medications that affect blood sugar levels and should be monitored regularly for worsening of glucose control. The liquid form (oral solution) of this medication may contain up to 15 grams of sugar per dose.[7]
Antipsychotics like aripiprazole and stimulant medications, such as
Chemistry
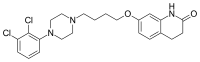
Aripiprazole belongs to the chemical class of drugs called
Pharmacology
Pharmacodynamics
Aripiprazole was discovered in 1988 by scientists at the Japanese firm Otsuka Pharmaceutical.[41][70][71][72][73]
| Site | Ki (nM) | IA (%) | Action | Ref |
|---|---|---|---|---|
| SERT | 900 – 1260 | Reuptake Inhibitor | [77][75] | |
| NET | 1340 – 2840 | Reuptake inbibitor | [75] | |
| DAT | 2560 – 3880 | Reuptake inhibitor | [75] | |
| 5-HT1A | 1.7 – 6.4 | ~75% | Partial agonist | [75][78][77] |
| 5-HT1B | 570 – 1090 | ND | [75] | |
| 5-HT1D | 57 – 79 | ND | [75] | |
| 5-HT1E | 3000 – >10,000 | ND | [75] | |
| 5-HT2A | 6.7 – 39 | 12.7% | Partial Agonist /
Functional Antagonist |
[77][75][78] |
| 5-HT2B | 0.25 – 0.47 | Inverse agonist | [75] | |
| 5-HT2C | 11 – 197 | 82% | Partial agonist | [77][75][78] |
| 5-HT3 | 520 – 740 | ND | [75] | |
| 5-HT5A | 960 – 1520 | Full Agonist | [75] | |
| 5-HT6 | 475 – 665 | Antagonist | [77][75][78] | |
| 5-HT7 | 6.6 – 14 | Partial Agonist /
Functional Antagonist |
[75][78][77] | |
| α1A | 26 | Antagonist | [75][78] | |
| α1B | 35 | Antagonist | [75] | |
| α2A | 74.3 | Antagonist | [75][78] | |
| α2B | 102 | ND | [75][78] | |
| α2C | 38 | Antagonist | [75][78] | |
| β1 | 141 | ND | [75] | |
| β2 | 163 | ND | [75] | |
D1 |
1290 – 2630 | Antagonist | [77][75] | |
D2 |
2.2 – 4.4 | ~60% | Partial agonist | [75] |
D2L
|
0.65 – 0.83 | Antagonist | ||
D3 |
4.3 – 15.1 | ~30% | Partial agonist | [77][75] |
D4 |
417 – 603 | Partial agonist | [77][75] | |
D5 |
1240 – 3940 | ND | [77][75] | |
H1 |
22.5 – 27.7 | Antagonist | [75][78][77] | |
H2 |
>10,000 | ND | [75] | |
H3 |
60 – 388 | ND | [75] | |
H4 |
>10,000 | ND | [75] | |
M1 |
6,780 | ND | [75] | |
M2 |
3,510 | ND | [75] | |
M3 |
4,680 | ND | [75][78] | |
M4 |
1,520 | ND | [75] | |
M5 |
2,330 | ND | [75] | |
| NMDA (PCP1) |
59 – 1824 | Antagonist | [75] | |
| Values are Ki (nM). The smaller the value, the more strongly the drug binds to the site. All data are for human cloned proteins, except 5-HT3 (rat), D4 (human/rat), H3 (guinea pig), and NMDA/PCP (rat).[75]
IA = Intrinsic Activity | ||||
Aripiprazole's mechanism of action is different from those of the other FDA-approved atypical antipsychotics (e.g., clozapine, olanzapine, quetiapine, ziprasidone, and risperidone).[79][80][81][82] It shows differential engagement at the dopamine receptor (D2[75]). Aripiprazole is a partial agonist at dopamine D2 receptors, partial agonist at 5-HT1A receptors and an antagonist at 5-HT2A, receptors.[83][84]
It appears to show predominantly antagonist activity on postsynaptic D2 receptors and partial agonist activity on presynaptic D2 receptors,
There are studies to date confirming aripiprazole as an antagonist at
As a pharmacologically unique antipsychotic with pronounced
Aripiprazole is also a partial agonist of the
Since the actions of aripiprazole differ markedly across receptor systems aripiprazole was sometimes an antagonist (e.g., at 5-HT6 and D2L), sometimes an inverse agonist (e.g., 5-HT2B), sometimes a partial agonist (e.g., D2L), and sometimes a full agonist (D3, D4). Aripiprazole was frequently found to be a partial agonist, with an intrinsic activity that could be low (D2L, 5-HT2A, 5-HT7), intermediate (5-HT1A), or high (D4, 5-HT2C). This mixture of agonist actions at D2-dopamine receptors is consistent with the hypothesis that aripiprazole has "functionally selective" actions.[100] The "functional-selectivity" hypothesis proposes that a mixture of agonist/partial agonist/antagonist actions are likely. According to this hypothesis, agonists may induce structural changes in receptor conformations that are differentially "sensed" by the local complement of G proteins to induce a variety of functional actions depending upon the precise cellular milieu. The diverse actions of aripiprazole at D2-dopamine receptors are clearly cell-type specific (e.g., agonism, antagonism, partial agonism), and are most parsimoniously explained by the "functional selectivity" hypothesis.[75]
Since 5-HT2C receptors have been implicated in the control of depression, OCD, and appetite, partial agonism at the 5-HT2C receptor might be associated with therapeutic potential in obsessive compulsive disorder, obesity, and depression. 5-HT2C agonism has been demonstrated to induce anorexia via enhancement of serotonergic neurotransmission via activation of 5-HT2C receptors; it is conceivable that the 5-HT2C partial agonist actions of aripiprazole may, thus, be partly responsible for the minimal weight gain associated with this compound in clinical trials. In terms of potential action as an antiobsessional agent, it is worthwhile noting that a variety of 5-HT2A/5-HT2C agonists have shown promise as antiobsessional agents, yet many of these compounds are hallucinogenic, presumably due to 5-HT2A activation. Aripiprazole has a favorable pharmacological profile in being a 5-HT2A antagonist and a 5-HT2C partial agonist. Based on this profile, one can predict that aripiprazole may have antiobsessional and anorectic actions in humans.[75]
Wood and Reavill's (2007) review of published and unpublished data proposed that, at therapeutically relevant doses, aripiprazole may act essentially as a selective partial agonist of the D2 receptor without significantly affecting the majority of serotonin receptors.[85] A positron emission tomography imaging study found that 10 to 30 mg/day aripiprazole resulted in 85 to 95% occupancy of the D2 receptor in various brain areas (putamen, caudate, ventral striatum) versus 54 to 60% occupancy of the 5-HT2A receptor and only 16% occupancy of the 5-HT1A receptor.[101][89] It has been suggested that the low occupancy of the 5-HT1A receptor by aripiprazole may have been an erroneous measurement however.[102]
Aripiprazole acts by modulating neurotransmission overactivity on the dopaminergic mesolimbic pathway, which is thought to be a cause of positive schizophrenia symptoms.[90] Due to its agonist activity on D2 receptors, aripiprazole may also increase dopaminergic activity to optimal levels in the mesocortical pathways where it is reduced.[90]
Pharmacokinetics
Aripiprazole displays linear kinetics and has an elimination half-life of approximately 75 hours. Steady-state plasma concentrations are achieved in about 14 days. Cmax (maximum plasma concentration) is achieved 3–5 hours after oral dosing. Bioavailability of the oral tablets is about 90% and the drug undergoes extensive hepatic metabolization (dehydrogenation, hydroxylation, and N-dealkylation), principally by the enzymes CYP2D6 and CYP3A4. Its only known active metabolite is dehydro-aripiprazole, which typically accumulates to approximately 40% of the aripiprazole concentration. The parenteral drug is excreted only in traces, and its metabolites, active or not, are excreted via feces and urine.[80][16]
| Medication | Brand name | Class | Vehicle | Dosage | Tmax | t1/2 single | t1/2 multiple | logPc | Ref |
|---|---|---|---|---|---|---|---|---|---|
| Aripiprazole lauroxil | Aristada | Atypical | Watera | 441–1064 mg/4–8 weeks | 24–35 days | ? | 54–57 days | 7.9–10.0 | |
| Aripiprazole monohydrate | Abilify Maintena | Atypical | Watera | 300–400 mg/4 weeks | 7 days | ? | 30–47 days | 4.9–5.2 | |
| Bromperidol decanoate | Impromen Decanoas | Typical | Sesame oil | 40–300 mg/4 weeks | 3–9 days | ? | 21–25 days | 7.9 | [103] |
Clopentixol decanoate |
Sordinol Depot | Typical | Viscoleob | 50–600 mg/1–4 weeks | 4–7 days | ? | 19 days | 9.0 | [104] |
Flupentixol decanoate |
Depixol | Typical | Viscoleob | 10–200 mg/2–4 weeks | 4–10 days | 8 days | 17 days | 7.2–9.2 | [104][105] |
Fluphenazine decanoate |
Prolixin Decanoate | Typical | Sesame oil | 12.5–100 mg/2–5 weeks | 1–2 days | 1–10 days | 14–100 days | 7.2–9.0 | [106][107][108] |
Fluphenazine enanthate |
Prolixin Enanthate | Typical | Sesame oil | 12.5–100 mg/1–4 weeks | 2–3 days | 4 days | ? | 6.4–7.4 | [107] |
| Fluspirilene | Imap, Redeptin | Typical | Watera | 2–12 mg/1 week | 1–8 days | 7 days | ? | 5.2–5.8 | [109] |
| Haloperidol decanoate | Haldol Decanoate | Typical | Sesame oil | 20–400 mg/2–4 weeks | 3–9 days | 18–21 days | 7.2–7.9 | [110][111] | |
Olanzapine pamoate |
Zyprexa Relprevv | Atypical | Watera | 150–405 mg/2–4 weeks | 7 days | ? | 30 days | – | |
| Oxyprothepin decanoate | Meclopin | Typical | ? | ? | ? | ? | ? | 8.5–8.7 | |
Paliperidone palmitate |
Invega Sustenna | Atypical | Watera | 39–819 mg/4–12 weeks | 13–33 days | 25–139 days | ? | 8.1–10.1 | |
Perphenazine decanoate |
Trilafon Dekanoat | Typical | Sesame oil | 50–200 mg/2–4 weeks | ? | ? | 27 days | 8.9 | |
| Perphenazine enanthate | Trilafon Enanthate | Typical | Sesame oil | 25–200 mg/2 weeks | 2–3 days | ? | 4–7 days | 6.4–7.2 | [112] |
Pipotiazine palmitate |
Piportil Longum | Typical | Viscoleob | 25–400 mg/4 weeks | 9–10 days | ? | 14–21 days | 8.5–11.6 | [105] |
Pipotiazine undecylenate |
Piportil Medium | Typical | Sesame oil | 100–200 mg/2 weeks | ? | ? | ? | 8.4 | |
| Risperidone | Risperdal Consta | Atypical | Microspheres |
12.5–75 mg/2 weeks | 21 days | ? | 3–6 days | – | |
Zuclopentixol acetate |
Clopixol Acuphase | Typical | Viscoleob | 50–200 mg/1–3 days | 1–2 days | 1–2 days | 4.7–4.9 | ||
Zuclopentixol decanoate |
Clopixol Depot | Typical | Viscoleob | 50–800 mg/2–4 weeks | 4–9 days | ? | 11–21 days | 7.5–9.0 | |
| Note: All by . Sources: Main: See template. | |||||||||
History

Aripiprazole was
It was approved by the US
Aripiprazole has been approved by the FDA for the treatment of both acute manic and mixed episodes, in people older than ten years.[119]
In 2006, the FDA required manufacturers to add a
In 2007, aripiprazole was approved by the FDA for the treatment of
In 2011 Otsuka and Lundbeck signed a collaboration to develop a depot formulation of aripiprazole.[122]
As of 2013, Abilify had annual sales of US$7 billion.[123] In 2013 BMS returned marketing rights to Otsuka, but kept manufacturing the drug.[124] Also in 2013, Otsuka and Lundbeck received US and European marketing approval for an injectable depot formulation of aripiprazole.[125][126]
Otsuka's US patent on aripiprazole expired on 20 October 2014, but due to a pediatric extension, a generic did not become available until 20 April 2015.[119] Barr Laboratories (now Teva Pharmaceuticals) initiated a patent challenge under the Hatch-Waxman Act in March 2007.[127] On 15 November 2010, this challenge was rejected by the U.S. District Court in New Jersey.[128]
Otsuka's European patent EP0367141 which would have expired on 26 October 2009, was extended by a
From April 2013 to March 2014, sales of Abilify amounted to almost $6.9 billion.[131]
In April 2015, the FDA announced the first generic versions.[132][133] In October 2015, aripiprazole lauroxil, a prodrug of aripiprazole that is administered via intramuscular injection once every four to six weeks for the treatment of schizophrenia, was approved by the FDA.[134][135]
In 2016, BMS settled cases with 42 US states that had charged BMS with off-label marketing to older people with dementia; BMS agreed to pay $19.5 million.[120][136]
In November 2017, the FDA approved Abilify MyCite, a digital pill containing a sensor intended to record when its consumer takes their medication.[137][138]
Aripiprazole was approved by the FDA for the treatment of bipolar disorder 1 and schizophrenia on April 27, 2023.[139][140]
In 2024, the European Commission approved apripiprazole for the maintenance treatment of schizophrenia.[141]
Society and culture
Legal status
| Regulatory administration (country)[142][143][144] | Schizophrenia | Acute mania | Bipolar maintenance | Major depressive disorder (as an adjunct) | Irritability in autism |
|---|---|---|---|---|---|
| Food and Drug Administration (US) | Yes | Yes | Yes (as an adjunct to lithium/valproate) | Yes | Yes (children and adolescents) |
| Therapeutic Goods Administration (AU) | Yes | Yes (as an adjunct to lithium/valproate) | Yes | No | No |
| Medicines and Healthcare products Regulatory Agency (UK) | Yes | Yes | Yes (to prevent mania) | No | No |
Classification
Aripiprazole has been described as the prototypical third-generation antipsychotic, as opposed to first-generation (typical) antipsychotics like haloperidol and second-generation (atypical) antipsychotics like clozapine.[145] It has received this classification due to its partial agonism of dopamine receptors, and is the first of its kind in this regard among antipsychotics, which before aripiprazole acted only as dopamine receptor antagonists.[145] The introduction of aripiprazole has led to a paradigm shift from a dopamine antagonist-based approach to a dopamine agonist-based approach for antipsychotic drug development.[145][146]
Brand names
- Abilify
- Aristada
- Arip MT
- Explemed
- Arivitae
Research
Attention deficit hyperactivity disorder
Aripiprazole was under development for the treatment of
Substance dependence
Aripiprazole has been studied for the treatment of amphetamine dependence and other substance use disorders, but more research is needed to support aripiprazole for these potential uses.[151][152][153][154] Available evidence of aripiprazole for amphetamine dependence is mixed.[151][152][153][154] Some studies have reported attenuation of the effects of amphetamines by aripiprazole, whereas other studies have reported both enhancement of the effects of amphetamines and increased use of amphetamines by aripiprazole.[151][152][153][154] As such, aripiprazole may not only be ineffective but potentially harmful for treatment of amphetamine dependence, and caution is warranted with regard to its use for such purposes.[151][152][153][154]
Other uses
Aripiprazole is under development for the treatment of
References
- ^ "Aripiprazole Use During Pregnancy". Drugs.com. 22 August 2019. Retrieved 7 February 2020.
- FDA. Retrieved 22 October 2023.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ a b c d e f g "Abilify (aripiprazole) tablet Abilify (aripiprazole) solution Abilify Discmelt (aripiprazole) tablet, orally disintegrating Abilify (aripiprazole) injection, solution [Otsuka America Pharmaceutical, Inc.]". DailyMed. Otsuka America Pharmaceutical, Inc. April 2013. Retrieved 22 October 2013.
- ^ a b c d e f g "Abilify Tablets, Orodispersible Tablets, Oral Solution – Summary of Product Characteristics (SPC)". electronic Medicines Compendium. Otsuka Pharmaceuticals (UK) Ltd. 20 September 2013. Archived from the original on 4 March 2016. Retrieved 22 October 2013.
- ^ a b c d e f g h "ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS" (PDF). European Medicines Agency. Otsuka Pharmaceutical Europe Ltd. Archived from the original (PDF) on 23 October 2013. Retrieved 22 October 2013.
- ^ a b c d e f g h i j k l m "Aripiprazole, ARIPiprazole Lauroxil Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 26 February 2019.
- ^ PMID 21833956.
- ^ a b c d e f g "Product Information for Abilify Aripiprazole Tablets & Orally Disintegrating Tablets". TGA eBusiness Services. Bristol-Myers Squibb Australia Pty Ltd. 1 November 2012. Retrieved 22 October 2013.
- ^ a b "Abilify Discmelt, Abilify Maintena (aripiprazole) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved 22 October 2013.
- ^ "Prescribing medicines in pregnancy database". Australian Government. 3 March 2014. Retrieved 22 April 2014.
- ISBN 978-0-85711-338-2.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Aripiprazole - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ a b c d e "Abilify- aripiprazole tablet Abilify- aripiprazole solution Abilify- aripiprazole tablet, orally disintegrating Abilify- aripiprazole injection, solution". DailyMed. Retrieved 20 October 2020.
- ^ "Psychosis and schizophrenia in children and young people: recognition and management | Guidance and guidelines | NICE". NICE. October 2016.
- ^ "Schizophrenia: aripiprazole prolonged-release suspension for injection | Guidance and guidelines | NICE". NICE. 24 July 2013.
- PMID 24385408.
- ^ "Levels of Evidence". Cochrane. Archived from the original on 23 September 2020. Retrieved 12 September 2019.
- ^ "Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis". The Lancet. 11 July 2019.
- S2CID 32085212.
- ^ S2CID 28750563.
- ^ S2CID 40089561.
- ^ "Bipolar disorder: assessment and management". Recommendations; Guidance and guidelines. UK National Institute for Health and Care Excellence (NICE). 24 September 2014.
1.1 Care for adults, children and young people across all phases of bipolar disorder
- PMID 24346956.
- S2CID 57249815.
- S2CID 21101054.
- S2CID 39065786.
- ^ "1 Recommendations - Bipolar disorder: assessment and management - Guidance". NICE. 24 September 2014. Retrieved 3 April 2023.
- ^ "Aripiprazole for treating moderate to severe manic episodes in adolescents with bipolar I disorder". NICE. May 2013. Retrieved 3 April 2023.
- ^ "The risks and benefits of highdose antipsychotic medication" (PDF). Royal College of Psychiatrists. 10 November 2014.
- ^ PMID 23554581.
- PMID 19687129.
- PMID 21154393.
- PMID 21739267.
- PMID 27747685.
- PMID 32845723.
- PMID 29955445.
- ^ PMID 27344135.
- ^ a b c d e f g "Aripiprazole". AdisInsight. Retrieved 21 September 2021.
- PMID 29154107.
- PMID 34244849.
- S2CID 13792422.
- PMID 29388585.
- S2CID 5353158.
- PMID 26220447.
- ^ PMID 25432131.
- PMID 25432131.
- PMID 17849776.
- PMID 19105842.
- S2CID 41312623.
- ^ "FDA Drug Safety Communication: FDA warns about new impulse-control problems associated with mental health drug aripiprazole (Abilify, Abilify Maintena, Aristada)". U.S. Food and Drug Administration (FDA). 9 February 2019. Retrieved 3 April 2023.
- ^ a b "Aripiprazole: MedlinePlus Drug Information". medlineplus.gov.
- ^ "DailyMed - ARIPIPRAZOLE- aripiprazole tablet". dailymed.nlm.nih.gov. Retrieved 4 November 2023.
BOXED WARNING WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS and SUICIDAL THOUGHTS AND BEHAVIORS WITH ANTIDEPRESSANT DRUGS Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Aripiprazole tablets are not approved for the treatment of patients with dementia-related psychosis [see WARNINGS AND PRECAUTIONS (5.1)]. Antidepressants increased the risk of suicidal thoughts and behavior in children, adolescents, and young adults in short-term studies. These studies did not show an increase in the risk of suicidal thoughts and behavior with antidepressant use in patients over age 24; there was a reduction in risk with antidepressant use in patients aged 65 and older [see WARNINGS AND PRECAUTIONS (5.3)]. In patients of all ages who are started on antidepressant therapy, monitor closely for worsening, and for emergence of suicidal thoughts and behaviors. Advise families and caregivers of the need for close observation and communication with the prescriber [see WARNINGS AND PRECAUTIONS (5. 3)].
- ^ a b "Aripiprazole (Abilify, Abilify Maintena, Aristada): Drug Safety Communication – FDA Warns About New Impulse-control Problems". U.S. Food and Drug Administration (FDA). 3 May 2016. Retrieved 4 May 2016.
- PMID 26658263.
- ISBN 978-0-85369-845-6.
Withdrawal of antipsychotic drugs after long-term therapy should always be gradual and closely monitored to avoid the risk of acute withdrawal syndromes or rapid relapse.
- ^ ISBN 978-0-19-852748-0.
- S2CID 6267180.
- ISBN 9788847026797.
- ISBN 978-0-9626523-7-0.
- PMID 25342720.
- ^ "Abilify (Aripiprazole) – Warnings and Precautions". DrugLib.com. 14 February 2007. Archived from the original on 4 December 2008. Retrieved 8 December 2008.
- PMID 20622942.
- ISBN 978-3-527-66445-0.
- ISBN 978-3-527-64564-0.
- S2CID 213824513.
- PMID 34703557.
- ISBN 9789811320927.
- PMID 26346901.
- ^ "Otsuka's Antipsychotic Abilify Is Approved in Japan, January 23, 2006|News Releases". Otsuka Pharmaceutical Co., Ltd. Retrieved 6 October 2022.
- PMID 33960741.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- ^ PMID 12784105.
- PMID 35145438.
- ^ S2CID 8654102.
- ^ PMID 12629531.
- S2CID 42636469.
- ^ a b c d e "Abilify (Aripiprazole) – Clinical Pharmacology". DrugLib.com. 14 February 2007. Retrieved 8 December 2008.
- ISBN 978-0-07-162442-8.
- ^ PMID 15592581.
- S2CID 19757151.
- PMID 28412910.
- ^ S2CID 42171115.
- ^ PMID 18418366.
- ^ PMID 12093598.
- ^ PMID 12063084.
- ^ PMID 17728427.
- ^ PMID 19909227.
- ^ "Aristada Initio (Aripiprazole Lauroxil Injectable Suspension): Uses, Dosage, Side Effects, Interactions, Warning". RxList. Retrieved 22 November 2022.
- S2CID 12919430.
- S2CID 31846731.
- S2CID 26101524.
- S2CID 33993650.
- PMID 15894311.
- S2CID 22568982.
- .
- PMID 16336943.
- PMID 10327430.
- PMID 26417330.
- S2CID 1891586.
- ^ Parent M, Toussaint C, Gilson H (1983). "Long-term treatment of chronic psychotics with bromperidol decanoate: clinical and pharmacokinetic evaluation". Current Therapeutic Research. 34 (1): 1–6.
- ^ PMID 6931472.
- ^ a b Reynolds JE (1993). "Anxiolytic sedatives, hypnotics and neuroleptics.". Martindale: The Extra Pharmacopoeia (30th ed.). London: Pharmaceutical Press. pp. 364–623.
- PMID 6143748.
- ^ PMID 444352.
- ^ Young D, Ereshefsky L, Saklad SR, Jann MW, Garcia N (1984). Explaining the pharmacokinetics of fluphenazine through computer simulations. (Abstract.). 19th Annual Midyear Clinical Meeting of the American Society of Hospital Pharmacists. Dallas, Texas.
- PMID 4992598.
- PMID 3545764.
- PMID 7185768.
- ^ Larsson M, Axelsson R, Forsman A (1984). "On the pharmacokinetics of perphenazine: a clinical study of perphenazine enanthate and decanoate". Current Therapeutic Research. 36 (6): 1071–88.
- ^ PMID 12790153.
- PMID 7616416.
- ^ "B-MS reveals Ph III aripiprazole data – Pharmaceutical industry news". The Pharma Letter. 17 May 2000.
- ^ "Abilify Product information". Union Register of medicinal products. 8 June 2004. Retrieved 1 October 2023.
- ^ Hitti M (20 November 2007). "FDA OKs Abilify for Depression". WebMD. Archived from the original on 5 December 2008. Retrieved 8 December 2008.
- ^ Keating G (23 November 2009). "FDA OKs Abilify for child autism irritability". Reuters. Retrieved 22 September 2010.
- ^ a b "Patent and Exclusivity Search Results". Electronic Orange Book. U.S. Food and Drug Administration (FDA). Retrieved 8 December 2008.
- ^ a b Mitchell M (8 December 2016). "Bristol-Myers Squibb Agrees to $19.5M Settlement Over Abilify Marketing". The Legal Intelligencer.
- ^ Staton T. "Pharma's Top 11 Marketing Settlements: Bristol-Myers Squibb – Abilify". FiercePharma. Retrieved 4 June 2017.
- ^ "Press Release: Lundbeck and Otsuka Pharmaceutical sign historic agreement to deliver innovative medicines targeting psychiatric disorders worldwide (OMX:LUN)". Lundbeck. 11 November 2011. Archived from the original on 1 April 2012. Retrieved 4 June 2017.
- ^ Megan Brooks (30 January 2014). "Top 100 Selling Drugs of 2013". Medscape. Retrieved 15 October 2015.
- ^ "BMS cuts salesforce on revised Abilify deal". PM Live. 7 November 2012.
- ^ Sagonowsky E (1 December 2016). "Lundbeck, Otsuka seek Abilify Maintena nod in bipolar disorder". FiercePharma.
- ^ "Abilify Maintena 300mg & 400mg powder and solvent for prolonged-release suspension for injection and suspension for injection in pre filled syringe – Summary of Product Characteristics (SPC)". UK Electronic Medicines Compendium. Retrieved 4 June 2017.
- ^ "Barr Confirms Filing an Application with a Paragraph IV Certification for Abilify Tablets" (Press release). Barr Pharmaceuticals, Inc. 20 March 2007. Retrieved 23 December 2008.
- ^ Decker S, Randall T (15 November 2010). "Bristol-Myers Partner Otsuka Wins Abilify Ruling – Bloomberg Business". Bloomberg L.P. Archived from the original on 22 July 2016. Retrieved 13 May 2015.
- ^ B1 EP application 0367141 B1, Oshiro Y, Sato S, Kurahashi N, "Carbostyril derivatives", published 1 October 1996, assigned to Otsuka Pharmaceutical Co., Ltd.
- ^ "Patent decision". UK Intellectual Property Office. 19 September 2006.
- ^ Michaelson J (9 November 2014). "Mother's Little Anti-Psychotic Is Worth $6.9 Billion A Year". The Daily Beast.
- ^ "FDA approves first generic Abilify to treat mental illnesses". Food and Drug Administration. Archived from the original on 1 May 2015. Retrieved 28 April 2015.
- ^ "Latest News | Teva Pharmaceuticals". www.tevapharm.com. 20 May 2023.
- S2CID 207208248.
- ^ "Aristada (aripiprazole lauroxil) FDA Approval History". Drugs.com. Retrieved 11 May 2018.
- ^ Staton T (14 December 2016). "Bristol-Myers to pay $19.5 million in Abilify off-label marketing settlement". FiercePharma.
- ^ "FDA approves pill with sensor that digitally tracks if patients have ingested their medication" (Press release). U.S. Food and Drug Administration (FDA). Retrieved 29 November 2017.
- ISSN 0362-4331. Retrieved 29 November 2017.
- ^ "Expert Q&A: Exploring a New Long-Acting Injectable for Bipolar I Disorder". bpHope.com. 8 May 2023. Retrieved 16 April 2024.
- ^ "FDA Approves Aripiprazole as First Once-Every-2-Months Long-Acting Injectable for Schizophrenia, Bipolar I Disorder". Pharmacy Times. 28 April 2023. Retrieved 16 April 2024.
- ^ "Otsuka and Lundbeck's schizophrenia treatment gains EC approval". Pharmaceutical Technology. 28 March 2024. Retrieved 16 April 2024.
- ^ Joint Formulary Committee. British National Formulary (BNF) 79. Pharmaceutical Pr; 2020.
- ^ "Australian Medicines Handbook 2013 [Internet]". Retrieved 20 September 2013.
- ^ Truven Health Analytics, Inc. DRUGDEX System (Internet) [cited 2013 Jun 25]. Greenwood Village, CO: Thomsen Healthcare; 2013.
- ^ PMID 28368577.
- ^ "Update on the Mechanism of Action of Aripiprazole: Translational Insights into Antipsychotic Strategies Beyond Dopamine Receptor Antagonism". NLM. 7 September 2015.
- ^ S2CID 24904076.
- ^ PMID 24141455.
- S2CID 33004517.
- PMID 18208634.
- ^ S2CID 37335642.
- ^ PMID 23104650.
- ^ PMID 20565449.
- ^ PMID 19042205.
Further reading
- Dean L (2016). "Aripiprazole Therapy and CYP2D6 Genotype". In Pratt VM, McLeod HL, Rubinstein WS, et al. (eds.). Medical Genetics Summaries. PMID 28520375. Bookshelf ID: NBK385288.
External links
- "Mechanism of Action of Aripiprazole". Psychopharmacology Institute.
