Asparagine
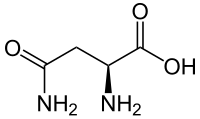 Skeletal formula of L-asparagine
| |||

| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Asparagine
| |||
| Other names
2-Amino-3-carbamoylpropanoic acid
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
ECHA InfoCard
|
100.019.565 | ||
| EC Number |
| ||
IUPHAR/BPS |
|||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H8N2O3 | |||
| Molar mass | 132.119 g·mol−1 | ||
| Appearance | white crystals | ||
| Density | 1.543 g/cm3 | ||
| Melting point | 234 °C (453 °F; 507 K) | ||
| Boiling point | 438 °C (820 °F; 711 K) | ||
| 2.94 g/100 mL | |||
| Solubility | soluble in | ||
| log P | −3.82 | ||
| Acidity (pKa) |
| ||
| -69.5·10−6 cm3/mol | |||
| Structure | |||
| orthorhombic | |||
| Thermochemistry | |||
Std enthalpy of (ΔfH⦵298)formation |
−789.4 kJ/mol | ||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 219 °C (426 °F; 492 K) | ||
| Safety data sheet (SDS) | Sigma-Alrich | ||
| Supplementary data page | |||
| Asparagine (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Asparagine (symbol Asn or N
The one-letter symbol N for asparagine was assigned arbitrarily,[3] with the proposed mnemonic asparagiNe;[4]
History
Asparagine was first isolated in 1806 in a crystalline form by French chemists Louis Nicolas Vauquelin and Pierre Jean Robiquet (then a young assistant). It was isolated from asparagus juice,[5][6] in which it is abundant, hence the chosen name. It was the first amino acid to be isolated.[7]
Three years later, in 1809, Pierre Jean Robiquet identified a substance from
The determination of asparagine's structure required decades of research. The empirical formula for asparagine was first determined in 1833 by the French chemists Antoine François Boutron Charlard and Théophile-Jules Pelouze; in the same year, the German chemist Justus Liebig provided a more accurate formula.[11][12] In 1846 the Italian chemist Raffaele Piria treated asparagine with nitrous acid, which removed the molecule's amine (–NH2) groups and transformed asparagine into malic acid.[13] This revealed the molecule's fundamental structure: a chain of four carbon atoms. Piria thought that asparagine was a diamide of malic acid;[14] however, in 1862 the German chemist Hermann Kolbe showed that this surmise was wrong; instead, Kolbe concluded that asparagine was an amide of an amine of succinic acid.[15] In 1886, the Italian chemist Arnaldo Piutti (1857–1928) discovered a mirror image or "enantiomer" of the natural form of asparagine, which shared many of asparagine's properties, but which also differed from it.[16] Since the structure of asparagine was still not fully known – the location of the amine group within the molecule was still not settled[17] – Piutti synthesized asparagine and thus published its true structure in 1888.[18]
Structural function in proteins
Since the asparagine side-chain can form hydrogen bond interactions with the peptide backbone, asparagine residues are often found near the beginning of
Asparagine also provides key sites for N-linked glycosylation, modification of the protein chain with the addition of carbohydrate chains. Typically, a carbohydrate tree can solely be added to an asparagine residue if the latter is flanked on the C side by X-serine or X-threonine, where X is any amino acid with the exception of proline.[19]
Asparagine can be hydroxylated in the HIF1
Sources
Dietary sources
Asparagine is not essential for humans, which means that it can be synthesized from central metabolic pathway intermediates and is not required in the diet.
Asparagine is found in:
- Animal sources: ]
- Plant sources: soy protein isolate, tofu[citation needed]
Biosynthesis and catabolism
The precursor to asparagine is

In reaction that is the reverse of its biosynthesis, asparagine is hydrolyzed to aspartate by asparaginase. Aspartate then undergoes transamination to form glutamate and oxaloacetate from alpha-ketoglutarate. Oxaloacetate, which enters the citric acid cycle (Krebs cycle).[21]
Acrylamide controversy
Heating a mixture of asparagine and
Function
Asparagine synthetase is required for normal development of the brain.
The addition of
References
- ISBN 978-1498754286.
- ^ "Nomenclature and Symbolism for Amino Acids and Peptides". IUPAC-IUB Joint Commission on Biochemical Nomenclature. 1983. Archived from the original on 9 October 2008. Retrieved 5 March 2018.
- .
- .
- .
- ^ Plimmer RH (1912) [1908]. Plimmer RH, Hopkins FG (eds.). The chemical composition of the proteins. Monographs on biochemistry. Vol. Part I. Analysis (2nd ed.). London: Longmans, Green and Co. p. 112. Retrieved January 18, 2010.
- ISBN 978-0-12-034226-6.
- ^ Robiquet PJ (1809). "Analyse de la racine de réglisse" [Analysis of licorice root]. Annales de Chimie et de Physique (in French). 72 (1): 143–159.
- ^ Plisson A (1828). "De l'indentité de l'asparagine avec l'agédoïte" [On the identity of asparagine with agédoïte]. Journal de Pharmacie et des Sciences Accessoires (in French). 14 (4): 177–182.
- ^ Felter HW, Lloyd JU (1898). "Glycyrrhiza (U. S. P.)—Glycyrrhiza". King's American Dispensatory. Henriette's Herbal Homepage.
- . The empirical formula of asparagine appears on p. 80.
- . The empirical formula appears on p. 149 ; the formula is correct if the subscripts are divided by 2.
- ^ See:
- Piria R (January 1846). "Studi sulla costituzione chimica dell' asparagina e dell' acido aspartico" [Studies of the chemical constitution of asparagine and aspartic acid]. Il Cimento (in Italian). 4: 55–73. S2CID 177614807.
- French translation: Piria R (1848). "Recherches sur la constitution chimique de l'asparagine et de l'acide aspartique" [Investigations into the chemical constitution of asparagine and of aspartic acid]. Annales de Chimie et de Physique. 3rd series (in French). 22: 160–179. From p. 175: " … on voit, en outre, que l'asparagine et l'acide aspartique lui-même se décomposent avec une facilité remarquable, sous l'influence de l'acide hyponitrique, en fournissant du gaz azote et de l'acide malique." ( … one sees, in addition, that asparagine and aspartic acid itself are decomposed with a remarkable ease under the influence of nitrous acid, rendering nitrogen gas and malic acid.)
- Piria R (January 1846). "Studi sulla costituzione chimica dell' asparagina e dell' acido aspartico" [Studies of the chemical constitution of asparagine and aspartic acid]. Il Cimento (in Italian). 4: 55–73.
- ^ Plimmer RH (1912). The Chemical Constitution of the Proteins. Part I: Analysis (2nd ed.). London, England: Longmans, Green and Co. p. 112.
- .
- .
- ^ The French chemist Edouard Grimaux thought that the amine group (–NH2) was located next to the amide group (–C(O)NH2), whereas the Italian chemist Icilio Guareschi thought that the amine group was located next to the carboxyl group (–COOH).
- Grimaux E (1875). "Recherches synthétiques sur le groupe urique" [Synthetic investigations of the uric group]. Bulletin de la Société Chimique de Paris. 2nd series (in French). 24: 337–355. On p. 352, Grimaux presented two putative structures for asparagine, and on p. 353, he favored structure (I.), which is incorrect. From p. 353: " … ce sont les formules marquées du chiffre I qui me semblent devoir être adoptées pour l'asparagine, … " ( … it is the formulas marked by the figure I which, it seems to me, should be adopted for asparagine, … )
- Guareschi I (1876). "Studi sull' asparagine e sull' acido aspartico" [Studies of asparagine and of aspartic acid]. Atti della Reale Academia del Lincei. 2nd series (in Italian). 3 (pt. 2): 378–393. On p. 388, Guareschi proposed two structures (α and β) for asparagine; he favored α, the correct one. From p. 388: "La formola α mi sembra preferibile per seguente ragione: … " (The formula α seems preferable to me for the following reason: … )
- English abstract in: Guareschi J (1877). "Asparagine and aspartic acid". Journal of the Chemical Society. 31: 457–459. See especially p. 458.
- ^ Piutti A (1888). "Sintesi e costituzione delle asparagine" [Synthesis and constitution of asparagine]. Gazzetta Chimica Italiana (in Italian). 18: 457–472.
- ISBN 978-0-07-074175-1.
- PMID 12080085.
- ^ ISBN 0716746840. Retrieved 27 May 2021.
- PMID 14705871.
- PMID 24139043.
- PMID 30996100.
- PMID 9878760.
- PMID 10600722.
- PMID 16584073.




