Aspergillus fumigatus
| Aspergillus fumigatus | |
|---|---|

| |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Fungi |
| Division: | Ascomycota |
| Class: | Eurotiomycetes |
| Order: | Eurotiales |
| Family: | Aspergillaceae |
| Genus: | Aspergillus |
| Species: | A. fumigatus
|
| Binomial name | |
| Aspergillus fumigatus Fresenius 1863
| |
| Synonyms | |
|
Neosartorya fumigata | |
Aspergillus fumigatus is a species of fungus in the genus Aspergillus, and is one of the most common Aspergillus species to cause disease in individuals with an immunodeficiency.
Aspergillus fumigatus, a
The fungus is capable of growth at 37 °C or 99 °F (
When the fermentation broth of A. fumigatus was screened, a number of indolic alkaloids with antimitotic properties were discovered.[6] The compounds of interest have been of a class known as tryprostatins, with spirotryprostatin B being of special interest as an anticancer drug.
Aspergillus fumigatus grown on certain building materials can produce
Genome
Aspergillus fumigatus has a stable
Pathogenesis
Aspergillus fumigatus is the most frequent cause of invasive fungal infection in immunosuppressed individuals, which include patients receiving immunosuppressive therapy for autoimmune or neoplastic disease, organ transplant recipients, and AIDS patients.[11] A. fumigatus primarily causes invasive infection in the lung and represents a major cause of morbidity and mortality in these individuals.[12] Additionally, A. fumigatus can cause chronic pulmonary infections, allergic bronchopulmonary aspergillosis, or allergic disease in immunocompetent hosts.[13]
Innate immune response
Inhalational exposure to airborne conidia is continuous due to their ubiquitous distribution in the environment. However, in healthy individuals, the innate immune system is an efficacious barrier to A. fumigatus infection.
Invasion

Immunosuppressed individuals are susceptible to invasive A. fumigatus infection, which most commonly manifests as invasive pulmonary aspergillosis. Inhaled conidia that evade host immune destruction are the progenitors of invasive disease. These conidia emerge from dormancy and make a morphological switch to hyphae by germinating in the warm, moist, nutrient-rich environment of the pulmonary alveoli.
Hypoxia response
As is common with tumor cells and other pathogens, the invasive hyphae of A. fumigatus encounters hypoxic (low oxygen levels, ≤ 1%) micro-environments at the site of infection in the host organism.[17][18][19] Current research suggests that upon infection, necrosis and inflammation cause tissue damage which decreases available oxygen concentrations due to a local reduction in perfusion, the passaging of fluids to organs. In A. fumigatus specifically, secondary metabolites have been found to inhibit the development of new blood vessels leading to tissue damage, the inhibition of tissue repair, and ultimately localized hypoxic micro-environments.[18] The exact implications of hypoxia on fungal pathogenesis is currently unknown, however these low oxygen environments have long been associated with negative clinical outcomes. Due to the significant correlations identified between hypoxia, fungal infections, and negative clinical outcomes, the mechanisms by which A. fumigatus adapts in hypoxia is a growing area of focus for novel drug targets.
Two highly characterized sterol-regulatory element binding proteins, SrbA and SrbB, along with their processing pathways, have been shown to impact the fitness of A. fumigatus in hypoxic conditions. The transcription factor SrbA is the master regulator in the fungal response to hypoxia in vivo and is essential in many biological processes including iron homeostasis, antifungal azole drug resistance, and virulence.[20] Consequently, the loss of SrbA results in an inability for A. fumigatus to grow in low iron conditions, a higher sensitivity to anti-fungal azole drugs, and a complete loss of virulence in IPA (invasive pulmonary aspergillosis) mouse models.[21] SrbA knockout mutants do not show any signs of in vitro growth in low oxygen, which is thought to be associated with the attenuated virulence. SrbA functionality in hypoxia is dependent upon an upstream cleavage process carried out by the proteins RbdB, SppA, and Dsc A-E.[22][23][24] SrbA is cleaved from an endoplasmic reticulum residing 1015 amino acid precursor protein to a 381 amino acid functional form. The loss of any of the above SrbA processing proteins results in a dysfunctional copy of SrbA and a subsequent loss of in vitro growth in hypoxia as well as attenuated virulence. Chromatin immunoprecipitation studies with the SrbA protein led to the identification of a second hypoxia regulator, SrbB.[21] Although little is known about the processing of SrbB, this transcription factor has also shown to be a key player in virulence and the fungal hypoxia response.[21] Similar to SrbA, a SrbB knockout mutant resulted in a loss of virulence, however, there was no heightened sensitivity towards antifungal drugs nor a complete loss of growth under hypoxic conditions (50% reduction in SrbB rather than 100% reduction in SrbA).[21][20] In summary, both SrbA and SrbB have shown to be critical in the adaptation of A. fumigatus in the mammalian host.
Nutrient acquisition
Aspergillus fumigatus must acquire nutrients from its external environment to survive and flourish within its host. Many of the genes involved in such processes have been shown to impact virulence through experiments involving genetic mutation. Examples of nutrient uptake include that of metals, nitrogen, and macromolecules such as peptides.[12][25]

Iron acquisition
Iron is a necessary
Nitrogen assimilation
Aspergillus fumigatus can survive on a variety of different
Proteinases
The human lung contains large quantities of
Unfolded protein response
A number of studies found that the unfolded protein response contributes to virulence of A. fumigatus.[39]
Secondary metabolism
Secondary metabolites in fungal development

The lifecycle of filamentous fungi including Aspergillus spp. consists of two phases: a
Gliotoxin
Gliotoxin is a mycotoxin capable of altering host defenses through immunosuppression. Neutrophils are the principal targets of gliotoxin.[46][47] Gliotoxin interrupts the function of leukocytes by inhibiting migration and superoxide production and causes apoptosis in macrophages.[48] Gliotoxin disrupts the proinflammatory response through inhibition of NF-κB.[49]
Transcriptional regulation of gliotoxin
LaeA and GliZ are transcription factors known to regulate the production of gliotoxin. LaeA is a universal regulator of secondary metabolite production in Aspergillus spp.
Current treatments to combat A. fumigatus infections
Current noninvasive treatments used to combat fungal infections consist of a class of drugs known as azoles.
Gallery
-
Conidia phialoconidia of A. fumigatus
-
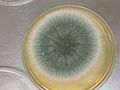
Colony in Petri dish
-
A. fumigatus isolated from woodland soil
-
Slide of an infected turkey brain
See also
- 2012 US meningitis outbreak
- Allergic bronchopulmonary aspergillosis
- Aspergilloma
- Diseases of the honeybee
- Fumagillin
- RodA
- Galactosaminogalactan
References
- PMID 30066670.
- S2CID 4371721.
- PMID 16607012.
- PMID 28203225.
- PMID 20974273.
- PMID 8823522.
- PMID 12324333.
- PMID 16372000.
- ^ PMID 16372009.
- PMID 16372010.
- ^ S2CID 28216163.
- ^ PMID 17890370.
- ^ PMID 19403905.
- ^ PMID 17196036.
- PMID 34781737.
- PMID 28968869.
- PMID 19462332.
- ^ PMID 22447924.
- PMID 20560863.
- ^ PMID 18989462.
- ^ PMID 25375670.
- PMID 27303716.
- PMID 26822492.
- PMID 23104569.
- ^ PMID 19597008.
- ^ PMID 17845073.
- S2CID 10989925.
- ^ PMID 15504822.
- PMID 16113265.
- ^ S2CID 27753283.
- PMID 12135576.
- PMID 6150137.
- PMID 6360904.
- PMID 11980964.
- PMID 2258912.
- PMID 8188335.
- PMID 7558282.
- PMID 9635248.
- PMID 22028661.
- ^ PMID 15075281.
- PMID 12208999.
- PMID 20966095.
- PMID 10066549.
- PMID 20507448.
- PMID 7773383.
- PMID 18199036.
- ^ PMID 17030582.
- PMID 16110799.
- PMID 15817772.
- ^ PMID 17432932.
- PMID 25328449.
- PMID 28638374.
- PMID 20729241.
- PMID 28992260.
- PMID 20385860.
- PMID 30940706.
- PMID 23226235.
- PMID 31969561.
External links
- Emergence of Azole Resistance in Aspergillus fumigatus and Spread of a Single Resistance Mechanism. at SciVee
- The Aspergillus Trust A registered UK charity engaged in support of people with aspergillus disease worldwide and research into cures
- The Fungal Research Trust
- Aspergillus info from DoctorFungus.org