Aryl hydrocarbon receptor
Ensembl | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) | |||||||||
| Location (UCSC) | Chr 7: 16.92 – 17.35 Mb | Chr 12: 35.55 – 35.59 Mb | |||||||
| PubMed search | [3] | [4] | |||||||
| View/Edit Human | View/Edit Mouse |
The aryl hydrocarbon receptor (also known as AhR, AHR, ahr, ahR, AH receptor, or dioxin receptor) is a
More recently, it has been discovered that AhR is activated (or deactivated) by a number of
The aryl hydrocarbon receptor is a member of the family of
Protein functional domains

The AhR
Ligands
AhR ligands have been generally classified into two categories, synthetic or naturally occurring. The first ligands to be discovered were synthetic and members of the halogenated aromatic hydrocarbons (
Research has focused on naturally occurring compounds with the hope of identifying an endogenous ligand. Naturally occurring compounds that have been identified as ligands of Ahr include derivatives of tryptophan such as indigo dye and indirubin,[18] tetrapyrroles such as bilirubin,[19] the arachidonic acid metabolites lipoxin A4 and prostaglandin G,[20] modified low-density lipoprotein[21] and several dietary carotenoids.[16] One assumption made in the search for an endogenous ligand is that the ligand will be a receptor agonist. However, work by Savouret et al. has shown this may not be the case since their findings demonstrate that 7-ketocholesterol competitively inhibits Ahr signal transduction.[22]
Carbidopa is a selective aryl hydrocarbon receptor modulator (SAhRM).[23] Other SAhRMs include microbial-derived 1,4-dihydroxy-2-napthoic acid[24] and plant-derived 3,3-diindolylmethane.[25]
Indolocarbazole (ICZ) is one of the strongest non-halogenated agonists for AhR in vitro reported.[26]
Ligand-independent AhR activity can be seen in mammalian AhR. The mammalian AhR needs no exogenous ligand-dependent activation to be functional, and this appears to be the case for its role in the regulation of the expression of some transforming growth factor-beta (TGF-b) isoforms. This is not to say that ligand-dependent AhR activation is not needed for the AhR to function in those cases, but that, if a ligand is needed, it is provided endogenously by the cells or tissues in question and its identity is unknown.[27]
Signaling pathway
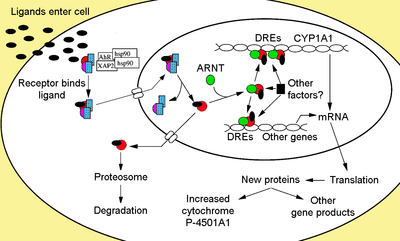

Cytosolic complex
Non-ligand bound AhR is retained in the
Transforming growth factor-beta (TGF-β) signaling pathway
TGF-β cytokines are members of a signaling protein family that includes activin, Nodal subfamily, bone morphogenetic proteins, growth and differentiation factors, and Müllerian inhibitor subfamily. TGF-β signaling plays an important role in cell physiology and development by inhibiting cell proliferation, promoting apoptosis, inducing differentiation, and determining developmental fate in vertebrates and invertebrates.[44] TGF-β activators include proteases such as plasmin, cathepsins, and calpains. Thrombospondin 1, a glycoprotein that inhibits angiogenesis, and matrix metalloproteinase 2 (MMP-2). The extracellular matrix itself appears to play an important regulatory role in TGF-β signaling.[45][46]
Receptor activation
Upon ligand binding to AhR, AIP is released resulting in exposure of the NLS, which is located in the bHLH region,[47] leading to import into the nucleus.[48] It is presumed that once in the nucleus, Hsp90 dissociates exposing the two PAS domains allowing the binding of ARNT.[40][49][50][51] The activated AhR/ARNT heterodimer complex is then capable of either directly or indirectly interacting with DNA by binding to recognition sequences located in the 5’- regulatory region of dioxin-responsive genes.[40][50][52]
DNA binding (xenobiotic response element – XRE)
The classical recognition motif of the AhR/ARNT complex, referred to as either the AhR-, dioxin- or xenobiotic- responsive element (AHRE, DRE or XRE), contains the core sequence 5'-GCGTG-3'
Functional role in physiology and toxicology
Role in development
In terms of evolution, the oldest physiological role of AhR is in development. AhR is presumed to have evolved from
Adaptive and innate response
The adaptive response is manifested as the induction of xenobiotic metabolizing enzymes. Evidence of this response was first observed from the induction of cytochrome P450, family 1, subfamily A, polypeptide 1 (Cyp1a1) resultant from TCDD exposure, which was determined to be directly related to activation of the AhR signaling pathway.[74][75][76] The search for other metabolizing genes induced by AhR ligands, due to the presence of DREs, has led to the identification of an "AhR gene battery" of Phase I and Phase II metabolizing enzymes consisting of CYP1A1, CYP1A2, CYP1B1, NQO1, ALDH3A1, UGT1A2 and GSTA1.[77] Presumably, vertebrates have this function to be able to detect a wide range of chemicals, indicated by the wide range of substrates AhR is able to bind and facilitate their biotransformation and elimination. The AhR may also signal the presence of toxic chemicals in food and cause aversion of such foods.[78]
AhR activation seems to be also important for immunological responses and inhibiting inflammation
Toxic response
Extensions of the adaptive response are the toxic responses elicited by AhR activation. Toxicity results from two different ways of AhR signaling. The first is a side effect of the adaptive response in which the induction of metabolizing enzymes results in the production of toxic metabolites. For example, the polycyclic aromatic hydrocarbon benzo[a]pyrene (BaP), a ligand for AhR, induces its own metabolism and bioactivation to a toxic metabolite via the induction of CYP1A1 and CYP1B1 in several tissues.[82] The second approach to toxicity is the result of aberrant changes in global gene transcription beyond those observed in the "AhR gene battery." These global changes in gene expression lead to adverse changes in cellular processes and function.[83] Microarray analysis has proved most beneficial in understanding and characterizing this response.[64][84][85][86]
Xenobiotic metabolizing enzymes help with the metabolic process by transforming and the excretion of chemicals. The most potent inducer of CYP1A1 is 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). In addition, TCDD induces a broad spectrum of biochemical and toxic effects, such as teratogenesis, immunosuppression and tumor promotion. Most, if not all, of the effects caused by TCDD and other PAHs are known to be mediated by AhR which has a high binding affinity to TCDD.[44]
Protein-protein interactions
In addition to the protein interactions mentioned above, AhR has also been shown to
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000106546 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000019256 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- PMID 26530806.
- PMID 27980293.
- PMID 29343438.
- PMID 1325649.
- PMID 7493958.
- PMID 15186484.
- PMID 1314586.
- ^ PMID 7559670.
- PMID 17227672.
- PMID 11551916.
- S2CID 29379967.
- ^ PMID 12540743.
- PMID 31721255.
- PMID 11425848.
- PMID 9380021.
- S2CID 21953408.
- PMID 17227852.
- PMID 11042205.
- PMID 29109131.
- PMID 27837168.
- PMID 22592002.
- PMID 19186062.
- S2CID 29379967.
- PMID 2844180.
- PMID 2843537.
- PMID 15270073.
- ^ PMID 10224120.
- PMID 11259606.
- ^ PMID 12623125.
- PMID 9447995.
- PMID 9083006.
- ^ PMID 25911105.
- PMID 9111057.
- PMID 7982913.
- PMID 1320028.
- ^ PMID 8384309.
- PMID 9837941.
- PMID 10986286.
- PMID 12431985.
- ^ PMID 12573486.
- PMID 15627472.
- PMID 18817753.
- PMID 9446600.
- PMID 10913191.
- PMID 1852076.
- ^ PMID 8396713.
- S2CID 34075046.
- PMID 8397410.
- PMID 1313023.
- PMID 8384216.
- PMID 1318077.
- PMID 7821222.
- PMID 7700240.
- PMID 7592839.
- PMID 15358164.
- PMID 15144902.
- PMID 16902966.
- PMID 9573046.
- PMID 10433921.
- ^ S2CID 1913812.
- PMID 19896476.
- S2CID 206537957.
- ^ S2CID 16090460.
- S2CID 4384276.
- PMID 16956419.
- PMID 16301529.
- PMID 20688981.
- PMID 21226706.
- PMID 34589904.
- PMID 6885786.
- PMID 6715350.
- PMID 8524325.
- PMID 10605936.
- PMID 21458548.
- PMID 21600206.
- PMID 24514067.
- PMID 26416282.
- PMID 14691214.
- S2CID 34036181.
- PMID 12377990.
- PMID 15598615.
- PMID 16984957.
- PMID 9079689.
- PMID 12917420.
- PMID 12612060.
- PMID 10620335.
- PMID 12024042.
- PMID 12215427.
- PMID 10428779.
- PMID 11114727.
- S2CID 2376236.
- PMID 17823304.
- PMID 9712901.
External links
- Aryl+hydrocarbon+receptor at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Human AHR genome location and AHR gene details page in the UCSC Genome Browser.
- Human ARNT genome location and ARNT gene details page in the UCSC Genome Browser.
