Arthropod
Arthropods (
Arthropods use combinations of
The evolutionary ancestry of arthropods dates back to the
Etymology
The word arthropod comes from the
Terrestrial arthropods are often called bugs.
Description
Arthropods are
Diversity

Arthropoda is the largest animal phylum with the estimates of the number of arthropod species varying from 1,170,000 to 5 to 10 million and accounting for over 80 per cent of all known living animal species.[29][30] One arthropod sub-group, the insects, includes more described species than any other taxonomic class.[31] The total number of species remains difficult to determine. This is due to the census modeling assumptions projected onto other regions in order to scale up from counts at specific locations applied to the whole world. A study in 1992 estimated that there were 500,000 species of animals and plants in Costa Rica alone, of which 365,000 were arthropods.[31]
They are important members of marine, freshwater, land and air ecosystems and one of only two major animal groups that have adapted to life in dry environments; the other is amniotes, whose living members are reptiles, birds and mammals.[32] Both the smallest and largest arthropods are crustaceans. The smallest belong to the class Tantulocarida, some of which are less than 100 micrometres (0.0039 in) long.[33] The largest are species in the class Malacostraca, with the legs of the Japanese spider crab potentially spanning up to 4 metres (13 ft)[34] and the American lobster reaching weights over 20 kg (44 lbs).
Segmentation
The embryos of all arthropods are segmented, built from a series of repeated modules. The last common ancestor of living arthropods probably consisted of a series of undifferentiated segments, each with a pair of appendages that functioned as limbs. However, all known living and fossil arthropods have grouped segments into tagmata in which segments and their limbs are specialized in various ways.[32]
The three-part appearance of many insect bodies and the two-part appearance of spiders is a result of this grouping.[36] There are no external signs of segmentation in mites.[32] Arthropods also have two body elements that are not part of this serially repeated pattern of segments, an ocular somite at the front, where the mouth and eyes originated,[32][37] and a telson at the rear, behind the anus.
Originally it seems that each appendage-bearing segment had two separate pairs of appendages: an upper, unsegmented

The most conspicuous specialization of segments is in the head. The four major groups of arthropods –
Working out the evolutionary stages by which all these different combinations could have appeared is so difficult that it has long been known as "The arthropod head problem".[45] In 1960, R. E. Snodgrass even hoped it would not be solved, as he found trying to work out solutions to be fun.[Note 2]
Exoskeleton
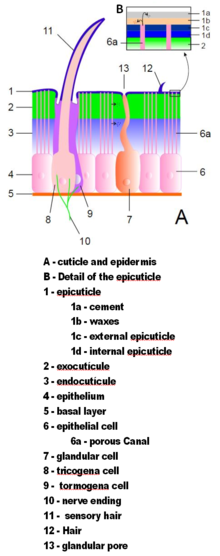
Arthropod exoskeletons are made of
The exoskeletons of most aquatic crustaceans are biomineralized with calcium carbonate extracted from the water. Some terrestrial crustaceans have developed means of storing the mineral, since on land they cannot rely on a steady supply of dissolved calcium carbonate.[48] Biomineralization generally affects the exocuticle and the outer part of the endocuticle.[47] Two recent hypotheses about the evolution of biomineralization in arthropods and other groups of animals propose that it provides tougher defensive armor,[49] and that it allows animals to grow larger and stronger by providing more rigid skeletons;[50] and in either case a mineral-organic composite exoskeleton is cheaper to build than an all-organic one of comparable strength.[50][51]
The cuticle may have
Although all arthropods use muscles attached to the inside of the exoskeleton to flex their limbs, some still use
Moulting

The exoskeleton cannot stretch and thus restricts growth. Arthropods, therefore, replace their exoskeletons by undergoing ecdysis (moulting), or shedding the old exoskeleton, the exuviae, after growing a new one that is not yet hardened. Moulting cycles run nearly continuously until an arthropod reaches full size. The developmental stages between each moult (ecdysis) until sexual maturity is reached is called an instar. Differences between instars can often be seen in altered body proportions, colors, patterns, changes in the number of body segments or head width. After moulting, i.e. shedding their exoskeleton, the juvenile arthropods continue in their life cycle until they either pupate or moult again.[54]
In the initial phase of moulting, the animal stops feeding and its epidermis releases moulting fluid, a mixture of
Because arthropods are unprotected and nearly immobilized until the new cuticle has hardened, they are in danger both of being trapped in the old cuticle and of being attacked by predators. Moulting may be responsible for 80 to 90% of all arthropod deaths.[54]
Internal organs
Arthropod bodies are also segmented internally, and the nervous, muscular, circulatory, and excretory systems have repeated components.
Respiration and circulation
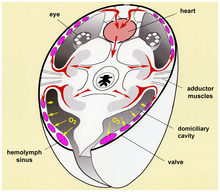
Arthropods have open
The heart is a muscular tube that runs just under the back and for most of the length of the hemocoel. It contracts in ripples that run from rear to front, pushing blood forwards. Sections not being squeezed by the heart muscle are expanded either by elastic ligaments or by small muscles, in either case connecting the heart to the body wall. Along the heart run a series of paired ostia, non-return valves that allow blood to enter the heart but prevent it from leaving before it reaches the front.[55]
Arthropods have a wide variety of respiratory systems. Small species often do not have any, since their high ratio of surface area to volume enables simple diffusion through the body surface to supply enough oxygen. Crustacea usually have gills that are modified appendages. Many arachnids have book lungs.[56] Tracheae, systems of branching tunnels that run from the openings in the body walls, deliver oxygen directly to individual cells in many insects, myriapods and arachnids.[57]
Nervous system

Living arthropods have paired main nerve cords running along their bodies below the gut, and in each segment the cords form a pair of
Excretory system
There are two different types of arthropod excretory systems. In aquatic arthropods, the end-product of biochemical reactions that
Senses

The stiff
Most arthropods lack balance and
The
Optical


Most arthropods have sophisticated visual systems that include one or more usually both of
Compound eyes consist of fifteen to several thousand independent
Olfaction
Reproduction and development
A few arthropods, such as
Most arthropods lay eggs,
Evolutionary history
Last common ancestor
Based on the distribution of shared
Fossil record

It has been proposed that the Ediacaran animals Parvancorina and Spriggina, from around 555 million years ago, were arthropods,[73][74][75] but later study shows that their affinities of being origin of arthropods are not reliable.[76] Small arthropods with bivalve-like shells have been found in Early Cambrian fossil beds dating 541 to 539 million years ago in China and Australia.[77][78][79][80] The earliest Cambrian trilobite fossils are about 520 million years old, but the class was already quite diverse and worldwide, suggesting that they had been around for quite some time.[81] In the Maotianshan shales, which date back to 518 million years ago, arthropods such as Kylinxia and Erratus have been found that seem to represent transitional fossils between stem (e.g. Radiodonta such as Anomalocaris) and true arthropods.[82][6][39] Re-examination in the 1970s of the Burgess Shale fossils from about 505 million years ago identified many arthropods, some of which could not be assigned to any of the well-known groups, and thus intensified the debate about the Cambrian explosion.[83][84][85] A fossil of Marrella from the Burgess Shale has provided the earliest clear evidence of moulting.[86]


The earliest fossil of likely
Arthropods provide the earliest identifiable fossils of land animals, from about 419 million years ago in the Late Silurian,[56] and terrestrial tracks from about 450 million years ago appear to have been made by arthropods.[92] Arthropods possessed attributes that were easy coopted for life on land; their existing jointed exoskeletons provided protection against desiccation, support against gravity and a means of locomotion that was not dependent on water.[93] Around the same time the aquatic, scorpion-like eurypterids became the largest ever arthropods, some as long as 2.5 m (8 ft 2 in).[94]
The oldest known
The oldest fossil record of
Evolutionary relationships to other animal phyla
From 1952 to 1977, zoologist
| ||||||||||||||||||||||||||||
Further analysis and discoveries in the 1990s reversed this view, and led to acceptance that arthropods are
A contrary view was presented in 2003, when Jan Bergström and
| Protostomes |
| |||||||||||||||||||||||||||||||||
Relationships of Ecdysozoa to each other and to annelids, etc.,[115][failed verification] including euthycarcinoids[116] |
||||||||||||||||||||||||||||||||||
Higher up the "family tree", the
In the 1990s,
If the Ecdysozoa hypothesis is correct, then segmentation of arthropods and annelids either has evolved convergently or has been inherited from a much older ancestor and subsequently lost in several other lineages, such as the non-arthropod members of the Ecdysozoa.[117][115]
Evolution of fossil arthropods
| Arthropod fossil phylogeny[118] | |||
| |||
| Summarized cladogram of the relationships between extinct arthropod groups. For more, see Deuteropoda. |
Aside from the four major living groups (
Modern interpretations of the basal, extinct
- The "Giant" or "Siberiid Lobopodians", such as Jianshanopodia, Siberion and Megadictyon, are the most basal grade in the total-group Arthropoda.
- The "Gilled Lobopodians", such as Kerygmachela, Pambdelurion and Opabinia, are the second most basal grade.
- The Radiodonta, which traditionally known as anomalocaridids come in third position, and are thought to be monophyletic.
- A possible "upper stem-group" assemblage of more uncertain position[118] but contained within Deuteropoda:[119] the Fuxianhuiida, Megacheira, and multiple "bivalved forms" including Isoxyida and Hymenocarina.
The
Controversies remain about the positions of various extinct arthropod groups. Some studies recover Megacheira as closely related to chelicerates, while others recover them as outside the group containing Chelicerate and Mandibulata as stem-group euarthropods.[120] The placement of the Artiopoda (which contains the extinct trilobites and similar forms) is also a frequent subject of dispute.[121] The main hypotheses position them in the clade Arachnomorpha with the Chelicerates. However, one of the newer hypotheses is that the chelicerae have originated from the same pair of appendages that evolved into antennae in the ancestors of Mandibulata, which would place trilobites, which had antennae, closer to Mandibulata than Chelicerata, in the clade Antennulata.[120][122] The fuxianhuiids, usually suggested to be stem-group arthropods, have been suggested to be Mandibulates in some recent studies.[120] The Hymenocarina, a group of bivalved arthropods, previously thought to have been stem-group members of the group, have been demonstrated to be mandibulates based on the presence of mandibles.[118]
- Radiodonts, Opabiniids, Gilled Lobopodians and the more traditional Lobopodians are all examples of basal stem-group arthropod lineages from the Cambrian
- Marrellomorphs, megacherians, funxianhuiids and phosphatocopines are some examples of Cambrian arthropods whose classification remains difficult
- Other examples of now extinct arthropod groups include
Evolution and classification of living arthropods
The phylum Arthropoda is typically
- trilobites.
- harvestmen, spiders, scorpions and related organisms characterized by the presence of chelicerae, appendages just above/in front of the mouthparts. Chelicerae appear in scorpions and horseshoe crabs as tiny claws that they use in feeding, but those of spiders have developed as fangs that inject venom.
- body segmentseach of which bearing one or two pairs of legs (or in a few cases being legless). All members are exclusively terrestrial.
- legs.
The phylogeny of the major extant arthropod groups has been an area of considerable interest and dispute.[124] Recent studies strongly suggest that Crustacea, as traditionally defined, is paraphyletic, with Hexapoda having evolved from within it,[125][126] so that Crustacea and Hexapoda form a clade, Pancrustacea. The position of Myriapoda, Chelicerata and Pancrustacea remains unclear as of April 2012[update]. In some studies, Myriapoda is grouped with Chelicerata (forming Myriochelata);[127][128] in other studies, Myriapoda is grouped with Pancrustacea (forming Mandibulata),[125] or Myriapoda may be sister to Chelicerata plus Pancrustacea.[126]
The following cladogram shows the internal relationships between all the living classes of arthropods as of the late 2010s,[129][130] as well as the estimated timing for some of the clades:[131]
| Arthropoda |
|
Entognaths | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Subphyla | Classes | Members | Example species |
|---|---|---|---|
| Chelicerata | Arachnida |
etc. | Araneae )
|
| Myriapoda | Chilopoda |
, etc. | Diplopoda, Spirostreptida )
|
Crustacea |
) | ||
| Hexapoda | Insecta Entognatha |
springtails , etc. |
Insecta, Lepidoptera )
|
Interaction with humans

However, the greatest contribution of arthropods to human food supply is by pollination: a 2008 study examined the 100 crops that FAO lists as grown for food, and estimated pollination's economic value as €153 billion, or 9.5 per cent of the value of world agricultural production used for human food in 2005.[144] Besides pollinating, bees produce honey, which is the basis of a rapidly growing industry and international trade.[145]
The red dye
The relative simplicity of the arthropods' body plan, allowing them to move on a variety of surfaces both on land and in water, have made them useful as models for
| Disease[154] | Insect | Cases per year | Deaths per year |
|---|---|---|---|
| Malaria | Anopheles mosquito | 267 M | 1 to 2 M |
| Dengue fever | Aedes mosquito | ? | ? |
| Yellow fever | Aedes mosquito | 4,432 | 1,177 |
| Filariasis | Culex mosquito | 250 M | unknown |
Although arthropods are the most numerous phylum on Earth, and thousands of arthropod species are venomous, they inflict relatively few serious bites and stings on humans. Far more serious are the effects on humans of diseases like
Many species of arthropods, principally insects but also mites, are agricultural and forest pests.
See also
- Dorsal lobe
- Invertebrate paleontology
- Minibeasts
Notes
- ^ "It would be too bad if the question of head segmentation ever should be finally settled; it has been for so long such fertile ground for theorizing that arthropodists would miss it as a field for mental exercise."[46]
- ^ The fossil was originally named Eotarbus but was renamed when it was realized that a Carboniferous arachnid had already been named Eotarbus.[96]
- ^ For a mention of insect contamination in an international food quality standard, see sections 3.1.2 and 3.1.3 of Codex 152 of 1985 of the Codex Alimentarius[140]
- ^ For examples of quantified acceptable insect contamination levels in food see the last entry (on "Wheat Flour") and the definition of "Extraneous material" in Codex Alimentarius,[141] and the standards published by the FDA.[142]
References
- ^ S2CID 258497632.
- ^ Gravenhorst, J. L. C. (1843). Vergleichende Zoologie. Breslau: Druck und Verlag von Graß, Barth und Comp.
- PMID 30963877.
- PMID 35125000.
- S2CID 246779634.
- ^ S2CID 226248177. Retrieved 8 December 2020.
- PMID 24077329.
- S2CID 252839113.
- .
- ^ Peel, J.S.; Stein, M. "A new Arthropod from the Lower Cambrian Sirius Passet Fossil-Lagerstätten of North Greenland" (PDF). Bulletin of Geosciences. 84 (4): 1158.
- PMID 28957518.
- (PDF) from the original on 2 December 2013, retrieved 11 June 2012
- S2CID 247123967.
- .
- S2CID 249652930.
- ISSN 1802-8225.
- S2CID 85744103.
- .
- ^ "Arthropoda". Online Etymology Dictionary. Archived from the original on 7 March 2013. Retrieved 23 May 2013.
- ^ Gravenhorst, J. L. C. (1843). Vergleichende Zoologie [Comparative Zoology] (in German). Breslau, (Prussia): Graß, Barth & Comp. p. foldout.
"Mit gegliederten Bewegungsorganen" (with articulated movement organs)
- ^ "What is a bug? Insects, arachnids, and myriapods" at Museum of New Zealand Te Papa Tongarewa website. Accessed 10 March 2022.
- ISBN 978-1-57859-049-0
- ^ ISBN 978-1-57859-049-0
- ISBN 978-0-226-84548-7
- S2CID 84995596
- ^ Australian Beetles Volume 2: Archostemata, Myxophaga, Adephaga, Polyphaga
- S2CID 23119386.
- ^ Amorphous calcium phosphate in the pupal cuticle of Bactrocera dorsalis Hendel (Diptera: Tephritidae): A new discovery for reconsidering the mineralization of the insect cuticle
- ^ Thanukos, Anna, The Arthropod Story, University of California, Berkeley, archived from the original on 16 June 2008, retrieved 29 September 2008
- (PDF) from the original on 26 December 2010, retrieved 6 May 2010
- ^ ISBN 978-0-226-79760-1
- ^ a b c d e f g h i j k l Ruppert, Fox & Barnes (2004), pp. 518–522
- S2CID 7325858.
- ISBN 978-0-521-31987-4
- ^ PMID 34330912.
- ^ a b Gould (1990), pp. 102–106.
- ^ PMID 27989966.
- ^ "Giant sea creature hints at early arthropod evolution". 11 March 2015. Archived from the original on 2 February 2017. Retrieved 22 January 2017.
- ^ S2CID 246608509.
- S2CID 22426697.
- PMID 18252674.
- ISBN 978-0-226-28497-2
- PMID 27240897.
- ^ Geological Survey of Canada Bulletin, 209: 1–24 Summarised in Gould (1990), pp. 107–121.
- S2CID 4310080.
- ^ Snodgrass, R. E. (1960), "Facts and theories concerning the insect head", Smithsonian Miscellaneous Collections, 142: 1–61
- ^ ISBN 978-0-691-08308-7.
- ISBN 978-0-19-504977-0
- OCLC 156823511
- ^ (PDF) from the original on 3 October 2008, retrieved 25 September 2008
- doi:10.1017/S1089332600002345. Archived from the original(PDF) on 3 October 2008.
- ISBN 978-0-632-04761-1
- (PDF) from the original on 3 October 2008, retrieved 25 September 2008
- ^ a b c Ruppert, Fox & Barnes (2004), pp. 523–524
- ^ a b c Ruppert, Fox & Barnes (2004), pp. 527–528
- ^ .
- ^ Ruppert, Fox & Barnes (2004), pp. 530, 733
- ^ Ruppert, Fox & Barnes (2004), pp. 531–532
- ^ a b c d Ruppert, Fox & Barnes (2004), pp. 529–530
- ^ a b c d e f g Ruppert, Fox & Barnes (2004), pp. 532–537
- ^ Ruppert, Fox & Barnes (2004), pp. 578–580
- doi:10.1016/S0167-9317(03)00102-3. Archived from the original(PDF) on 1 October 2008.
- ^ a b c d Ruppert, Fox & Barnes (2004), pp. 537–539
- ISBN 978-0-470-01617-6.
- S2CID 11617147.
- PMID 3324702.
- ^ "Facts About Horseshoe Crabs and FAQ". Retrieved 19 January 2020.
- ISBN 978-87-7934-001-5, archived(PDF) from the original on 3 October 2008, retrieved 28 September 2008
- (PDF) from the original on 3 October 2008. Retrieved 28 September 2008.
- ^ Smith, G., Diversity and Adaptations of the Aquatic Insects (PDF), New College of Florida, archived from the original (PDF) on 3 October 2008, retrieved 28 September 2008
- ^ ISBN 978-0-8493-3498-6
- ^ McKeever, Conor (30 September 2016). "Arthropod ancestor had the mouth of a penis worm". Natural History Museum. Archived from the original on 2 February 2017.
- Transactions of the Royal Society of South Australia. 81: 185–188. Archived from the original(PDF) on 16 December 2008.
- S2CID 85821717.
- ^ McMenamin, M.A.S (2003), "Spriggina is a trilobitoid ecdysozoan" (abstract), Abstracts with Programs, 35 (6): 105, archived from the original on 30 August 2008, retrieved 21 October 2008
- PMID 29784780.
- S2CID 129651908. Archived from the original(PDF) on 18 July 2011.
- .
- ISSN 0810-8889
- from the original on 19 October 2008, retrieved 21 October 2008
- ^ a b "A 520-million-year-old, five-eyed fossil reveals arthropod origin". phys.org. Retrieved 8 December 2020.
- ^ Whittington, H. B. (1979). Early arthropods, their appendages and relationships. In M. R. House (Ed.), The origin of major invertebrate groups (pp. 253–268). The Systematics Association Special Volume, 12. London: Academic Press.
- OCLC 15630217
- ^ Gould (1990), p. [page needed].
- S2CID 40015864.
- ^ . Retrieved 5 January 2024.
- ISSN 0036-8075.
- PMID 11739918
- ISSN 0031-0239.
- S2CID 4329196.
- PMID 14731304.
- ISBN 978-0-632-04444-3.
- PMID 18029297.
- ^ Dunlop, J. A. (September 1996). "A trigonotarbid arachnid from the Upper Silurian of Shropshire" (PDF). Palaeontology. 39 (3): 605–614. Archived from the original (PDF) on 16 December 2008.
- S2CID 83825904.
- PMID 19104044.
- S2CID 26323977.
- doi:10.1146/annurev.ecolsys.37.091305.110221. Archived from the original(PDF) on 9 December 2008.
- ISSN 0031-0239.
- S2CID 4327169.
- ISSN 1618-1077.
- ISSN 0031-0220.
- S2CID 4431205.
- ^ PMID 28584727.
- Witwatersrand University Press. Archived from the original(PDF) on 11 September 2008. Retrieved 21 October 2008.
- ^ .
- ISBN 978-0-306-44967-3.
- ^ Adrain, J. (15 March 1999). "Arthropod Fossils and Phylogeny, edited by Gregory D. Edgecomb". Book review. Palaeontologia Electronica. Archived from the original on 8 September 2008. Retrieved 28 September 2008.
- The book is
JSTOR 3515467. - S2CID 32142337.
- S2CID 4341971.
- ISBN 978-0-19-850681-2.
- S2CID 83582128.
- Cambridge University. 17 August 2014. Archivedfrom the original on 7 January 2017. Retrieved 24 January 2017.
- ^ PMID 18192181.
- S2CID 4419235.
- S2CID 46920478.
- ^ S2CID 225478171.
- ^ S2CID 7751936.
- ^ S2CID 243269510.
- PMID 21672726.
- ^ Dunlop, Jason A. (31 January 2011). "Fossil Focus: Chelicerata". Palaeontology Online. pp. 1–8. Archived from the original on 12 September 2017. Retrieved 15 March 2018.
- ^ "Arthropoda". Integrated Taxonomic Information System. Retrieved 15 August 2006.
- PMID 17767736.
- ^ S2CID 4427443.
- ^ PMID 22049065.
- (PDF) from the original on 10 January 2011. Retrieved 16 April 2010.
- ISBN 978-0-8493-3498-6. Archived(PDF) from the original on 16 September 2006. Retrieved 23 August 2006.
- PMID 31270537.
- S2CID 189926344.
- S2CID 36008925.
- ISBN 978-0-632-05464-0. Archivedfrom the original on 6 December 2008. Retrieved 3 October 2008.
- ^ Bailey, S., Bugfood II: Insects as Food!?!, University of Kentucky Department of Entomology, archived from the original on 16 December 2008, retrieved 3 October 2008
- ^ Unger, L., Bugfood III: Insect Snacks from Around the World, University of Kentucky Department of Entomology, archived from the original on 10 October 2008, retrieved 3 October 2008
- Sunday Telegraph, archivedfrom the original on 18 July 2009, retrieved 24 August 2009
- ^ "Spiderwomen serve up Cambodia's creepy caviar", ABC News Online, 2 September 2002, archived from the original on 3 June 2008, retrieved 24 August 2009
- ISBN 978-1-74059-111-9.
- ISBN 978-0-452-28700-6, archived from the originalon 11 May 2011, retrieved 3 October 2008
- ^ Taylor, R. L. (1975), Butterflies in My Stomach (or: Insects in Human Nutrition), Woodbridge Press Publishing Company, Santa Barbara, California
- ^ Codex commission for food hygiene (1985), "Codex Standard 152 of 1985 (on "Wheat Flour")" (PDF), Codex Alimentarius, Food and Agriculture Organization, archived (PDF) from the original on 31 December 2010, retrieved 8 May 2010.
- ^ "Complete list of Official Standards", Codex Alimentarius, Food and Agriculture Organization, archived from the original on 31 January 2010, retrieved 8 May 2010
- ^ The Food Defect Action Levels, U. S. Food and Drug Administration, archived from the original on 18 December 2006, retrieved 16 December 2006
- ISBN 978-1-57808-339-8
- from the original on 3 December 2008. Retrieved 3 October 2008.
- ^ Apiservices — International honey market — World honey production, imports & exports, archived from the original on 6 December 2008, retrieved 3 October 2008
- ^ Time line of fabrics, Threads In Tyme, LTD, archived from the original on 28 October 2005, retrieved 14 July 2005
- ^ Jeff Behan, The bug that changed history, archived from the original on 21 June 2006, retrieved 26 June 2006
- ^ Canary Islands cochineal producers homepage, archived from the original on 24 June 2005, retrieved 14 July 2005
- ^ Heard, W., Coast (PDF), University of South Florida, archived from the original (PDF) on 19 February 2017, retrieved 25 August 2008
- ISBN 978-0-8493-8120-1
- PMID 20957283.
- S2CID 21564918. Archived from the original(PDF) on 10 March 2012.
- ISBN 978-4-431-24164-5
- ^ ISBN 978-0-412-49800-8
- ISBN 978-1-55581-238-6, retrieved 29 March 2010
- ^ Potter, M. F., Parasitic Mites of Humans, University of Kentucky College of Agriculture, archived from the original on 8 January 2009, retrieved 25 October 2008
- ^ Klenerman, Paul; Lipworth, Brian, House dust mite allergy, NetDoctor, archived from the original on 11 February 2008, retrieved 20 February 2008
- ^ ISBN 978-0-471-24483-7
- ^ Gorham, J. Richard (1991), "Insect and Mite Pests in Food: An Illustrated Key" (PDF), Agriculture Handbook Number 655, United States Department of Agriculture, pp. 1–767, archived from the original (PDF) on 25 October 2007, retrieved 6 May 2010
- .
- ISBN 978-0-471-58957-0
- ISBN 978-0-470-01617-6
- S2CID 10823576
Bibliography
- ISBN 978-0-09-174271-3.
- Ruppert, E. E.; R. S. Fox; R. D. Barnes (2004). Invertebrate Zoology (7th ed.). ISBN 978-0-03-025982-1.
External links
- "Arthropod" at the Encyclopedia of Life
- Venomous Arthropods chapter in United States Environmental Protection Agency and University of Florida/Institute of Food and Agricultural Sciences National Public Health Pesticide Applicator Training Manual
- Arthropods – Arthropoda Insect Life Forms