Aromatization
Aromatization is a chemical reaction in which an aromatic system is formed from a single nonaromatic precursor. Typically aromatization is achieved by dehydrogenation of existing cyclic compounds, illustrated by the conversion of cyclohexane into benzene. Aromatization includes the formation of heterocyclic systems.[1]

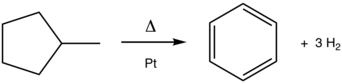
The conversion of methylcyclohexane to toluene is a classic aromatization reaction. This platinum (Pt)-catalyzed process is practiced on scale in the production of gasoline from petroleum.
Industrial practice
Although not practiced under the name, aromatization is a cornerstone of
Biochemical processes
Aromatization pathways
Oxidative dehydrogenation
For cyclohexane, cyclohexene, and cyclohexadiene, dehydrogenation is the conceptually simplest pathway for aromatization. The activation barrier decreases with the degree of unsaturation. Thus, cyclohexadienes are especially prone to aromatization. Formally, dehydrogenation is a redox process. Dehydrogenative aromatization is the reverse of arene hydrogenation. As such, hydrogenation catalysts are effective for the reverse reaction. Platinum-catalyzed dehydrogenations of cyclohexanes and related feedstocks are the largest scale applications of this reaction (see above).[1]
Sulfur and selenium are traditionally used in aromatization, the leaving group being hydrogen sulfide.[9]
Soluble transition metal complexes can induce oxidative aromatization concomitant with complexation.
Oxidative dehydrogenation of dihydropyridine results in aromatization, giving pyridine.[11]
Dehydration

Non-aromatic rings can be aromatized in many ways.
Tautomerization

The isomerization of cyclohexadienones gives the aromatic tautomer phenol.[13][14] Isomerization of 1,4-naphthalenediol at 200 °C produces a 2:1 mixture with its keto form, 1,4-dioxotetralin.[15]
Hydride and proton abstraction
Classically, aromatization reactions involve changing the C:H ratio of a substrate. When applied to cyclopentadiene, proton removal gives the aromatic conjugate base cyclopentadienyl anion, isolable as sodium cyclopentadienide:[16]
- 2 Na + 2 C5H6 → 2 NaC5H5 + H2
Aromatization can entail removal of hydride. Tropylium, C
7H+
7 arises by the aromatization reaction of cycloheptatriene with hydride acceptors.
- C
7H
8 + Br
2 → C
7H+
7 + Br−
+ HBr

From acyclic precursors
The aromatization of acyclic precursors is rarer in organic synthesis, although it is a significant component of the BTX production in refineries.
Among acyclic precursors, alkynes are relatively prone to aromatizations since they are partially dehydrogenated. The
See also
- Aromatase
- Aromatic hydrocarbon
References
- ^ ISBN 978-0-471-72091-1
- ISBN 0-8247-7150-8.
- .
- S2CID 11987113.
- ^ ISBN 9780080559629.
- ISBN 9783852001814.
- ISBN 9783774198463.
- PMID 5167256.
- PMID 20251415.
- ISBN 9780470132524.
- ISBN 3527306730.
- .
- ISBN 9780198503460.
- .
- PMID 16304647.
- ISBN 9780471199571.
- PMID 23600723.