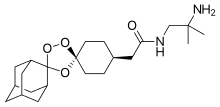
Arterolane
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Arterolane, also known as OZ277 or RBx 11160, is a substance that was tested for
antimalarial activity[1] by Ranbaxy Laboratories.[2] It was discovered by US and European scientists who were coordinated by the Medicines for Malaria Venture (MMV).[3] Its molecular structure is uncommon for pharmacological compounds in that it has both an ozonide (trioxolane) group and an adamantane substituent.[4]
Initial results were disappointing, and in 2007 MMV withdrew support, after having invested $20M in the research;[5] Ranbaxy said at the time that it intended to continue developing the drug combination on its own.[2] Ranbaxy started a Phase II clinical trial of arterolane, in combination with piperaquine in 2009 that published in 2015.[6][7]
In 2012, Ranbaxy obtained approval to market the arterolane/piperaquine combination drug in India, under the brand name Synriam,[5] and in 2014 received approval to market it in Nigeria, Uganda, Senegal, Cameroon, Guinea, Kenya and Ivory Coast; it had already received approval in Uganda.[8]
References
- PMID 19924861.
- ^ a b Unnikrishnan CH (21 September 2007). "Blow to Ranbaxy drug research plans". LiveMint.com. Archived from the original on 25 September 2007.
- S2CID 4320974.
- ^ Lowe D (23 November 2009). "Ozonides As Drugs: What Will They Think Of Next?". In the Pipeline. Sciencemag.org. Retrieved 17 November 2015.
- ^ a b Rathi A (3 May 2012). "Ranbaxy launches new anti-malarial Synriam". Chemistry World.
- ^ "Phase II trial of dispersible fixed dose combination of arterolane (RBx 11160) maleate and piperaquine phosphate in pediatric patients with acute uncomplicated Plasmodium falciparum malaria". India Clinical trials registry.
- PMID 26608469.
- ^ Staff (16 December 2014). "Ranbaxy receives approval for malaria drug Synriam from 7 African countries". Business Standard.
